P5.1 The nucleus
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
Describe the composition of the nucleus
Composed of positively charged protons and neutrally charged neutrons
The nucleus has a positive charge
State the relative charges, mass and location of the main subatomic particles
Subatomic Particle | Location | Mass | Relative Charge |
Protons | Nucleus | 1 | +1 |
Neutrons | Nucleus | 1 | No charge |
Electrons | Shell | 0 | -1 |
How do you calculate nuclear charge?
number of protons in the nucleus x relative charge of a proton (1)
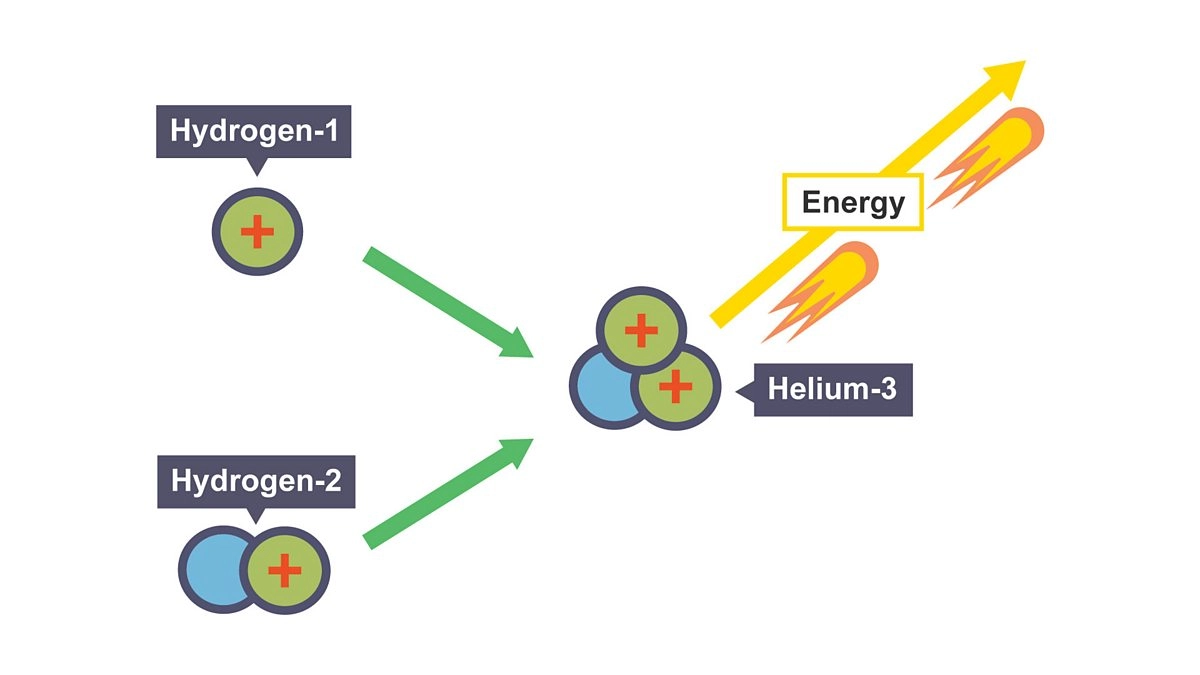
Define proton number (Z)
Proton number: amount of protons in an atom, determines the atomic number
Number of protons determines the element it is
Define nucleon number (A)
Nucleon number: total number of particles in the nucleus of an atom (protons + neutrons), determines mass number
How to calculate neutron number?
nucleon number - proton number
Use nuclide notation
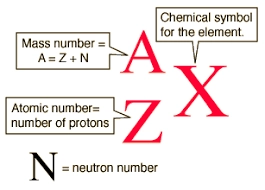
What is nuclear fission?
The process of splitting a large, unstable nucleus into two smaller nuclei
Describe the process of nuclear fission
Neutron collides with the large, unstable nucleus (reactants)
The nucleus splits into two smaller nuclei (products)
Gamma rays are emitted during this process
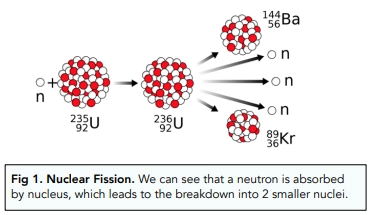
What is nuclear fusion?
The process of joining two light nuclei to form a single, heavier nucleus
Describe the process of nuclear fusion
Two light, atomic nuclei merge to form a single, heavier nucleus
Energy is released during this process
Because of this, the mass of the product will be less than the combined mass of the reactants since some of it was converted into energy