module 2—chemical levels of organization
1/211
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
212 Terms
Human chemistry includes
organic molecules (carbon based)
biochemicals (produced by the body)
elements (inorganic and organic)
All matter in the world is composed of one or more…
elements
Element definition
a pure substance that is distinguished from all other matter by the fact that it cannot be broken down by ordinary means
The body cannnot make what
elements
Elements that are used in the body are derived from…
food and the air
What element is essential to the human body
calcium
What are the most abundant elements in the body
oxygen, carbon, hydrogen, nitrogen
T or F: elements rarely occur alone in nature
true
Compound definition
a substance composed of two or more elements joined by chemical bonds
Glucose always has these components
6 carbon, 6 oxygen, 12 hydrogen
Free radical definition
an atom or molecule with an unpaired electron in the outermost shell
Characteristics of free radicals
unstable
highly reactive
destructive to nearby molecules
How to stabilize a free radical
give up the unpaired electron
obtain an electron
Why does stabilizing a free radical disrupt homeostasis?
it breaks the molecule
Atom definition
the smallest quantity of an element that retains the unique properties of that element
Subatomic particles
proton, neutron, electron
Atomic number definition
the number of protons in the nucleus of the atom
The atomic number identifies…
the element
the usual number of electrons
Mass number defintion
the sum of the number of protons and neutrons in its nucleus
Ion definition
an atom that has an electric charge
Positively charged ion
cation
Negatively charged ion
anion
most individual atoms exist as…
ions
Ionic bond definition
close association between ions of opposite charge
Ionic bonds contain
1 metal element and 1 non metal element
Ions…
disassociate in water
Disassociation of ions in water produce…
electrical charges within the body
Behavior of ions can produce…
waves on EKGs or EEGs
Covalent bond definition
share electrons in a mutually stabilizing relationship
Covalent bonds involve…
2 non metal elements
Two kinds of covalent bonds
polar and non polar
Non polar covalent bond definition
covalently bonded molecules that are electrically balanced
sharing of negative electrons is relatively equal in the electrical pull of the positive protons in the nuclei
In a single covalent bond, how many pairs of electrons are shared
one pair
Examples of non polar molecules
fats and oils
What type of phobic are nonpolar covalent bonds
hydrophobic
Polar molecule definition
a molecule that contains regions that have opposite electrical charges
Polar molecules occur when atoms share electrons _____ in polar covalent bonds
unequally
Example of polar molecule
water
Characteristics of polar covalent molecules
dipoles
hydrophilic
Hydrogen bonds are a type of…
intermolecular bond
Hydrogen bonds are formed when a…
weakly positive hydrogen atom is attracted to a highly electronegative atom
What electronegative atoms are seen in hydrogen bonds
nitrogen, oxygen, fluorine, halogens
Van der waals forces are a type of…
intermolecular bond
Van der waals forces are…
weak and temporary attractions between nearby molecules
Van der waals forces are due to…
transient unequal charge distributions
Ionic compounds are dissolvable in…
polar solutions
Polar molecules are dissolvable in…
polar solutions
Non polar molecules are dissolvable in…
non polar solutions
What do polar and non polar interactions mean for human physiology?
creates essential compartments and biological structures
Ionic or covalent? H2O
covalent
Ionic or covalent? NaCl
ionic
Ionic or covalent? CO2
covalent
Ionic or covalent? NaSO4
ionic
Polar or nonpolar? H2O
polar
Polar or nonpolar? Cl2
nonpolar
Polar or nonpolar? CCl4
nonpolar
Polar or nonpolar? HCl
polar
Chemical energy is…
absorbed, stored, and released by chemical bonds
Mechanical energy…
directly powers the movement of matter
Example of mechanical energy
lifting a brick to place on a wall
the muscles provide the mechanical energy that moves the brick
Radiant energy is…
energy emitted and transmitted as waves rather than matter
The types of waves that can be emitted from decaying atomic nuclei
radio waves (long), microwaves, gamma rays (short)
The full spectrum of radiant energy is referred to as the…
electromagnetic spectrum
The body uses the ultraviolet energy of sunlight to convert a compound in the skin cells to…
vitamin D
The human eye has evolved to see wavelengths from red to violet in the spectrum called…
“visible light”
Electrical energy is supplied by…
electrolytes in the cells and body fluids
Electrolytes contributes to the…
voltage changes that help transmit impulses in nerve and muscle cells
A + B→AB
synthesis reaction
AB→A+B
decomposition reaction
A+BC→AB+CA+BC→AB+C,
AB+CD→AC+BDAB+CD→AC+BD, AB+CD→AD+BCAB+CD→AD+BC
exchange (subsitution) reaction
CxHx+O2 → CO2 + H2O
combustion reaction
Any chemical reaction can proceed in ____ ______ under the right conditions
either direction
What type of arrow shows reversibility of a chemical reaction
double equilibrium arrow
Law of mass action
direction of the reaction is influenced by the concentration of the reactants and products
The greater the surface area of the reactants…
the more readily they will interact
The larger the molecule…
the greater number of total bonds
Reactions involving smaller molecules with fewer total bonds are expected to…
proceed faster
Reactions that involve hydrogen are fairly…
reactive
Reactions involving helium are likely…
not to occur due to its stability
Nearly all chemical reactions occur at a faster rate at…
higher temperatures
Higher temperature means…
particles move faster and bump into each other more frequently
The more particles present in a given space…
the more likely they are to bump into each other
Chemist can speed up chemical reactions by…
increasing the concentration of particles
decreasing the volume of the space/increasing the pressure
Catalyst definition
a substance that increases the rate of a chemical reaction without undergoing any change
Enzyme definition
a catalyst composed of protein or ribonucleic acid (RNA)
How do enzymes work?
they lower the level of energy that needs to be invested in a chemical reaction
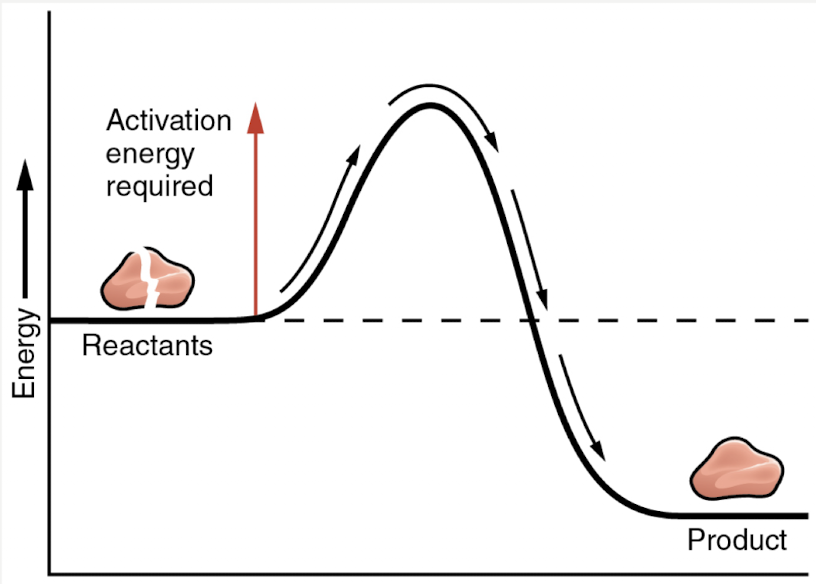
Is this reaction with or without an enzyme
without
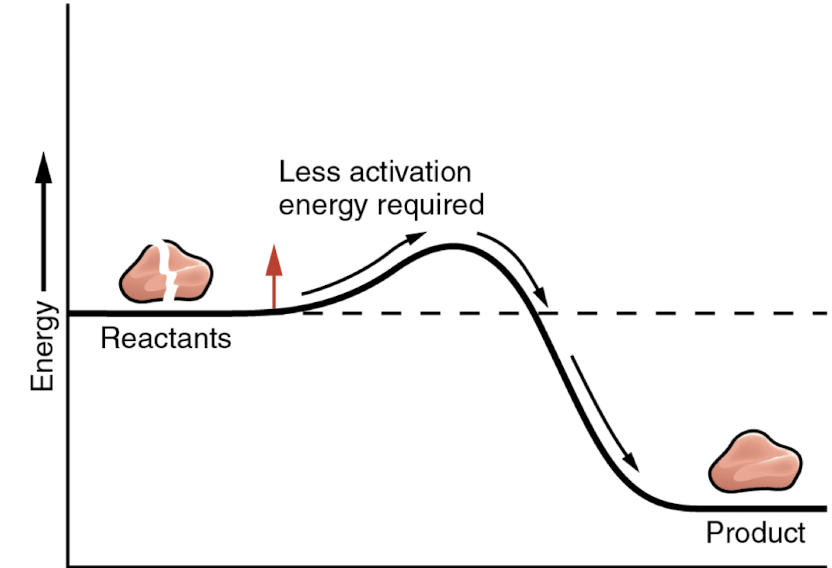
Is this reaction with or without an enzyme?
with
Inorganic compound definition
a substance that does not contain both carbon and hydrogen
Examples of inorganic compounds
H2O, HCl, CO2
Organic compound definition
a substance that contains both carbon and hydrogen
Organic compounds are synthesized via…
covalent bonds within living organisms
What is the percentage of water that makes up an adult’s body weight
70
Where is the water contained in the human body
within cells
between cells that make up tissues and organs
Water in synovial fluid…
lubrictes the actions of body joints
Water in pleural fluid…
helps the lungs expand and recoil with breathing
Watery fluids help the GI tract by…
keeping flood flowing
ensures movement of adjacent abdominal organs are frictionless
Water also protects cells and organs from…
physical trauma
Water does what for the brain
cushions it within the skull
Water protects…
the delicate nerve tissue of the eyes