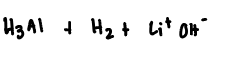Reducing Benzil using Sodium Borohydride
1/42
Earn XP
Description and Tags
OCHEM 2 Lab Final
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
Reduction
hydrogen is added to or oxygen is lost from an organic molecule
Ex: LiAlH4 + NaBH4
LiAlH4
reduces variety of compounds like carboxylic acids, esters, nitriles, amides, aldehydes and ketone
hydrogen is more electronegative than Li —> reacts violently with protic solvents ( water or alcohols to form flammable H gas)
must be used in inert, anhydrous solvent ( diethyl or tetrahydrofuran)
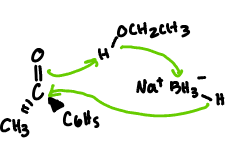
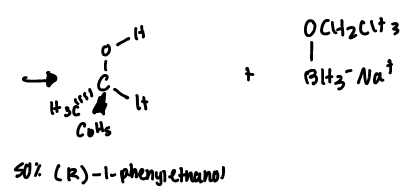
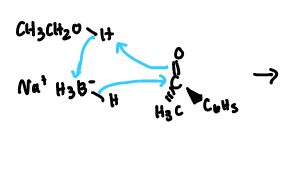
NaBH4
reduces only aldehydes and ketones
easier to use b/c reacts slowly with protic solvents (can use OH)
decomposes in acidic functional groups
first acidic groups neutralized with base (NaOH) before aldehyde and ketone can be reduced
one molecule is capable of reducing up to four molecules of a ketone that contains a single carbonyl group
acq. acid is used to free the reduce acid from the complex mixture
Enantiomers
non-superimposable mirror image stereoisomers
Racemic mixture
mixture of enantiomers
Vicinal diols
alcohols containing two OH groups attached to adjacent carbon atoms
Meso compound
a molecule that is optically inactive even tho it contains one or more chiral center
Diastereomers
stereoisomers that are not mirror images
What happens when two different compounds with the same melting point are mixed together?
the resulting mixture has a lower + broader melting point range
What are the reactions?
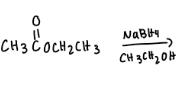

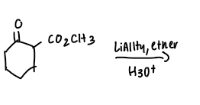
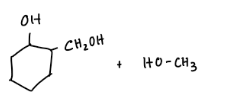
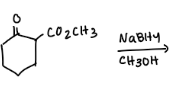
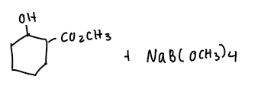
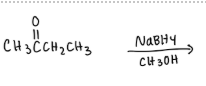
Explain why one molecule of NaBH4 will reduce only two molecules of m-acetylbenzaldehyde?
One molecule of NaBH4 will reduce only two molecules of m-acetyl benzaldehyde because NaBH4 only has four hydride ions, and each m-acetyl benzaldehyde has two functional groups, one ketone and one aldehyde.
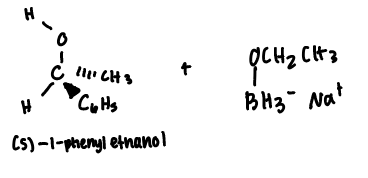
Predict the stereochemical outcome of the following reaction?
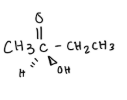
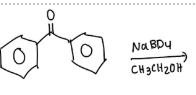
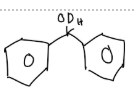
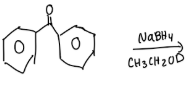
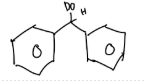
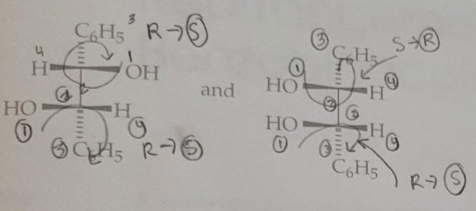

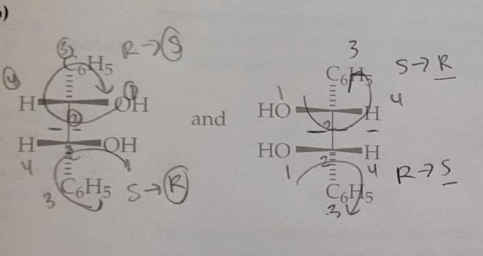

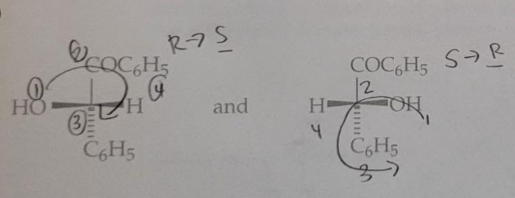

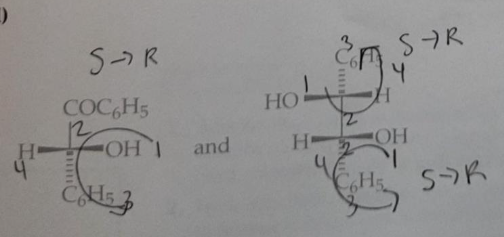

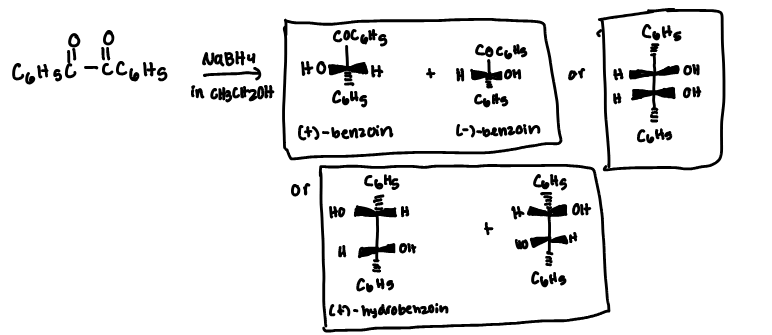
Calculate the theoretical mass of NaBH4 needed to reduce 100 mg of benzil to (+)-benzoin.
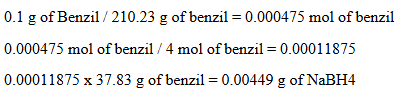
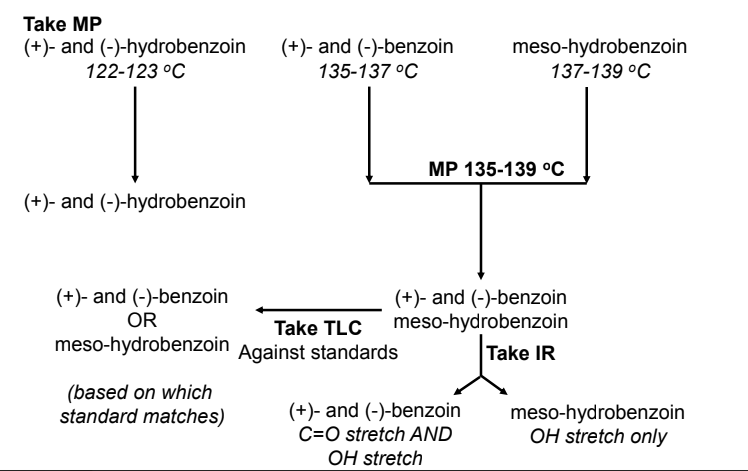
How can you melting point to determine the identity of your product?
The melting point can be used to determine the product because the melting point of each product is identifiable.
Racemic hydrobenzoin has a melting point range of 122-123 °C.
Racemic benzoin has a melting point range of 135-137 °C.
Meso hydrobenzoin has a melting point range of 137-139°C.
When taking the melting point of the product, a comparison of the melting point and those listed can be done.
How can you use TLC to determine the identity of your product?
Doing the TLC of the product can assess the polarity of the product formed along with the expected products as meso hydrobenzoin has a higher polarity than racemic hydrobenzoin
Racemic benzoin has lower polarity in comparison, as it only has one OH group.
How can you use IR Spec to determine the identity of your product?
If there is a strong carbonyl present in the 1600-1700 cm-1 range, the product is most likely racemic benzoin due to its carbonyl
A broad range around 3300 cm-1 indicates a strong OH group is present, and with the absence of a carbonyl peak, it can indicate that the product might be meso hydrobenzoin.
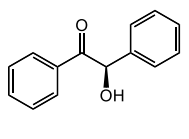
(+/) benzoin
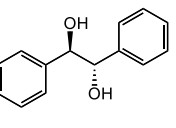
meso-hydrobenzoin
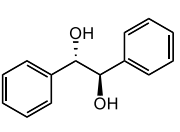
(-) hydrobenzoin
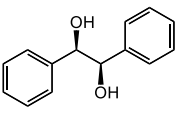
(+) hydrobenzoin
flowchart
Benzil hazard
Irritant
Prevent eye, skin and clothing contact
Avoid inhaling the vapors and ingesting the compound
Ethanol, ethyl acetate, hexane: hazard
Flammable fire hazards.
Potassium bromide hazard
Irritant, hygroscopic (sensitive to contact with water)
Prevent eye, skin and clothing contact. Leave capped.
Sodium borohydride hazard
Flammable and corrosive. Avoid eye, skin, and clothing contact. Leave capped when not in use.
Steps
Heat benzil in ethanol
cool to room temp + ice bath
add NaBH4 in 8 portions over 2 steps (let stand for 10 min)
add hot water + mixture heated to boiling
filter ( if not clear) + add more water
vacuum filtration
melting point + tlc
Why do we heat the benzil in ethanol?
it acts as a solvent
cooled to room temp to recrystallize
Why do we add NaBH4 in 8 portions in 2 minutes while the flask is in the ice bath?
prevents reaction from overheating or becoming too vigorous
swirling after each time makes sure its well mixed
allow the mixture to stand for 10 minutes so the reaction can be completed
Why is hot water added to the mixture and heated to boiling?
helps destroy any remaining intermediates and ensures all compounds are dissolved
filter to remove impurities
add more water ( anti-solvent ) step
organic product is less soluble than in ethanol - product is forced to precipitate out of the solution
TLC reagents
Eluent: 2:1 hexane: ethyl acetate
For spotting: 1-2 mg in 2-3 drop ethyl acetate
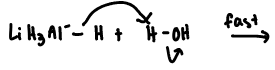
li reacts violently side reaction