18. Properties of period 3 elements and their oxides
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
Describe the general trend for first ionisation energy across a period.
Increases. HOWEVER:
● Dip between Group 2 and 3, as electrons start to fill the p subshell.
● Dip between Group 5 and Group 6, as pairs of electrons start to fill the same orbital.
How many atoms of the following can bond together:
P
S
Cl
What about Si?
P₄
S₈
Cl₂
Si can form macromolecules of unlimited size; giant covalent lattice
Order the melting points of the period 3 melting points.
Si>S>P>Cl>Ar
To some extent, we can generalise the trends we have seen in Period 3 to other periods.
For example, we can generalise the trends in...
Atomic radius
First ionisation energy
NOT melting point!
Appearance and physical properties of sodium?
Metallic, shiny, conducts electricity (worst conductor out of the metals)
Reaction of sodium with acid?
Na + acid → H₂ + salt
Which elements react with cold or room temp water?
Na
Mg
Cl₂
ALL redox reactions.
Reaction of sodium with cold water - equation and observations?
Vigorous reaction, floats on water, fizzes, melts due to -ΔH of the reaction.
NaOH is pH 13-14.
2Na (s) + 2H₂O(l) → 2NaOH (aq)+ H₂ (g)
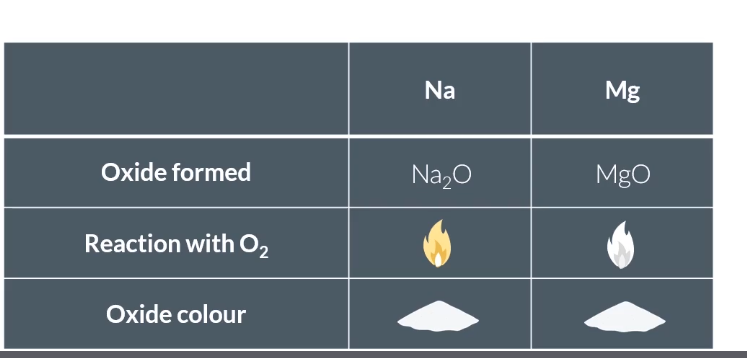
Reaction of sodium with oxygen (equation and observations)?
Bright yellow flame, forms white powder of Na₂O.
2Na + ½ O₂ → Na₂O
4Na + O₂ → 2Na₂O
Why might we observe some yellow powder instead of white powder when sodium reacts with oxygen?
Some Na₂O₂ is formed
Appearance and physical properties of magnesium?
Metallic, shiny, conducts electricity (between Na and Al in terms of how well it does).
Reaction of Mg with cold water (observations and equation)?
Very slow reaction. pH = 10 as Mg(OH)₂ is sparingly soluble. Mg + 2H₂O → H₂ + Mg(OH)₂
Reaction of Mg with steam (observations and equation)?
Much faster.
Mg (s) + H₂O (g) → MgO (s) + H₂ (g)
Reaction of Mg with oxygen (observations and equation)?
Bright white flame, forms white powder of MgO.
2Mg + O₂ → 2MgO
Does sodium or magnesium form less alkaline solutions?
Mg(OH)₂ as it is has a lower solubility then NaOH
Appearance and physical properties of aluminium?
Metallic, shiny, best conductor of electricity in period 3
Reaction of Al with cold water?
No reaction
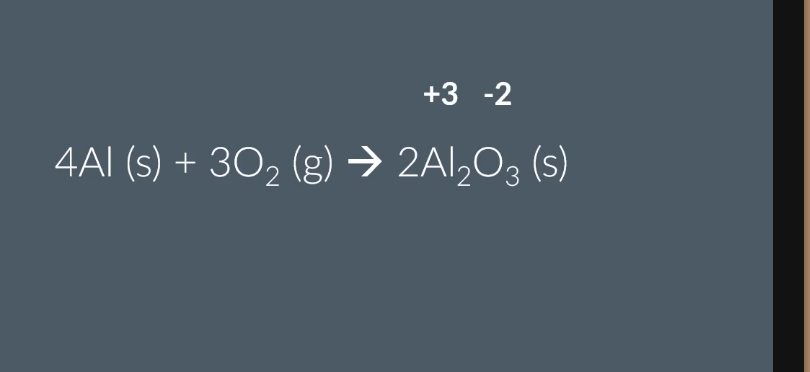
Reaction of Al with oxygen (equation and observations)?
Heat and lower into jar of O₂ → bright flame, forms white powder.
4Al (s) + 3O₂ (g) → 2Al₂O₃ (s)
Why is aluminium considered unreactive even though the metal itself is reactive?
Covered by Al₂O₃ from where oxygen has oxidised the surface of the Al.
Al₂O₃ is unreactive.
Makes it useful for saucepans, window frames etc.
If Al₂O₃ is scratched off, Al reacts rapidly with air to form a new coat.
Appearance and physical properties of silicon?
Semimetal (metalloid), semiconductor (conducts electricity to an extent and in certain conditions)
Reaction of silicon with oxygen (observations and equation)?
Heat strongly.
Si + O₂ → SiO₂ (white powder)
Appearance and physical properties of phosphorus?
Non-metal, low m.p. And b.p., does not conduct electricity.
Red Phosphorus as a crystal structure White Phosphorus as P4
Reaction of phosphorous in excess oxygen (observations and equation)?
Red P needs to be heated significantly first, white P ignites spontaneously in air
Phosphorus pentoxide (white crystalline solid) formed
4P + 5O₂ → P₄O₁₀
Reaction of phosphorous in limited oxygen (observations and equation)?
Incomplete combustion: colourless liquid phosphorus trioxide is formed.
4P + 3O₂ → P₄O₆
Appearance and physical properties of sulfur?
Non-metal, low mp and bp, does not conduct electricity.
Reaction of sulfur with oxygen (observations and equation)?
Need to heat and lower into a jar of oxygen. Colourless gas sulfur dioxide formed. Some SO₃ also formed.
S + O₂ → SO₂
Give the formula of the highest oxide of sulfur.
Give the formula of the most common oxide of sulfur.
SO₃
SO₂
(a) How are the atoms in a molecule of SO₂ held together?
(b) What forces hold the molecules of SO₂ together?
(a) Covalent bonds
(b) Van der Waals forces + Dipole-dipole forces
What are the different types of structure and bonding displayed by the period 3 oxides?
Na - ionic lattice
Mg - ionic lattice
Al - ionic lattice with covalent character; Al³⁺ distorts O²⁻ electron cloud
Si - giant covalent
P - simple molecular covalent
S - simple molecular covalent
Explain how the covalent character of aluminium oxide arises.
● The aluminium cation is very small so is closer to the oxide ion.
● The cation is also highly charged enough to distort the electron cloud.
● So the electron cloud appears more covalent.
Draw a table giving the bonding, structure and relative melting points of the period 3 oxides.
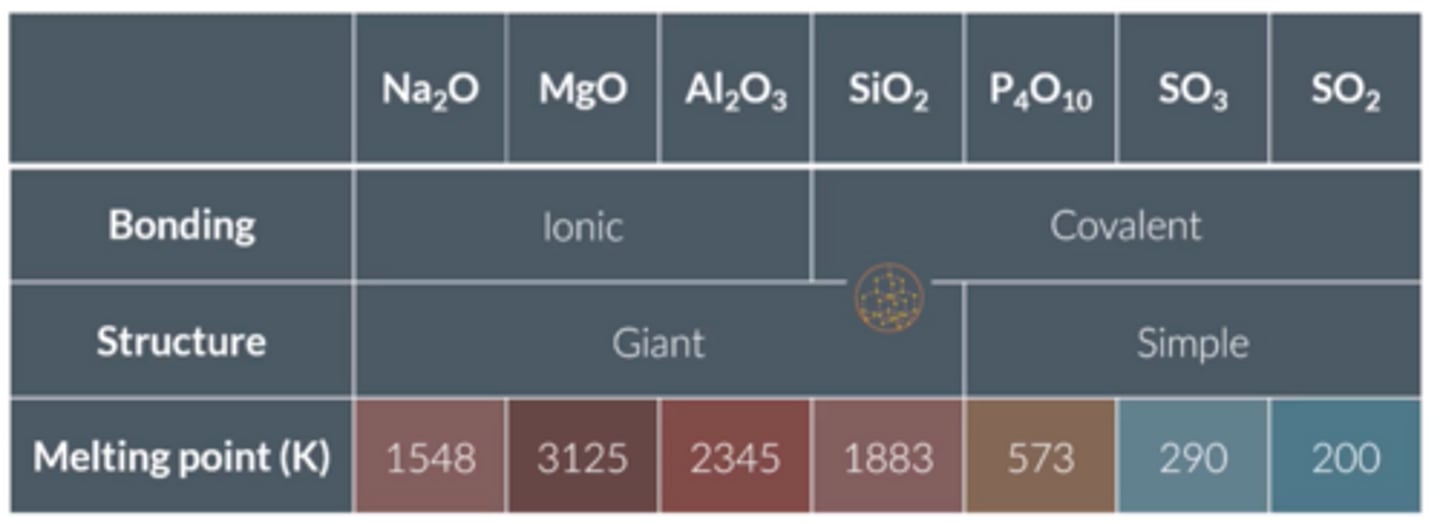
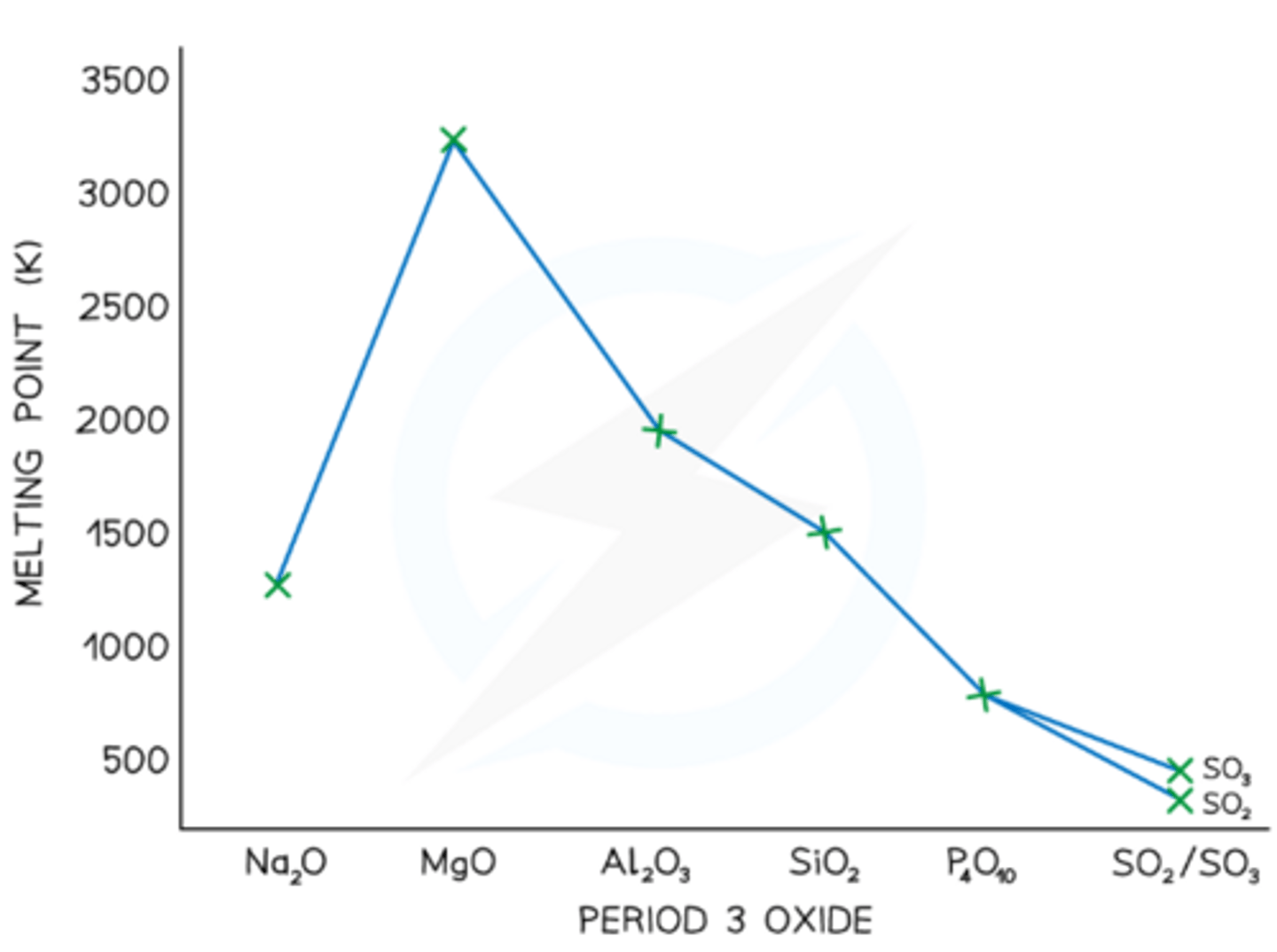
What is the trend in melting points for period 3 oxides (state highest and lowest and why and draw graph)?
● Highest is MgO, lowest is SO₂;
● Increases along group for metals for Na₂O and MgO due to stronger ionic bonding, but Al₂O₃ is lower due to the covalent character of the bonding.
● Decreases with size for simple molecular molecules, weaker van der waals: P₄O₁₀>SO₃>SO₂
Which metal oxides are insoluble in water?
SiO₂ and Al₂O₃
because of the high strength of the Al₂O₃ ionic lattice and the SiO₂ macromolecular structure, so they give a neutral pH 7
Which period 3 oxides are basic?
Na₂O and MgO
Reaction of Na oxide with water? pH?
Na₂O + H₂O → 2Na⁺ + 2OH⁻
pH = 14
Reaction of Mg oxide with water? pH?
MgO + H₂O → Mg²⁺ + 2OH⁻ ⇌ Mg(OH)₂
pH = 9 as Mg(OH)₂ is sparingly soluble
Give an equation for the reaction between magnesium oxide and sulfuric acid.
Include state symbols.
MgO (s) + H₂SO₄(aq) → MgSO₄(aq) + H₂O(l)
What are the products if Na₂O and MgO are reacted with acid?
Salt and water only
Is aluminium oxide acidic or basic?
It is amphoteric; can act as either an acid or a base
Is aluminium oxide soluble?
Not in water
Reaction of aluminium oxide with HCI?
Al₂O₃ + 6HCl → 3H₂O + 2AlCl₃
Reaction of aluminium oxide with NaOH?
Al₂O₃ + 2NaOH + 3H₂O → 2NaAl(OH)₄;
Sodium aluminate is formed
Is silicon dioxide soluble in water?
No
In what conditions will silicon dioxide act as an acid?
Reacts as a weak acid with a strong base (e.g. hot, conc NaOH)
Reaction of silicon dioxide with hot, conc NaOH?
Give full and ionic equation.
SiO₂ + 2NaOH → Na₂SiO₃ + H₂O
sodium silicate
SiO₂ + 2OH⁻ →SiO₃²⁻ + H₂O
How is silicon dioxide used in the production of Fe?
SiO₂ + CaO → CaSiO₃
calcium silicate
Give the reaction of phosphorus pentoxide with water.
pH?
P₄O₁₀ + 6H₂O → 4H₃PO₄
pH = 2
Give the reaction of phosphorus pentoxide with NaOH?
Give the full and ionic equation.
P₄O₁₀ + 12NaOH → 4Na₃PO₄ + 6H₂O
P₄O₁₀ + 12OH⁻ → 4PO₄³⁻ + 6H₂O
Dissociation of the acid formed (H₃PO₄)?
H₃PO₄ → H⁺ + H₂PO₄⁻
Reaction of P₄O₁₀ with NaOH?
H₃PO₄ + 3NaOH → Na₃PO₄ + 3H₂O
Reaction of SO₂ with water? pH?
SO₂ + H₂O → H₂SO₃
weak acid → pH = 2-3
Reaction of SO₃ with water? pH?
SO₃ + H₂O → H₂SO₄
strong acid → pH = 0-1
Reaction of SO₂ with NaOH? (2 stages)
SO₂ + NaOH → NaHSO₃
NaHSO₃ + NaOH → Na₂SO₃ + H₂O
How can flue gases be removed by CaO?
CaO + SO₂ → CaSO₃ (calcium sulfite)
Draw the structure of H₃PO₄
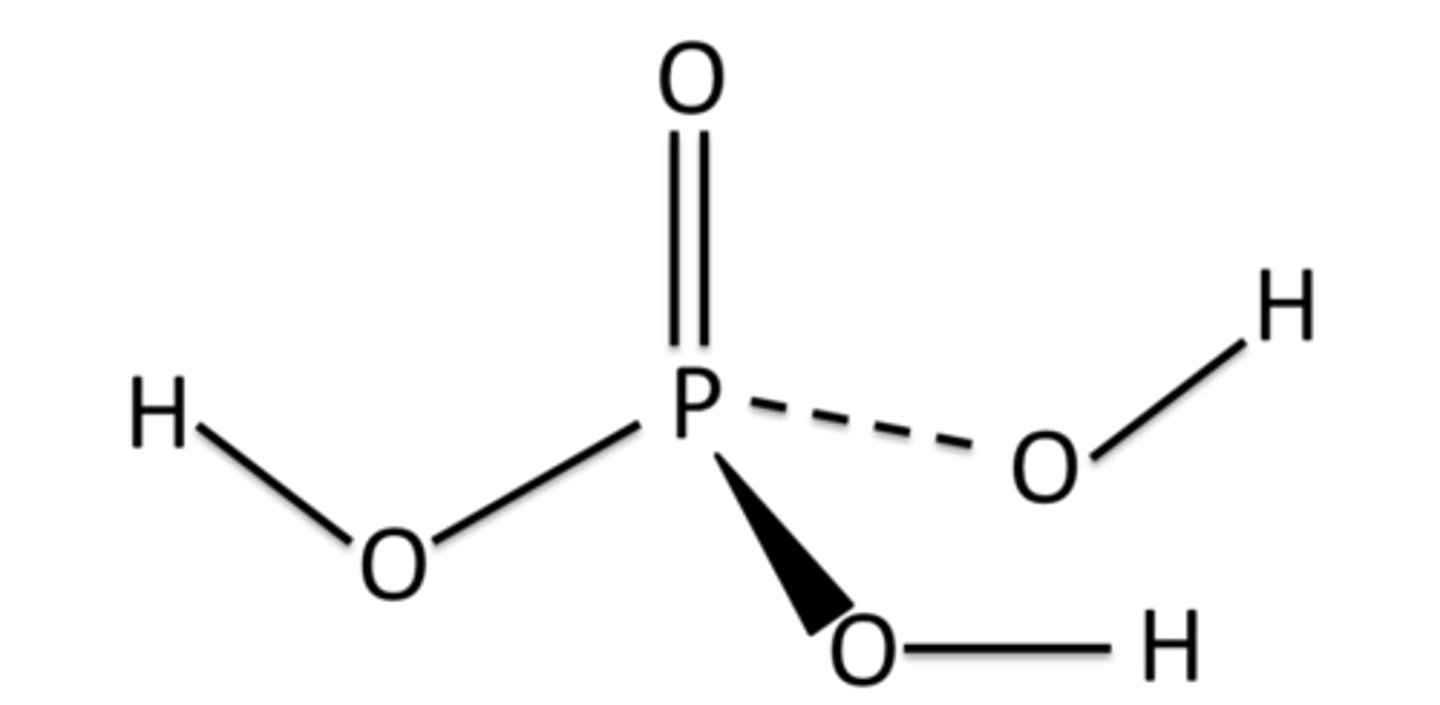
How many electrons does P have in its outer shell in Н₃PO₄?
10
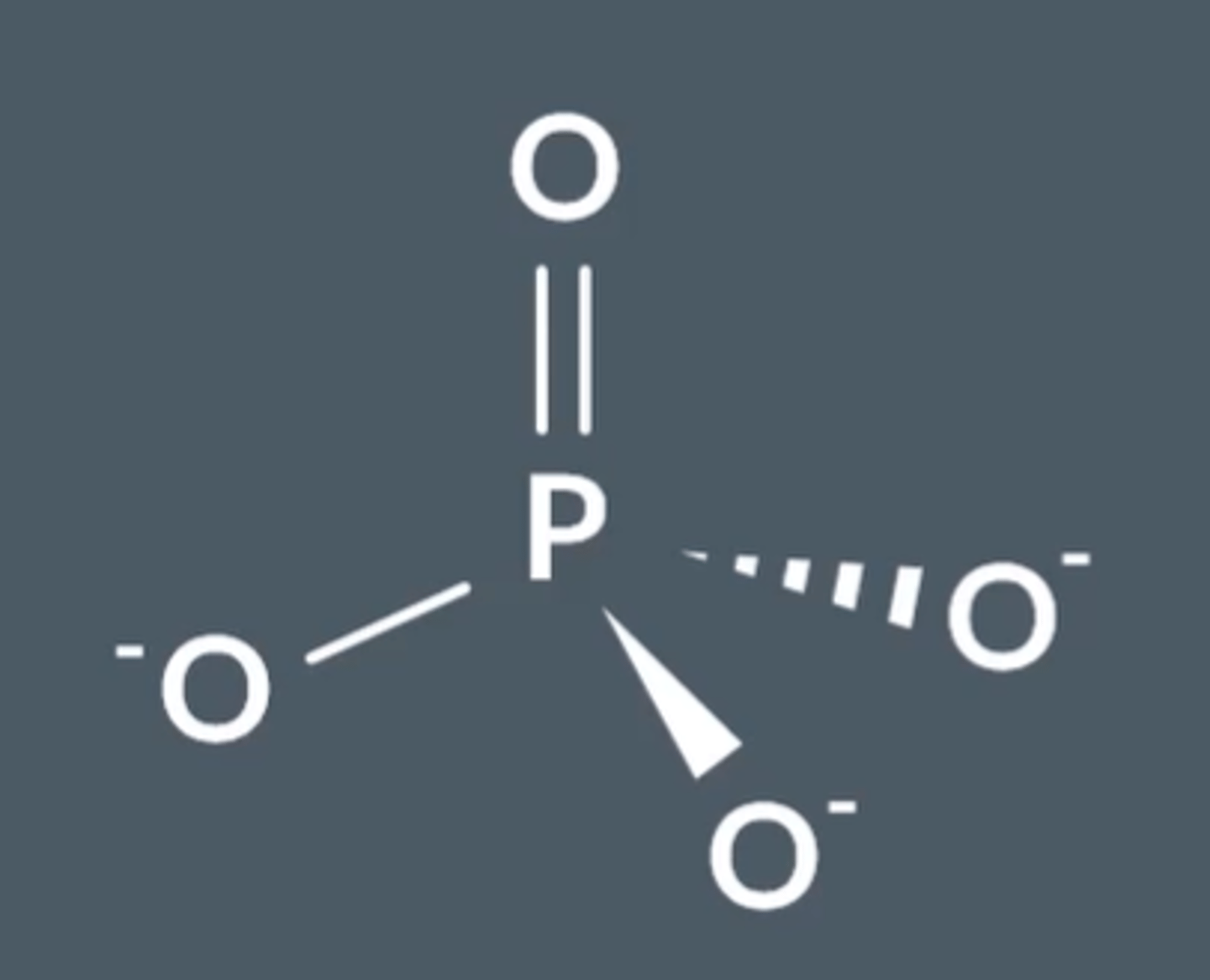
What is the shape and bonding in PO₄³⁻ ?
Electrons delocalise to give tetrahedral structure with 109.5° bond angle.
Each P-O bond is the same length
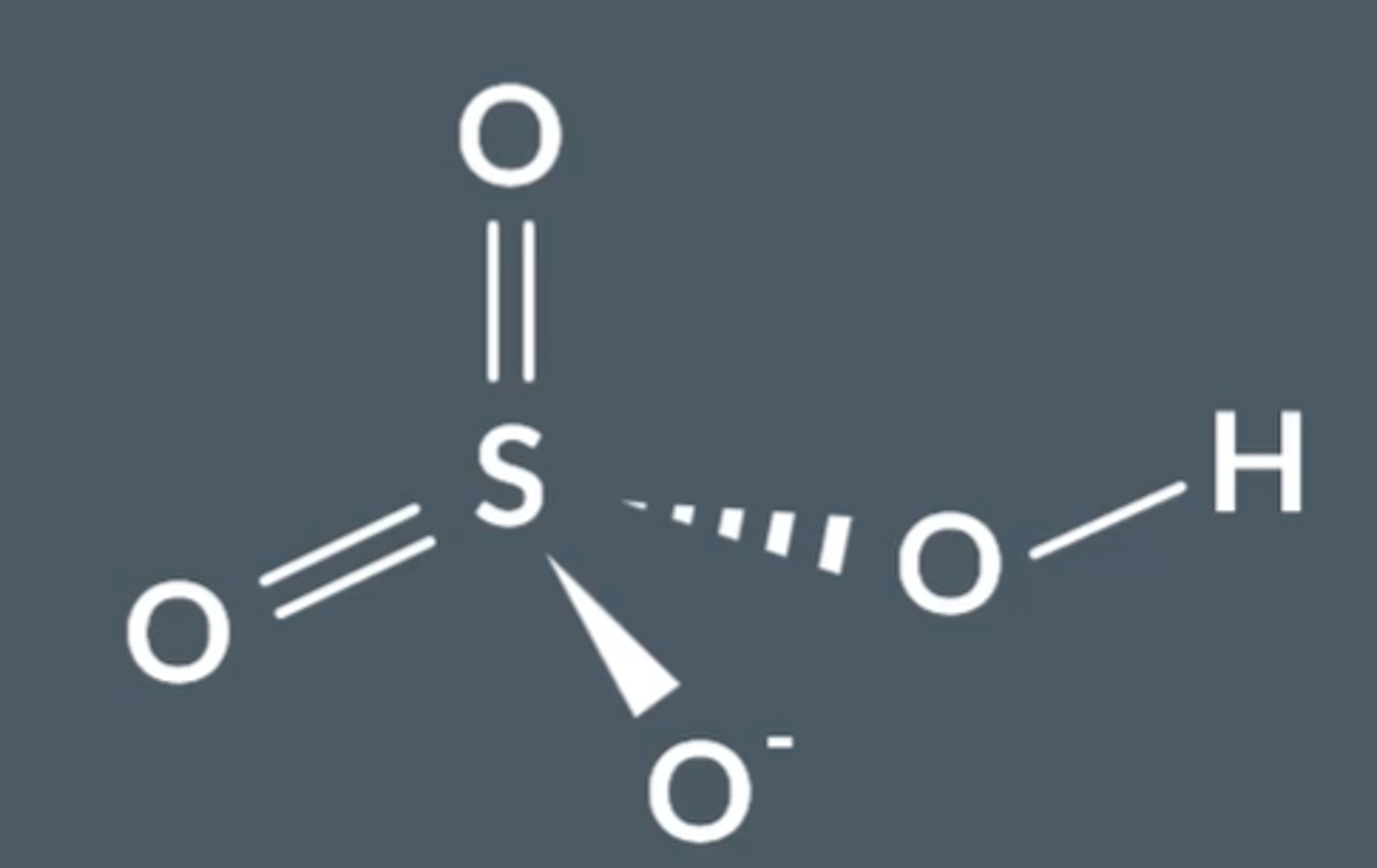
Draw the structure and bonding of H₂SO₄
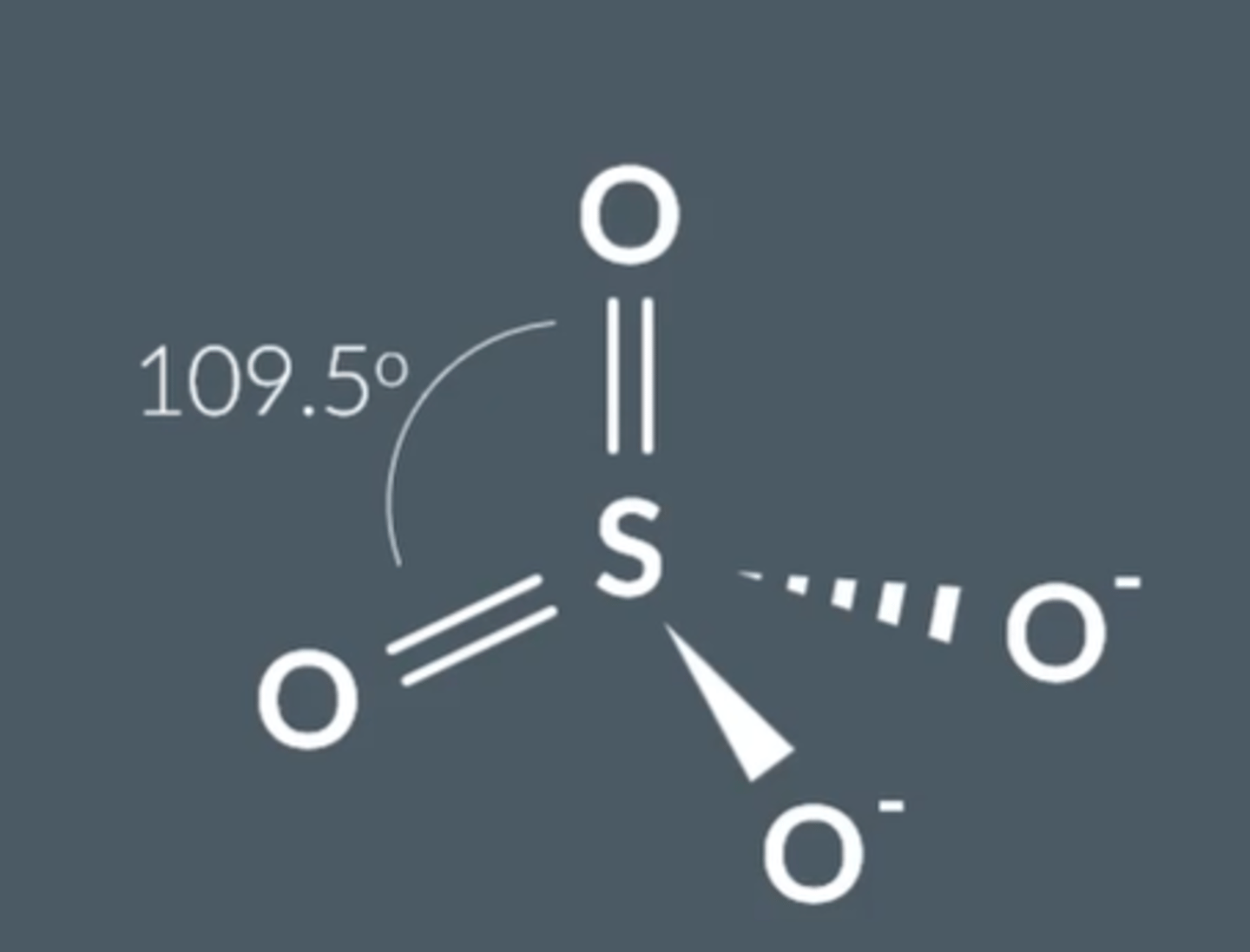
What is the structure, bonding and shape of SO₄²⁻ ?
Electrons delocalise → tetrahedral with 109.5° bond angle.
Each S-O bond is the same
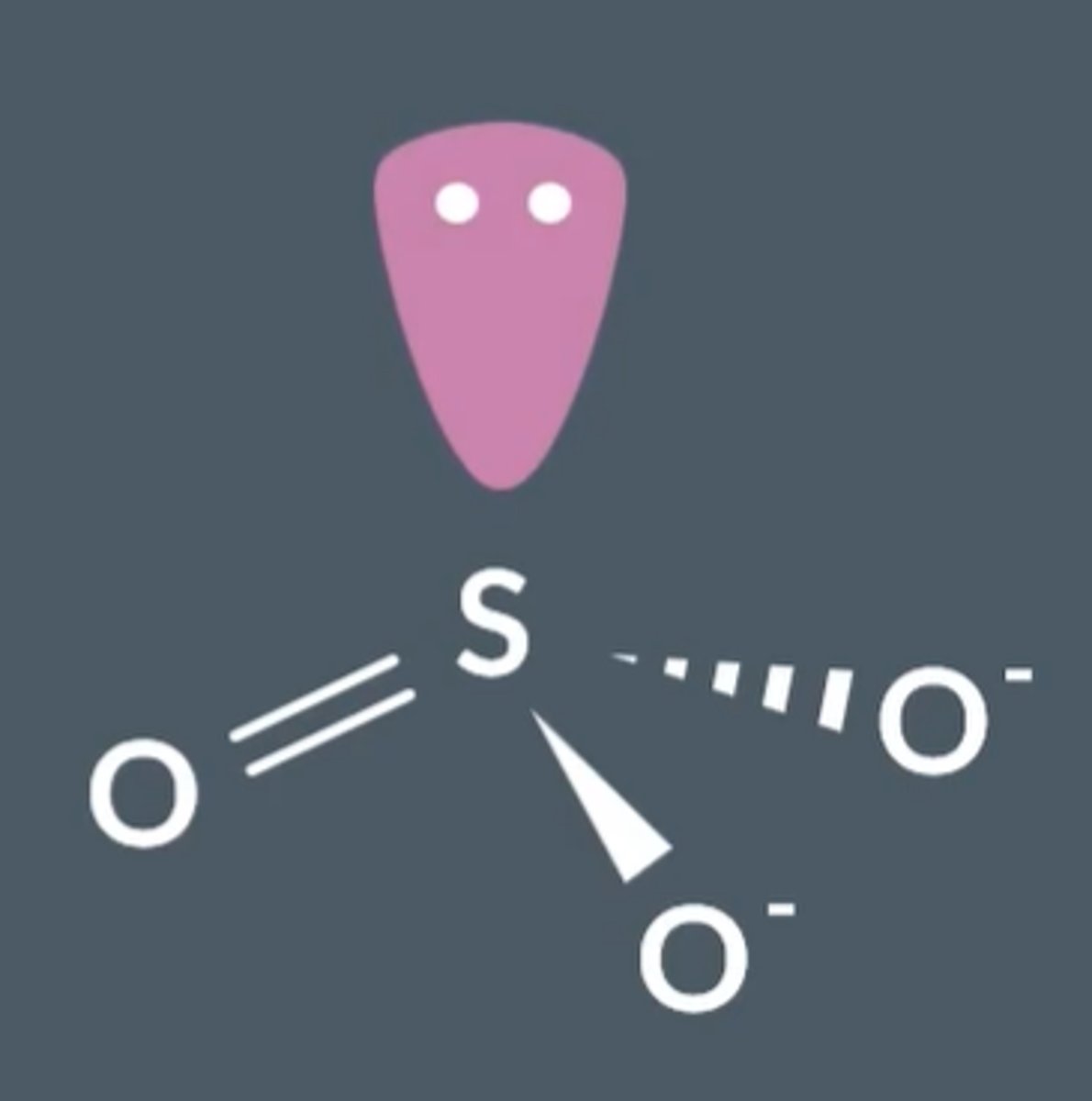
What is the structure, bonding and shape of SO₃²⁻?
Bond angle = 107° (trigonal pyramid shape), each S-O bond is the same, S has one lone pair of electrons.
Give the equation for sulfuric acid reacting with sodium hydroxide.
H₂SO₄ + 2NaOH → Na₂SO₄ + 2H₂O
Uses of MgO?
Additive for cattle feed
How useful is Al₂O₃?
Oxide layer on Aluminium makes it very useful as it is unreactive and returns quickly if it is scratched off
Uses of SO₂
Reactant in contact process (making H₂SO₄)
Would Lithium oxide or sodium oxide have a higher melting point? Why?
Li₂O has a higher m.p., since Li⁺ is a smaller ion than Na⁺, so the O₂ and Li⁺ charge centres are closer together and there is a greater electrostatic force of attraction between the
oppositely charged ions.