3.1.3.1 ionic bonding
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
what is ionic bonding?
the strong electrostatic force of attraction between the oppositely charged ions in a lattice
explain what will happen to metals during ionic bonding
metals lose electrons when bonded to non-metals in order to achieve the nearest noble gas electronic configuration
the metal is left as a positive ion
explain what will happen to non-metals during ionic bonding
non-metals gain electrons in order to achieve the nearest noble gas electronic configuration
the non-metal is left as a negative ion
in ionic bonding, the metal _______ its electrons to the non-metal which results in what?
transfers
so the stability of each atom has now increased
there is now a strong electrostatic force of attraction between the oppositely charged ions
draw a dot + cross diagram for NaF
*outer shells are only shown
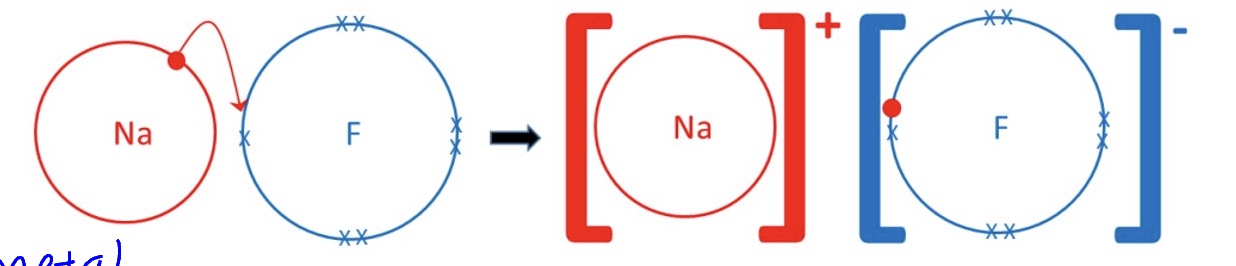
however the + and - charges do not just point in one direction, explain what they do instead + what this results in
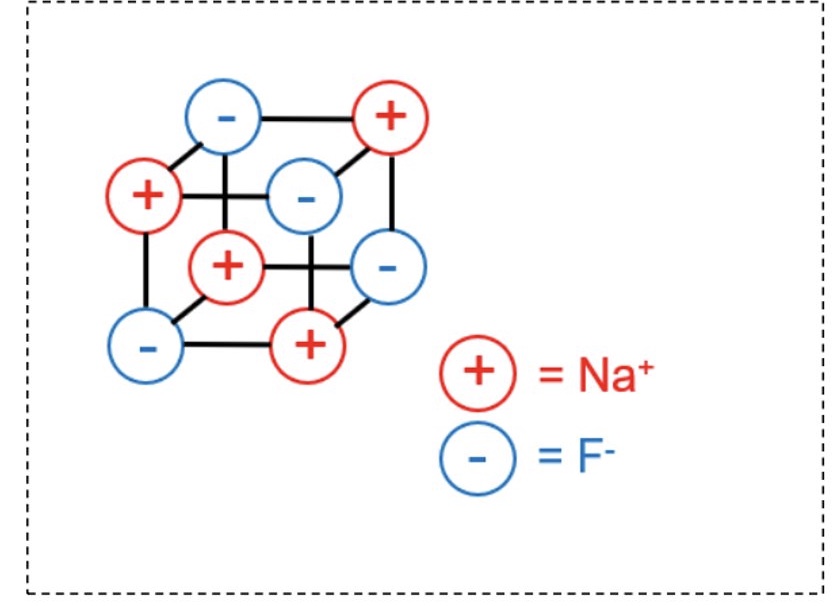
they span out to create a giant 3D structure where there is a constant state of repulsion between ions with the same charge + attraction between ions of opposite charge
as a result, all the different forces find a balance as the ions form a giant ionic lattice
draw the structure of a general giant ionic lattice
eg for NaF
give 3 properties of ionic compounds
they are always solids at room temperatures
they will not conduct electricity in the solid state but will in the molten state
they are brittle + will shatter upon impact
explain why ionic compounds are solids at room temp
they have a giant ionic lattice structure + their melting + boiling points are very high
this is due to the strong electrostatic attraction between the oppositely charged ions
so lots of energy is required to overcome the forces
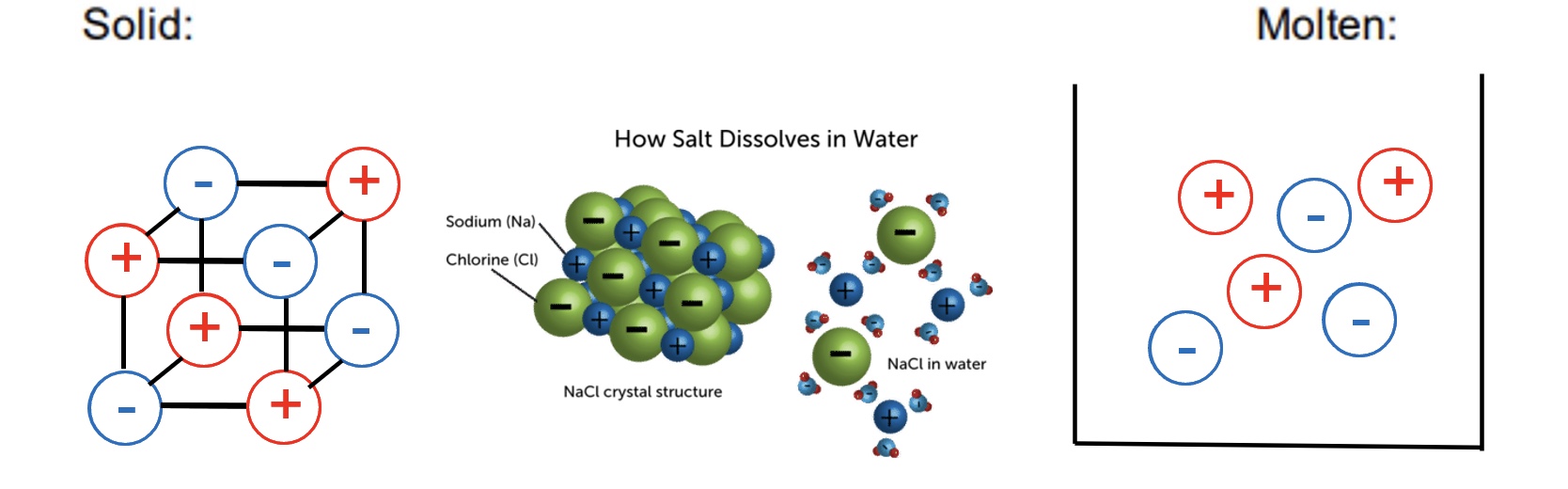
explain why ionic compounds will not conduct electricity in the solid state but will when molten
solid → ions are not free to move or flow so cannot carry a charge
when molten or dissolved in water → ions are free to move/flow so are able to carry a charge
why are ionic compounds brittle + will shatter upon impact?
upon impact, the like charges will line up + repel, this repulsion results in the shattering
give 2 factors that increase the strength of ionic bonds
the greater the ion charge, the stronger the bond
the smaller the ionic radius, the stronger the bond
state which groups in the periodic table with become cations and which will become anions when reacting
groups 1, 2, 3 → form cations (+, +2, +3)
groups 5, 6, 7 → form anions (-3, -2, -)
(the charge is determined from which group the element is in)
which groups will not tend to form ions and why?
groups 4 + 8
elements in group 8 are energetically stable, they have fully filled shells/orbitals
elements in group 4 do not because they would have to either lose 4 electrons or gain 4 → they are relatively stable
what type of ions do the transition metals form?
atoms of these elements can lose different numbers of electrons depending on what they are bonding to and what the environment is → so they can form a variety of ions
what tells us the charge on the ions of transition metals?
these ions will have a roman numeral after the name eg iron(III) showing us that its Fe³⁺
how do you construct formulas for ionic compounds?
you have to balance the charges of the cation and the anion so the negative + positive charges cancel out
eg magnesium chloride → Mg²⁺ Cl⁻
therefore 2 CL are needed to balance the Mg charge
an easier way to do is is to just swap the charges over
give the formulas of the compound ions: hydroxide, nitrate, carbonate, sulfate + ammonium
hydroxide → OH⁻
nitrate → NO₃⁻
carbonate → CO₃²⁻
sulfate → SO₄²⁻
ammonium → NH₄⁺