Pancreas anatomy and physiology
1/85
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
86 Terms
Describe the location of the pancreas
one part along the greater curvature of the stomach, other part cranial to the duodenum
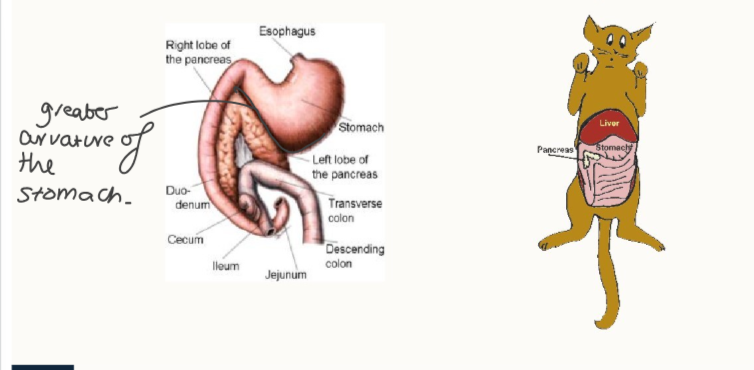
What is the exocrine function of the pancreas?
digestive enzymes - in response to GI hormones
bicarbonate to neutralise stomach acid → response to secretin
What are the 3=6 digestive enzymes produced by the pancreas?
trypsin and carboxypeptidase → proteins/peptides
lipase
amylase
phospholipase
ribonuclease
deoxyribonuclease
What are the 3 cells with endocrine function in the pancreas? What are their relative proportions?
beta - insulin
alpha - glucagon
delta - somatostatin
In order of percentage (60-70% beta, 20-25% alpha, 10% delta)
Outline somatostatin:
where is it produced
paracrine function
hypothalamus (PVN), stomach, intestine, pancreas
suppresses insulin and glucagon secretion from beta and alpha cells
Outline insulin:
type of hormone
secreted by?
synthesis chain
peptide
beta cells
preprohormone → prohomone, proinsulin - > insulin
How is proinsulin stored?
intracytoplasmic - ready to be released quickly when needed
What must be removed from proinsulin to activate insulin?
C-peptide
Outline insulin structure:
chains
connection
2 chains: alpha and beta
two disulphide bonds
Outline how insulin is degraded - 2 ways
liver/kidney via cleavage of 2 disulphide bonds
within target cells after receptor binding
Outline C-peptide and secretion:
what is it
how is it released
active?
what do we measure it for
connecting peptide for insulin
equal amounts to insulin
biologically inactive → removed at a slower rate
pancreatic function: if we have insulin but no C-peptide, tells us it’s injected, not self-produced insulin
Outline insulin kinetics:
half life
where does it enter into circulation?
first organ it acts on?
C-peptide in liver?
5-8 mins
veins → portal system
liver → general circulation
not removed
3 methods of insulin secretion
glucose and amino acids
GI hormones (GIP and GLP-1)
ANS: PNS and SNS
Insulin:
results in what process (building or breaking?)
method of release from beta cells
overall purpose
anabolic
calcium-mediated exocytosis
energy → storage
How do GI hormones trigger insulin release?
increase in concentration of GIP and GLP-1 when food reaches intestine → insulin release (in advance of absorption)
Outline how the PSNS affects insulin release
increased PSNS → insulin secretion
via vagus nerve
vagus nerve increases GI motility and digestion
higher insulin secretion after feeding
Outline how the SNS affects insulin release
direct innervation
indirect response via adrenaline
stress response - hyperglycaemia
insulin secretion is inhibited (as well as its action)
What is meant by:
first phase insulin release
second phase?
insulin released from its vesicles in response to a stimulus
active production of insulin by beta cells in pancreas to maintain levels required according to stimulus
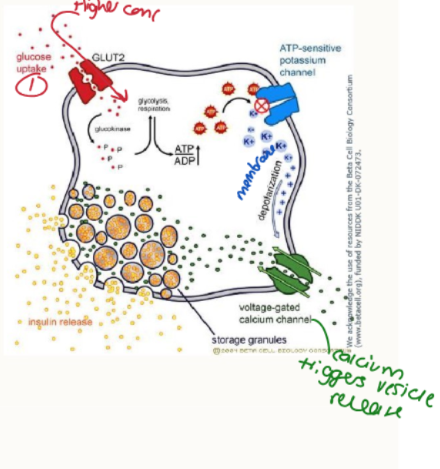
How does insulin release from beta cells work?
increased blood glucose
GLUT-2 transporters allow glucose to enter
phosphorylation of glucose
ATP production
ATP inhibits potassium channel (drug target)
increased K+ intracellularly → depolarisation
VG Ca2+ channels open
influx of Ca2+ triggers insulin exocytosis
Is insulin always needed to promote glucose uptake?
NO
insulin dependent cells don’t need insulin receptor for glucose uptake
insulin dependent cells need it
What type of hormone is insulin?
water soluble
What transmission pathway does insulin use?
tyrosine kinase
what is a determinant of insulin sensitivity in a cell
density of receptors
Outline GLUT-4 receptor:
what does insulin stimulate
what are GLUT-4 proteins?
what are two main tissues we find this occur in
translocation of GLUT4 to membrane
insulin-responsive glucose transporters
muscle and adipose tissues
Outline these 3 transporter types and where we find them:
GLUT-1
GLUT-2
GLUT-4
insulin independent glucose transport - CNS, RBC
facilitated insulin independent glucose transport - liver, islet glucose sensor
insulin-dependent glucose transport - muscle and fat
What are the 3 macronutrients insulin acts on and what 4 main organs
carbs, protein and fat
liver, brain, muscle, fat
How does insulin act in muscle
promotes glucose uptake
increases glycogenesis
Outline insulin influence in the liver:
what transporter takes up glucose
what does it inhibit
what does it promote
GLUT-2
glycogen phosphorylase (glycogenolysis) and gluconeogenesis
glycogen synthase (glycogenesis) and lipogenesis
What glucose transporter do we find in the brain, what is the exception?
GLUT-1: based on conc gradient, no insulin involvement
EXCEPT in the satiety centre
Why do we see increased hunger in diabetes mellitus?
insulin doesn’t trigger satiety centre in brain → appetite increase
Insulin and fat metabolism:
inhibits
stimulates
promotes?
hormone sensitive lipase (HSL) → decreases lipolysis
de novo lipogenesis
lipoprotein liipase → increases delivery of fatty acid into tissues
How does excess glucose → fat precursor?
glucose → increased TCA cycle intermediates (citrated)
activate acetyl CoA carboxylase (ACC)
malonyl CoA (fat precursor) produced
Insulin and protein metabolism:
increases
inhibits
depresses?
amino acid uptake by tissue, rate of transcription and translation
catabolism of proteins
rate of gluconeogenesis within liver - inhibiting enzymes and lowering supply of aas from tissues
Outline glucagon:
cells secreted by?
structure
preprohormone?
half-life
metabolised where?
alpha cells in Islets of Langerhans
29 aas
preproglucagon
5-6mins
liver and kidneys
Glucagon actions:
builds or breaks?
primary site of action
responds to what?
what ratio determines actions?
what do alpha cells respond to?
catabolic
liver
glucagon increasing in conc, insulin decreasing in conc
insulin: glucagon
low blood glucose level
Glucagon and the liver:
function
when is it activated?
stimulates?
maintains blood glucose levels in the inter-prandial period
during negative energy balance (starvation)
glycogenolysis, gluconeogenesis
How is glycogen converted into glucose?
Glycogen is phosphorylated (ATP→ AMP), glycogen phosphorylase enzyme
→ glucose 1-phosphate → glucose 6-phosphate → glucose
how is glucose converted into glycogen by insulin?
glucose → glucose—6-phosphate → glucose-1-phosphate → UDP, glycogen synthase → glycogen
Outline glucagon secretion
inhibited by high glucose levels, decrease → glucagon secretion increases
stimulated by high amino acid levels (alanine and arginine are most important) → gluconeogenesis
what does glucagon protect from?
post-prandial hypoglycaemia
maintains glucose levels, especially after protein-rich meal (insulin is released), otherwise low blood glucose would occur
Fill in the table
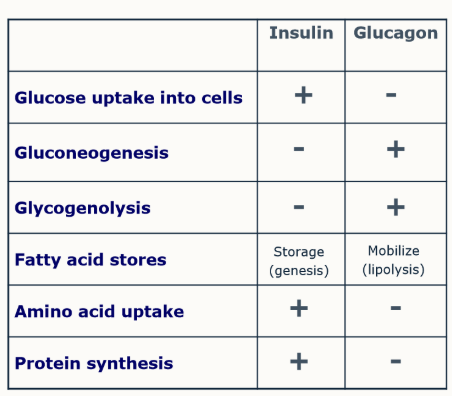
What is meant by hormonal antagonism?
two hormones that have opposing effects e.g. insulin and glucagon
importance in recognising, both can be present and have different effects - the ratio affects the outcome
Outline diabetes mellitus:
what is it described as?
how does it affect tissue use of different micronutrients?
what affect does it have on other organs?
result in the blood?
relative/absolute insulin deficiency
decreased tissue utilisation of glucose, increased utilisation of aas and fatty acids.
increased hepatic glycogenolysis and gluconeogenesis
hyperglycaemia → glucose accumulates in circulation
What are the two types of diabetes mellitus?
Type 1 - immune-mediated or idiopathic
Type 2 - insulin resistance w. relative deficiency
What causes type 1 diabetes?
beta-cell destruction → usually absolute insulin deficiency
What are other potential causes of diabetes mellitus (not type 1/2)
diseases of exocrine pancreas
endocrinopathies: cushing’s, acromegaly, phaeochromocytoma, glucagonoma, hyperthyroidism
genetic defects, drug/chemical induced or infections
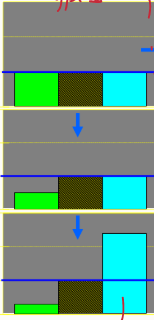
How do these diagrams represent type 1 diabetes?
normal levels on insulin, some naturally occurring resistance to insulin (brown) and normal glucose levels
damage leading to less insulin. For a time, the body can cope and insulin works overtime
Insulin levels are too low, blood glucose increases - no control. The liver also starts producing more glucose → increases levels even further (not inhibited by insulin)
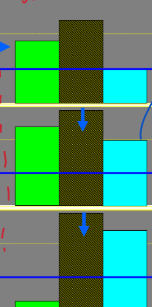
Outline how these diagrams represent type 2 diabetes
too much insulin and an increased resistance in the tissues, glucose levels are under control.
due to increased resistance to insulin, more is produced → more resistance. This causes glucose levels to increase
Insulin is no longer effective and cannot be produced (almost become type 1 diabetic), almost total tissue resistance, no blood glucose control → glucotoxicity
What does glucotoxicity effect?
Beta cell function
What 7 hormones/states can lead to glucose toxicity (think type 2 diabetes)
progesterone/agen
growth hormone
glucocorticoids
glucagon
catecholamines
thyroid
obesity
What are potential sites of dysfunction/causes of dysfunction as a result of glucose toxicity that can lead to diabetes mellitus (5)
pancreatectomy
pancreatitis
auto-immunity
islet cell hypoplasia
chemical toxicity
What are some causes of insulin resistance?
Physiological:
pregnancy, stress
Pathological
obesity
hereditary
concurrent disease
endocrinopathies - hyperadrenocorticism, acromegaly (GH excess)
Outline how insulin resistance can lead to diabetes
insulin resistance - compensated by extra insulin
IR → uncompensated (insufficient even if extra insulin)
e.g. obese horses
Type 2 diabetes mellitus (NIDDM)
In which species do we tend to find type 2 diabetes mellitus?
most cats
rare in horses (may be present in late stage PPID)
Physiologically, how does uncompensated IR lead to diabetes mellitus?
islet failure/glucotoxicy and hyperglycaemia → glucosuria and PU/PD
For diabetes mellitus in dogs, outline the most common causes
genetic suceptibility (consider different species in different countries that are more prone than others)
immune mediated destruction beta cells
pancreatitis with beta cells destruction
obesity-induced insulin resistance
insulin-antagonistic disease/conditions → cushing’s, met/dioestrus
insulin-antagonistic drugs - glucocorticoids
What is the pathogenesis for these developments of canine diabetes mellitus:
immune-mediated destruction of Beta cells
pancreatitis with beta cell destruction
Autoantibodies against insulin and/or beta cells OR progressive decrease in glucose-stimulated insulin secretion
spontaneous inflammation
How may insulin antagonism lead to canine DM?
specific hormones antagonise insulin action
cortisol and GH → induce peripheral insulin resistance
State 5 counter regulatory hormones
cortisol (hyperadrenocorticism)
GH
catecholamines
glucagon
progesterone
For each of these hormones, state what they can cause which in turn may lead to DM?
cortisol (hyperadrenocorticism)
GH
catecholamines
glucagon
progesterone
hyperadrenocorticism - spontaneous and iatrogenic
acromegaly
phaeochromocytoma
glucagonoma
dioestrus/gestation (these lead to a surge in GH production, in turn risking acromegaly formation)
For cats, state 6 common causes of diabetes mellitus
obesity/diet-induced insulin resistance
islet amyloidosis
pancreatitis
insulin-antagonistic drugs
insulin-antagonisti disease (acromegaly)
genetics
Why do we obesity related insulin resistance in cats?
high carbohydrate diet → glucose toxicity
cats can cope with a higher level of blood glucose before insulin is released, if this is constantly high, we result in DM
Define insulin resistance
diminished ability of cells to respond to the action of insulin in transporting glucose from the blood into tissues
Why does obesity lead to insulin resistance?
inadequate number of insulin receptors
defective insulin receptor structure
cell signalling pathway
defective GLUT-4 transport proteins
problems with translocation of GLUT-4 to membrane
interference with function of GLUT-4
What is amylin
co-secreted with insulin by feline beta cells
it’s deposited in islets by amyloid
What is the issue with amyloid fibrils?
cytotoxic → apoptosis of islet cells → defective insulin secretion
if it’s progressive deposition → diabetes mellitus
when does chronic increased amylin secretioin occur in felines?
obestiy
insulin resistant states
consequence of chronic hyperglycaemia/glucose toxicity
Can we reverse glucotoxicity?
yes, initially
may not be after prolonged periods of time
Outline the pathway from obesity → islet destruction in cats
obesity, insulin resistance
increased insulin secretion
increased amylin co-secretion
progressive amyloid deposition in islets
islet destruction
With felines and type 2 DM
is it always the case with cats
is it always reversible?
no
no - often when they present, they’ve been hyperglycaemic for so long, they’re closer to being type 1 diabetic (due to beta cell destruction, think about amylin production)
Insulin-dependent diabetes mellitus: IDDM
how common in dogs and cats?
treatment?
almost 100% dogs, 50-70% cats
permament insulin deficiency, therefore needs exogenous insulin
Non-insulin dependent diabetes mellitus (NIDDM)
in cats?
in dogs?
obesity/diet induced insulin resistance
insulin - antagonism, drugs - glucocorticoids, progestogens, conditions e.g. dioestrus
Outline equine metabolic syndrome:
strong link to…?
primary disorder is?
most common clinical sign?
what do we see high levels of?
obesity/regional adiposity
insulin resistance
laminitis
insulin and glucose
What are the 4 stages of diabetes presentation?
polyuria
polydipsia
polyphagia (remember, satiety centre won’t be suppressed)
weight loss
For diabetes mellitus and hyperglycaemia:
canine blood glucose
feline blood glucose
general guidline?
>10-12.2mmol/L
>11 to 15.5mmol/L
>10mmol/L
What is the physiological description of the cause of polyuria associated with DM
osmotic diuresis
Why do we see weight loss with diabetes mellitus
insulin:glucagon ratio falls → starvation process promoted
continuous inter-prandial period (mobilised stores, catabolic)
amino acids used for gluconeogenesis
increased protein breakdown → muscle wasting
Why can we see cataract formation with DM?
glucose uptake into lens
normally - metabolised into lactate, which diffuses out
glucose → fructose → sorbitol (doesn’t diffuse)
trapped → water drawn in
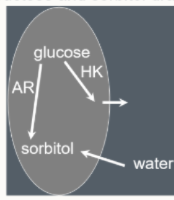
Outline ketoacidosis
unbalanced insulin:glucagon
shift to fat metabolism for energy
more fatty acids used produces ketones (ketoacids)
ketones build up → metabolic acidosis
What clinical signs will we see with keoacidosis?
vomiting
diarrhoea
anorexia → contribute to dehydration
What may a blood glucose between 5.5 and >10mmol/L
pre-diabetic
What is feline stress-induced hyperglycaemia
stress induced cortisol and catecholamines and hyperglycaemia
usually NO glycosuria unless prolonged stress
need to recheck urine in a non-stressed environment
How can we diagnose using fructosamine?
glycosylated serum proteins (albumin)
non-enzymatic reaction: proportional to blood glucose concentration
reflects previous 2-3 weeks of blood glucose
above 400umon/L = orolonged period of high glucose
What level of fructosamine confirms DM?
>400umol/L
What lab abnormalities may be associated with DM?
hypercholesterolemia
hypertriglyceridemia
visible lipid in serum/plasma (due to mobilisation of fatty acid from adipose tissue)
hepatic lipidosis (increased liver enzymes)
what will we excpect in urinalysis of DM?
USG often >1.025
glucose
+_ ketones
UTI: wbc, rbc, bacteria and proteins