The Periodic Table - Chp 4 nf
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Pauli Exclusion Principle
states that no more than two electrons may occupy an orbital and they must have opposite spin
Hund's Rule of Maximum Multiplicity
states tha when two or more orbitals of equal energy are available, the electrons occupy them singly before filling them in pairs
Aufbau Principle
when building up the electron configuration of an atom in its ground state, the electrons occupy the lowest available energy levels
Isotopes
are atoms of the same element (ie they have the same atomic number) which have difference mass numbers due to the different number of neutrons in the nucleus
Relative atomic mass
defined as
- the average of the mass numbers of the isotopes of the element
- as they occur naturally.
- Taking their abundances into account and
- expressed on a scale in which the atoms of the carbon-12 isotope have a mass of exactly 12 units
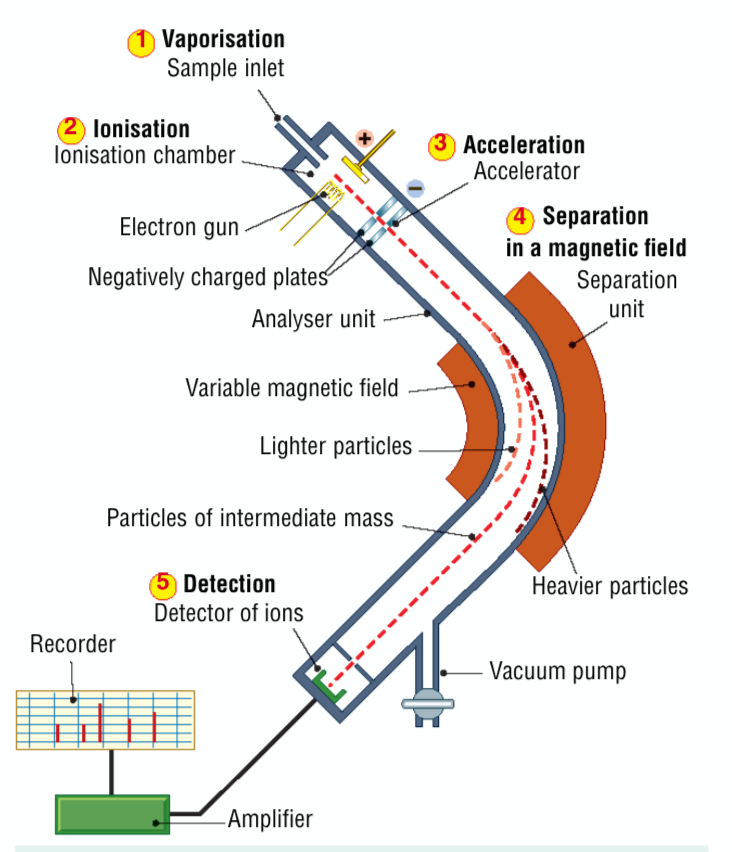
What 5 processes occur in a mass spectrometer?
- vaporisation - the sample is turned into a gas
- ionisation - an electron gun fires high speed electrons at the gas sample and creates positive ions
- acceleration - the positive ions are attracted towards a negatively charged plate
- separation - the positive ions travelling at speed are separated by passing them through a strong magnetic field, lighter particles are more affected than heavy particles
- detection - the number of ions of each mass are detected
What is mass spectrometry used for?
- drug testing atheletes
- measure relative atomic masses and relative molecular mass
- measure relative abundance of isotopes
- identify unknown compounds
Mass spectrometer diagram
What did Dobereiner do?
- Arranged the elements with similar properties into triads
- Eg Cl, Br, I
What did Newlands do?
- arranged the elements in order of relative atomic mass, forming Newlands' octaves
- only worked for the first 16 elements
What did Mendeleef do?
- arranged the elements in order of increasing atomic weight placing those with similar properties in groups
- he left gaps as he correctly predicted that there were some other elements not yet discovered
- he reversed the order of I and Te as he knew that their properties meant that they were better suited to those groups, he did not fully understand why but it was proven to be correct by Mosley
What did Moseley do?
arranged the elements in order of increasing atomic number keeping those with similar properties underneath each other
How did Moseley determine the atomic number of an element?
he studied the frequencies of the X-rays emitted by atoms of different elements
found that frequencies varied with the quantity of positive charge in the nucleus
What are the differences between Mendeleef's and the modern Periodic Table?
- elements are now arranged in order of atomic number rather than atomic weight
- separate block for transition metals
- no gaps in the modern periodic table
- more elements (man-made) on the modern table
The modern Periodic Table
an arrangement of elements in order of increasing atomic number
Modern Periodic Law
when elements are arranged in order of increasing atomic number, the properties of the elements recur periodically, ie. the properties displayed by an element are repeated at regular intervals in other elements
Atomic number
of an atom is the number of protons in the nucleus of that atom
Mendeleev's Periodic Law
when elements are arranged in order of increasing atomic weight (relative atomic mass), the properties of the elements recur periodically, ie. the properties displayed by an element are repeated at regular intervals in other elements
Newlands' Octaves
Arrangements of elements in which the first and the eighth element, counting from a particular element, have similar properties
Triad
is a group of three elements with similar chemical properties, arranged in order of increasing atomic mass, where the atomic weight (relative atomic mass) of the middle element is approximately equal to the average of the other two
Element
a substance that cannot be split into simpler substances by chemical means
Mass number
of an element is the sum of the number of protons and neutrons in the nucleus of an atom of that element
Principle of Mass spectrometry
charged particles moving in a magnetic field
are deflected to different extents according to their masses and
are thus separated according to these masses
Atom
the smalles particle of an element that can exist independently while retaining the properties of that element
Compound
a substance made up of two or more substances chemically combined
Element
a substance that cannot be split into simpler substances by chemical means