CHE 2C | ELECTROCHEMISTY/ TRANS METALS | EXAM I
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
In an electrochemical cell, where does oxidation occur?
ANODE (LHS)
are ligands considered a lewis base or a Lewis acid?
Ligands are considered Lewis bases because they donate electron pairs to form coordinate covalent bonds with metal ions.
difference between electron geometry and molecular geometry
example of EG: Linear, tetrahedral, octahedral (the general terms, basically)
example of MG: Square planar, octahedral (the more specific shape)
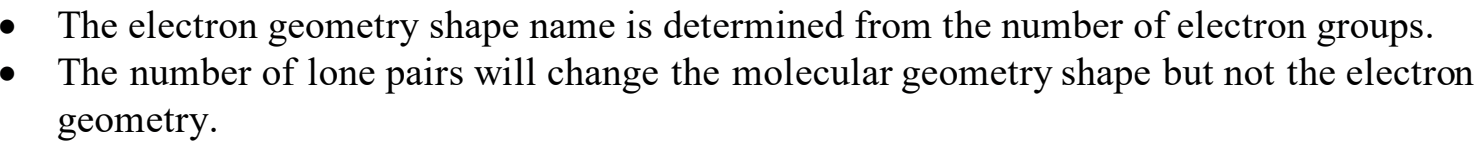
are structures with linear EG able to be chiral?
No, (linear electron geometry structures cannot be chiral due to their symmetrical nature)
are structures with tetrahedral EG able to be chiral?
yes, its possible
are structures with octahedral EG able to be chiral?
yes, it’s possible
are structures with tetrahedral EG AND square planar MG able to be chiral?
NO
what kind of trans. metals have no visible color?
if they have no electrons in their d orbital OR their d orbital is full
how is A and C related?
1 A= 1 C/sec

how would u solve for Ksp in a question like this?
how do u tell if something is a spectator ion, and that u can ignore it?
if throughout the run, the ox state STAYS THE SAME, that is a spectator ion!!
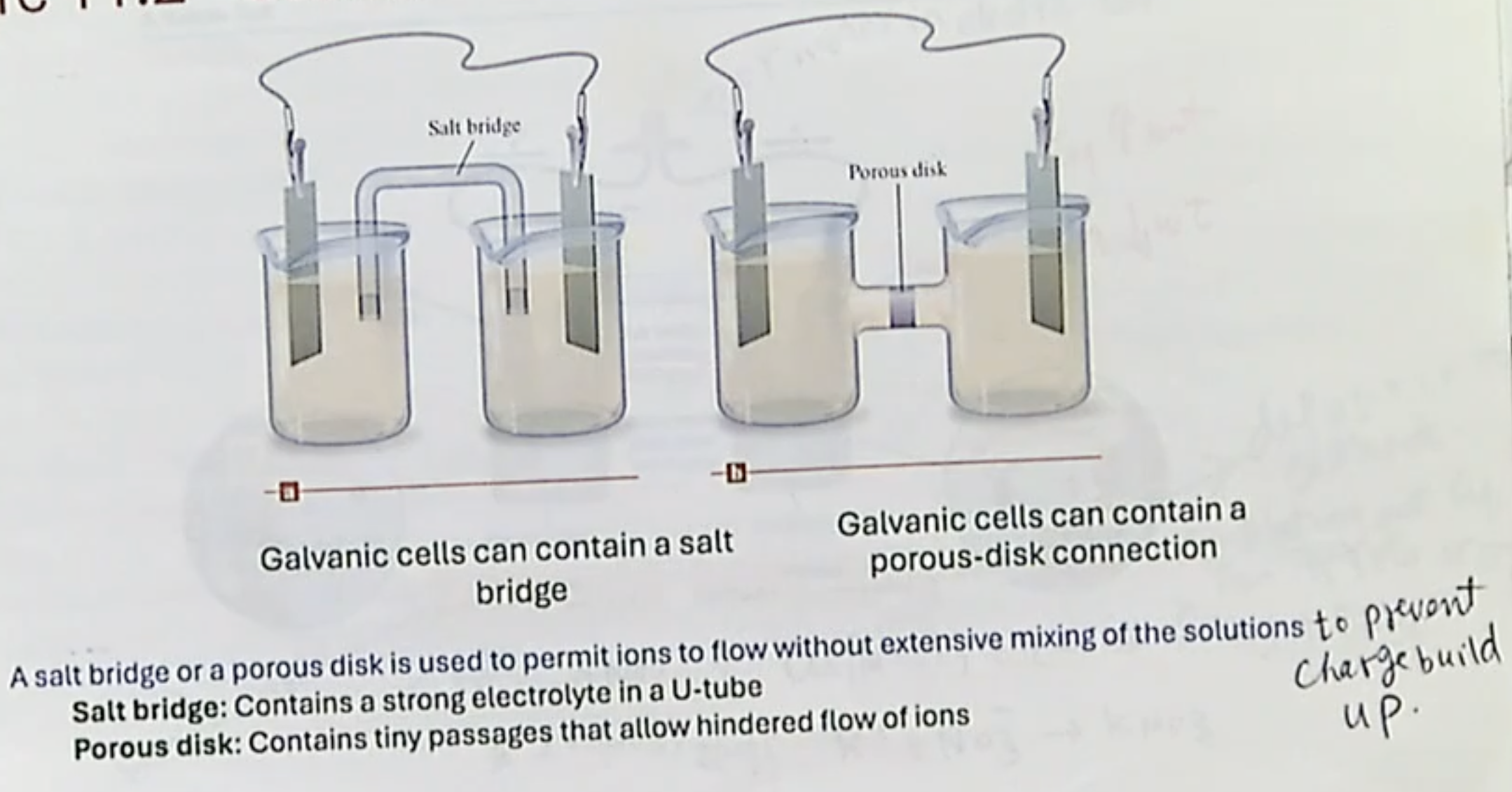
what does a salt bridge/Porous disk do, and how are they different?
differences:

equation for solving the Ecell with tabulated values
USE THE REDUCTION POTENTIAL VALUES FOR THE ANODE AND CATHODE! if u want you can also do Ecell = Ered + Eox and then for the Eox you put the oxidation potential (the given reduction potential FLIPPED)

In an electrochemical cell, where does reduction occur?
CATHODE, (RHS)
In an electrochemical cell, what determines the voltage of a reaction? What’s it measured in?
Voltmeter, which finds the VOLTAGE (which is in volts (V) which is J/C )
What 2 things is Voltage equal to?
electromotive force (emf) and cell potential (ξ° or ECell)
In an electrochemical cell, what is usually used for the electrode?
The soild metal in the half reaction
In an electrochemical cell, if the reaction doesn’t have a solid, what do you use as the electrode?
Either Pt (s) or C (graphite)
In an electrochemical cell, why are salt bridges important?
It prevents charge buildup, because it allows ions to exchange between the bridge and the half cell
In an electrochemical cell, what does the wire do? Which direction will they flow?
it allows electrons to transfer from the oxidation to reaction half-reaction. The e-’s flow in the SPONTANEOUS DIRECTION
In an electrochemical cell, what compound do you use for the salt bridge?

In an electrochemical cell, what if the reaction has more than one aqueous compound present?

In an electrochemical cell, what if a gas is present?

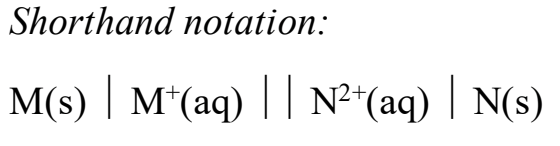
For shorthand notation of an electrochemical cell, what do the single lines mean? What about the parallel lines? Does the anode and cathode go on the right or left?
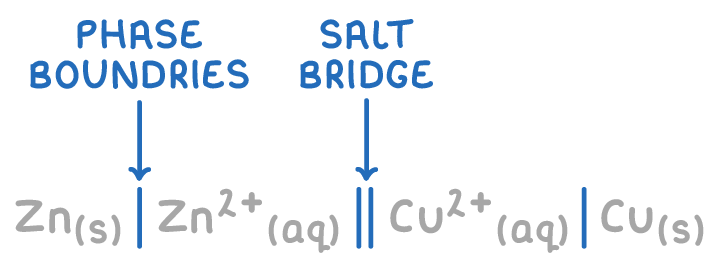

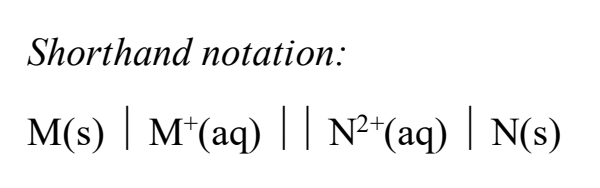
For shorthand notation, what is the order of place the different types of phases (aq, s, l, g)

(SHORTHAND NOTATION) what do you do if there are more than one species in the same phase in a half cell
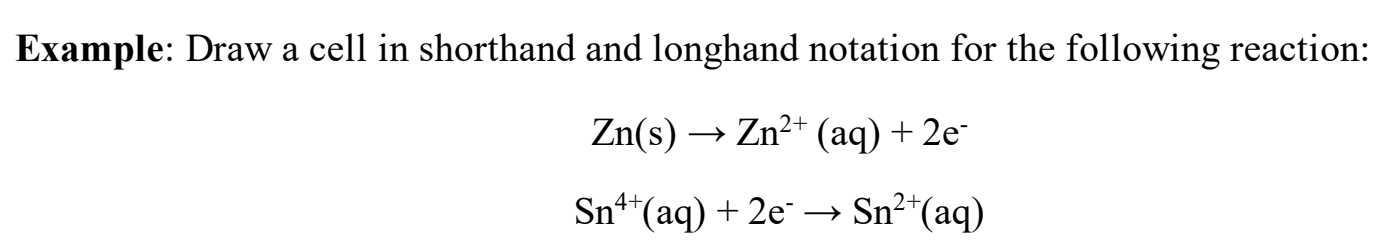
Not rlly looking for the full answer but review this for a general idea of what longhand/shorthand notation will look like
oxidation state vs formal charge
formal charge: The hypothetical charge assigned to an atom, assuming all bonds are perfectly covalent (shared equally)
oxidation number: The hypothetical charge an atom would have if all bonds were 100% ionic (electrons assigned to the more electronegative atom).
what are componds’ elemental forms you need to remember?

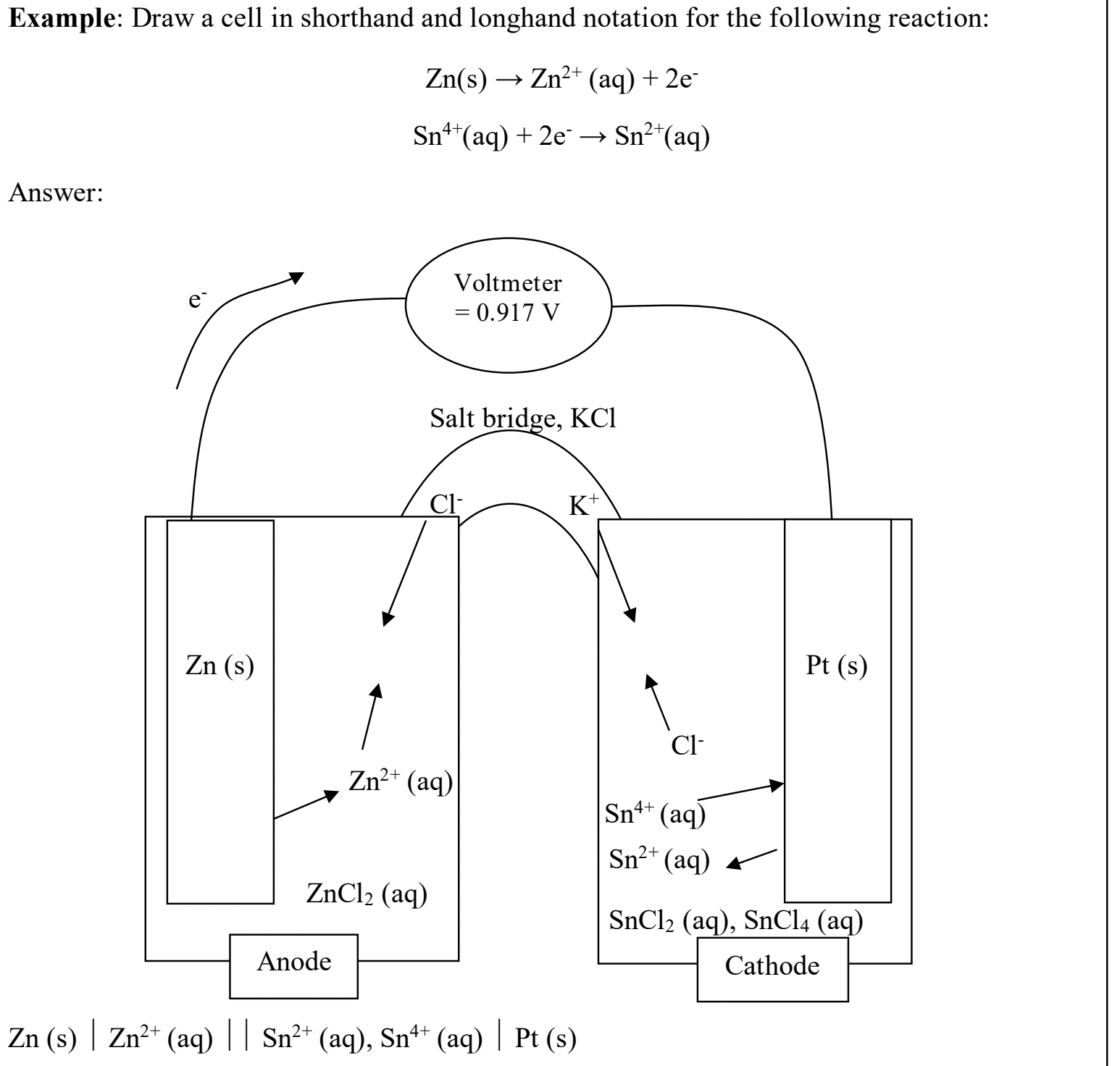
Say ur given a problem like this, where you know what’s in each half cell, but no information is given about the state symbols or which one is being oxidized or reduced. How would u figure this out?
Reference the Standard Reduction Potentials Chart (which is a given on tests). (look at pic attached) now that we have that, know that the half-reaction with higher reduction potential will be reduced (IT’LL BE IN THE CATHODE). But first, since we’ve been told to assume all these problems are spontaneous (MEANING, IF U WERE TO ADD THESE 2 VALUES TOGETHER, THE REDUCTION POTENTIAL VALUE WILL BE POSITIVE), manipulate the half cell with Cr so it’s reversed, making it the oxidized one (and that Ecell will be a positive value now)!
ALSO for the state symbols look at the given equation and figure it out LOL

for electrochemical cells, on which side would the oxidizing agent go on?
It’ll be in the cathode!
for electrochemical cells, on which side would the reducing agent go on?
It’ll be in the anode!
for electrochemical cells, on which side does oxidation occur?
in the ANODE
for electrochemical cells, on which side does reduction occur?
in the CAHTODE
study snack 👅

corrosion

difference between Fe and other metals like Al in regards of when it corrodes
Fe2O3 will FLAKE OFF. Al2O3 forms a protective coating, actually. Gold is the only metal that doesn’t corrode.
with rusting- where is the anode area?
in the water droplet
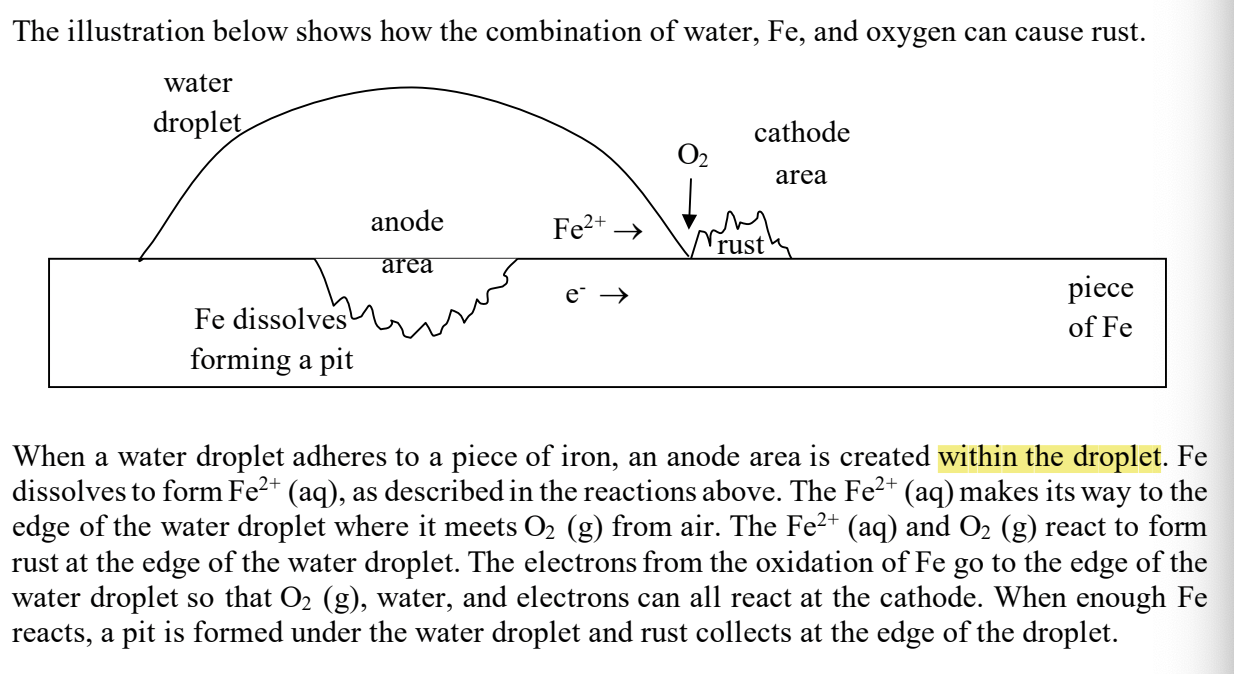
with rusting- where is the cathode area?
around the rust spot outside the water
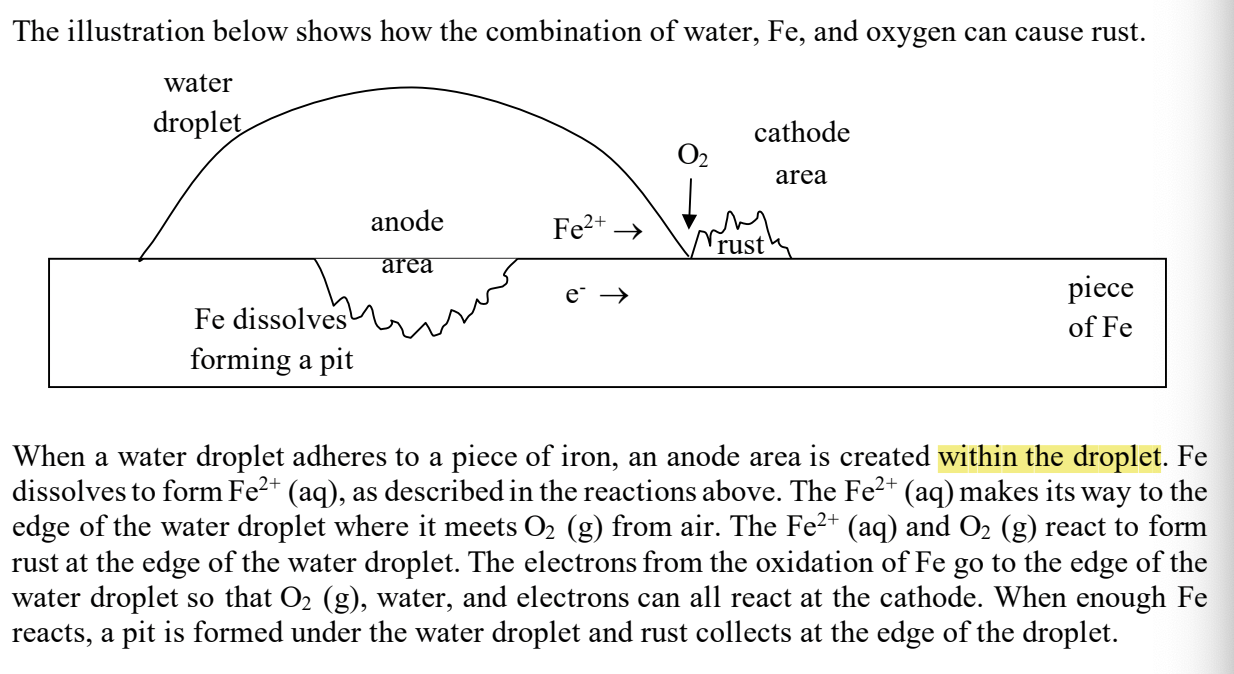
with rusting- where does the Fe+2 and e-’s go?
to the cathode area (like a galvanic cell!) where they combine with the oxygen
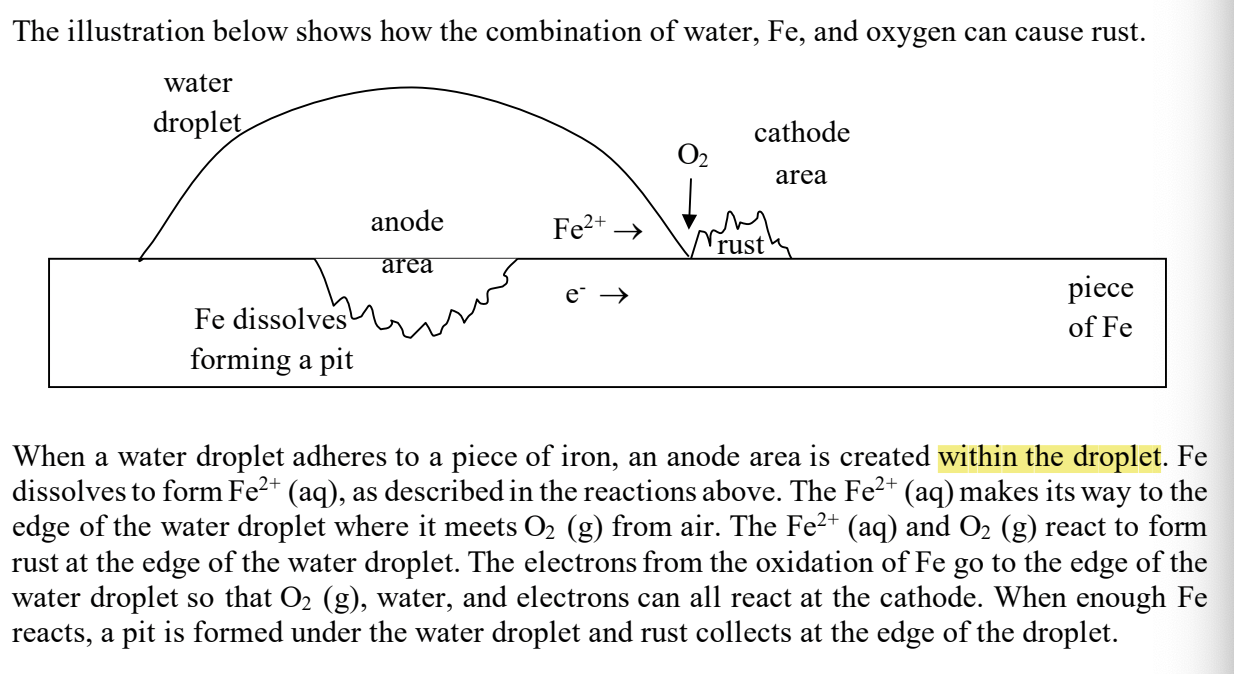
with rusting- what happens the to Fe that’s in contact with the water droplet
the Fe dissolves, forming a pit
what is cathode protection? what is a sacrificial anode? irl example?
its a technique that’s used for controlling the process of corrosion of a metal
sac anodes: are attached to the metal, so that they will oxidize first.
they are attached to the bottoms of ships (that are near the water)

what is plating?
adding a day anode onto another metal

for plating, what’s the key characteristic you’d want the metal u choose to have?
IT NEEDS A LARGER OXIDATION POTIENTIAL!! (opposite of reduction potential)
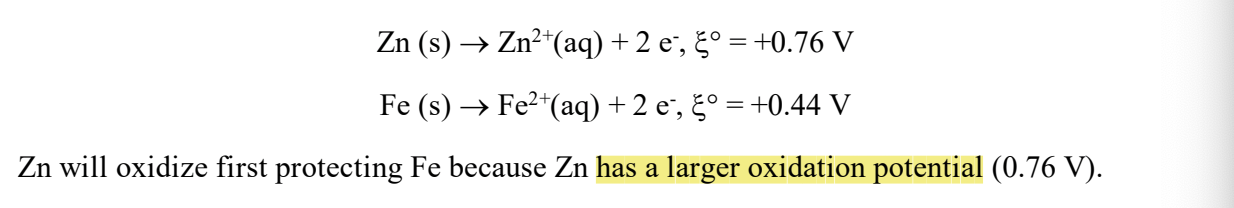
what an alloy, and what’s an example
a mixture of metals that may/may not be homogeneous.
example; stainless steel
in a galvanic cell, if the voltmeter reads a negative voltage, what does that mean?
reaction is NON-SPON!
study snack pt2 👅

What is the lore for the SHE (what is the reaction, what are its conditions, what does it stand for, what does it do, what is the voltage value, is it special?)
REACTION: 2H+ + 2e- → H2
Conditions: [H+] =1 M, pH=0, (with a H2 gas at [?]) P= 1 bar/~1 atm, 298 K. A Pt (s) electrode is used.
Stands for: STANDARD HYDROGEN ELECTRODE
What it does: It serves as the reference electrode for measuring standard reduction potentials, assigned a voltage value of 0 V (the ABSOLUTE potential is not 0V, its thought to be ~4.4V, but for practical use, it is defined as 0V.). All other electrode potentials are measured relative to it.
Voltage Value: 0V
Special?: NO. any run could’ve been the reference electrode.
primary cell- reversible? Can it die?
IRREVERSIBLE! (not chargeable) so it will eventually die
secondary cell- reversible? Can it die?
they CAN be reversed by charging it- they will eventually degrade and die but only after being used thru hundreds of cycles.
2 types of primary cells:
Leclanché (or dry) cell
Button battery (silver-zinc cell)
Leclanché (or dry) cell- irl examples, general facts, voltage, why is zinc used?
can hold charge for a while.
AA, AAA
~1.5V
Zinc is used for the anode bc it does corrode quickly in an alkaline environment
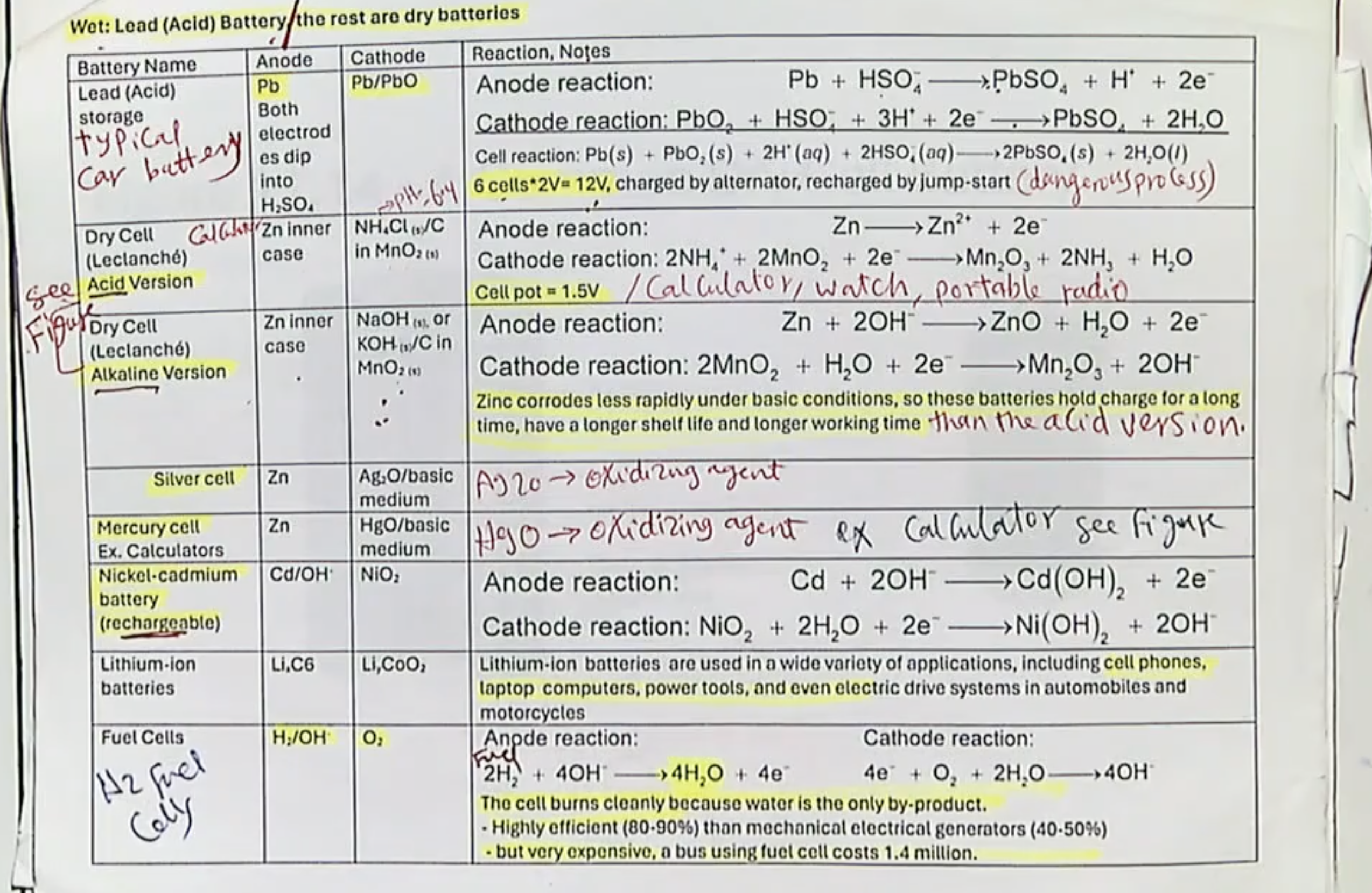
Button Battery/Silver-zinic cell- irl examples, general facts, voltage, is it alkaline or acidic?
Used in watches/hearing aids.
They are small with high storage capacity
~1.8V
Cell is alkaline (in the run, notice the OH- ions.)
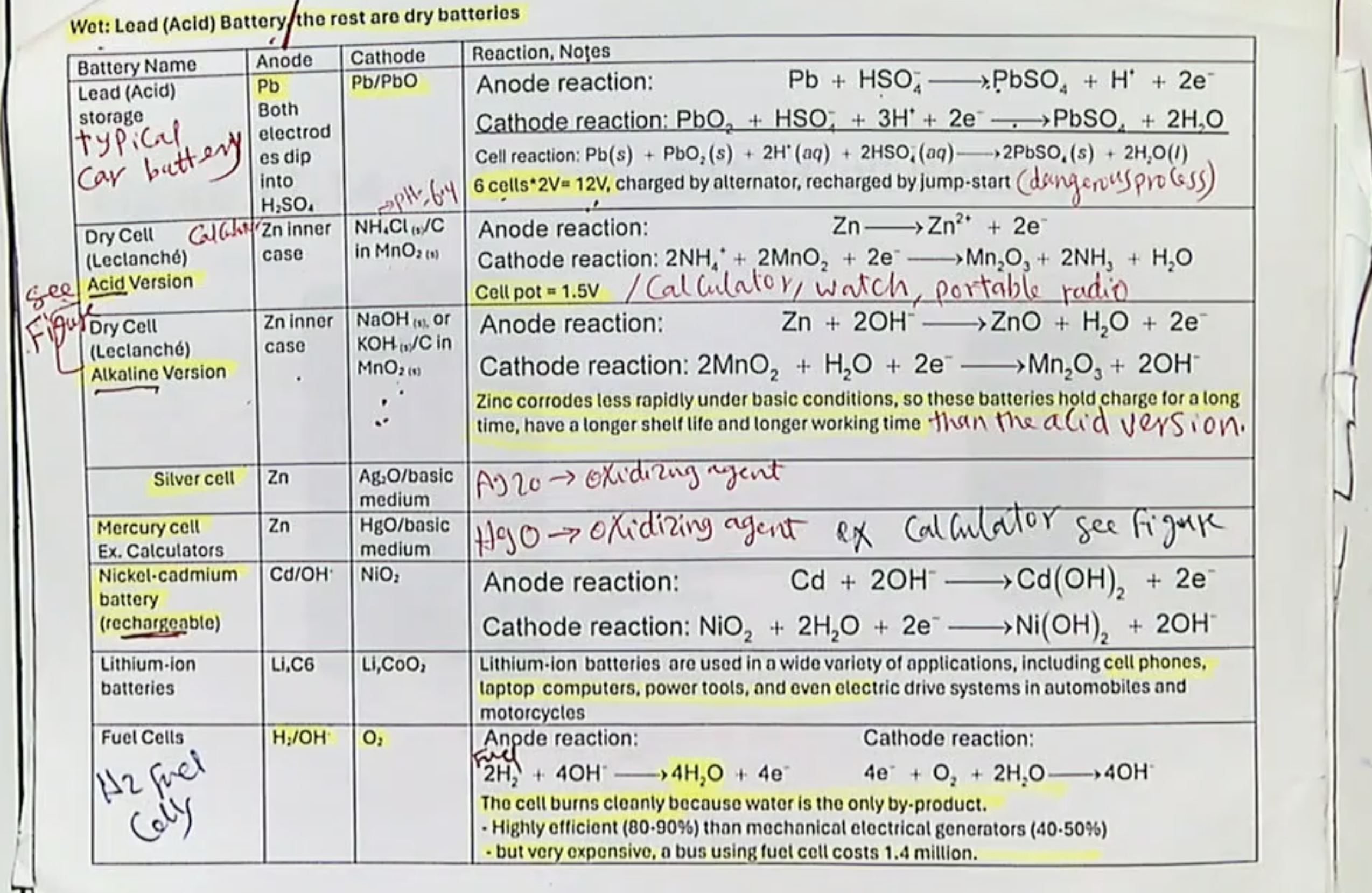
3 types of secondary cells:
Lead-acid (or Storage) Battery, Nickel-Cadminum (or Nicad) Battery, and Lithium Battery (AKA Li-Ion Cell)
Storage Battery- Voltage, are the products and reactants similar? Example?
~2V
Yes, the species in the products and reactants are similar.
Example- Car Battery
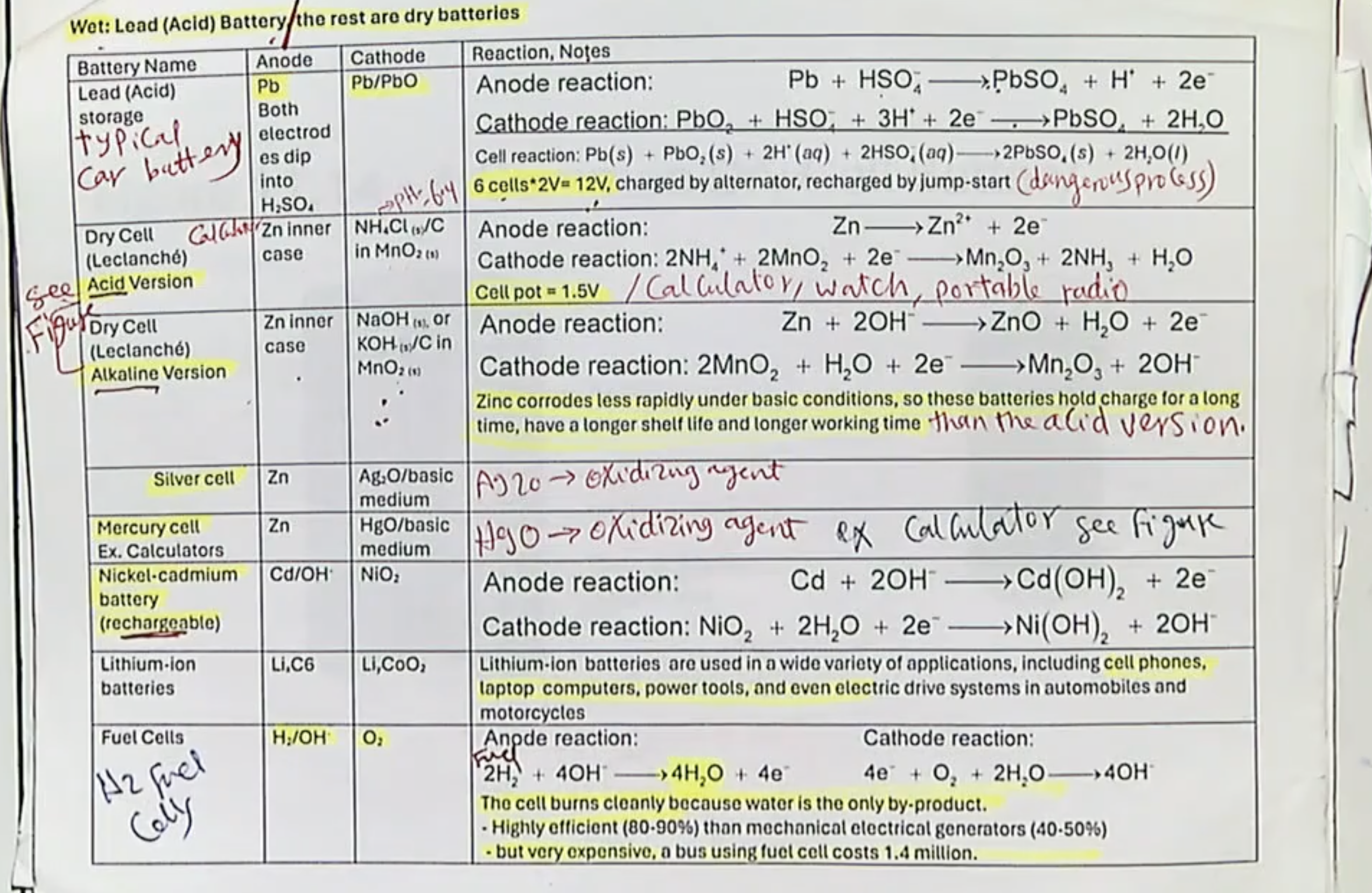
NiCad Battery- Voltage, are they rechargeable? Examples? Why do they decrease in capacity over time? Basic or acidic?
~1.2V
Yes, they are rechargeable
Examples are rechargeable AA/AAA Batteries
The capacity decreases over time is due to the "memory effect," where repeated partial discharges cause internal crystal growth of the Cd that reduces capacity.
They are alkaline (notice the OH- ions)
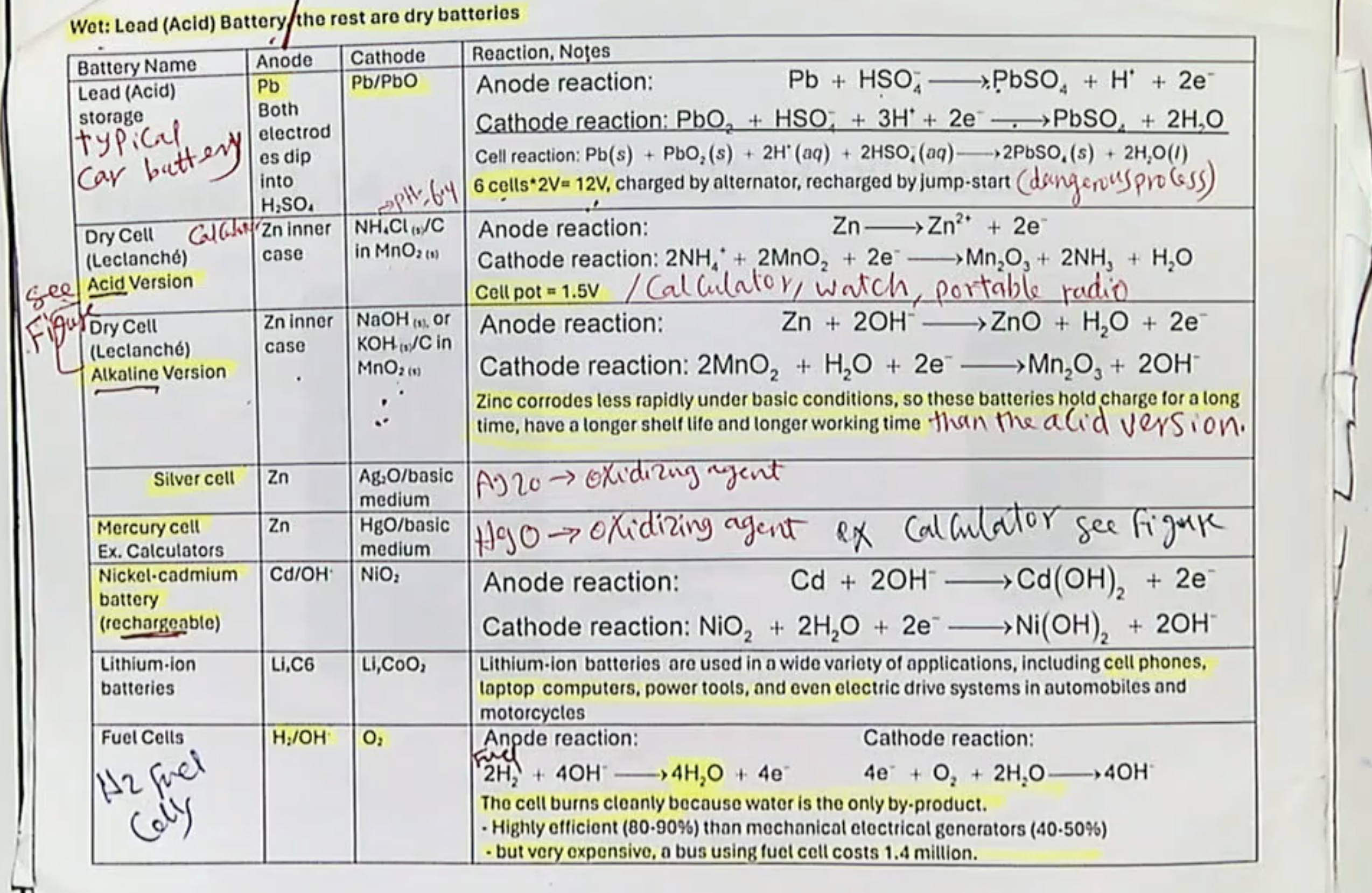
Li-Ion Cells- basic info, examples, do they like heat, do they will being completely charged up?
rechargeable with a long lifetime
found in smartphones and laptops
no, they dont like heat
no, charging too much can heat up the device. For example, with phones, u usually only want them to be charged to ~80%
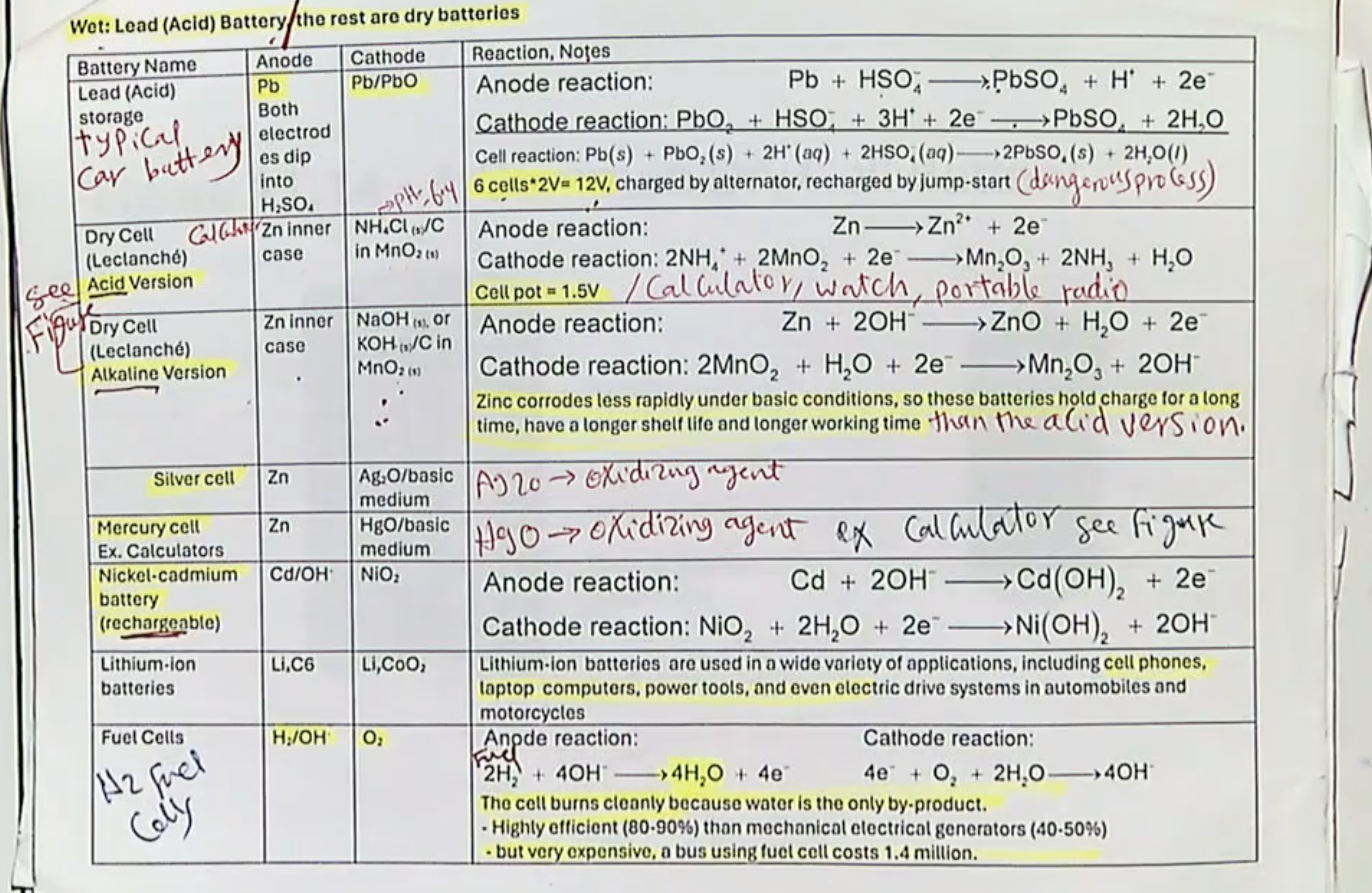
What are the 2 types of Flow/Fuel Cells?
Fuel cells and Air batteries (idk if we need to know stuff about air batteries tho LOL)
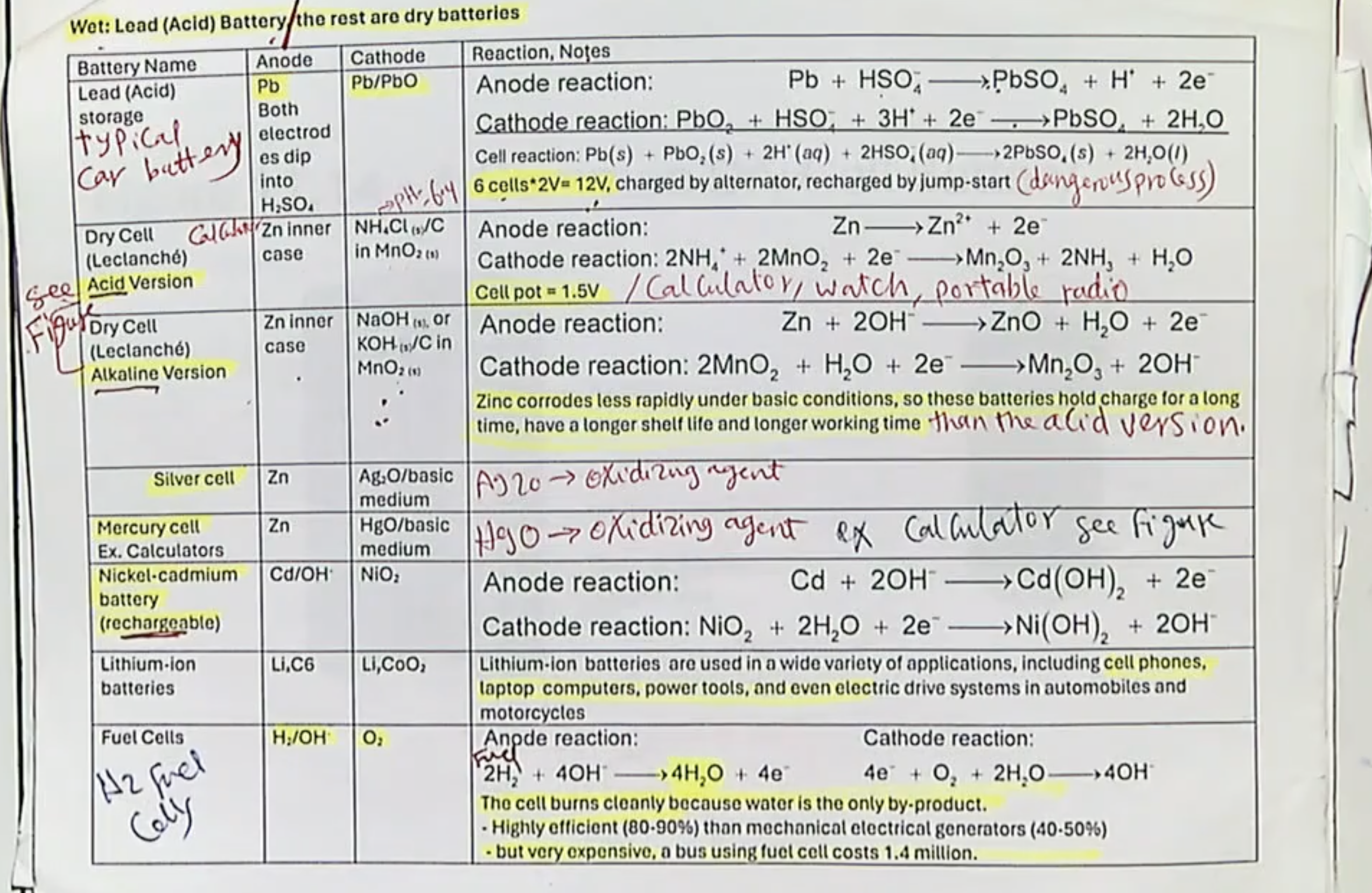
basic description of Flow/Fuel Cells:
Fuel cells
lazy for this one LMAO

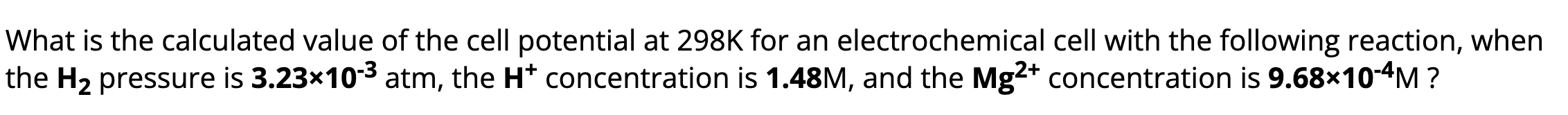
what do u do if your given the atm of a gas like this? do u do anything with it?
put it in there when calculating the Q value like this. if the pressure isn’t given, assume its 1.
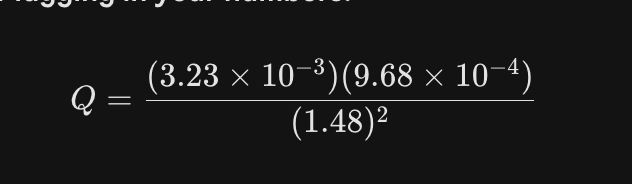
what a VERY IMPORTANT REASON to be careful when using standard reduction potential charts?
CUZ WE GOT SHT LIKE THIS?!?!? WTAF??? HOW MANY HGs DO WE FUCKING NEED

what happens when H2CO2 is formed?
It is actually written as H2O + CO2
Tungsten

MANGANESE

TECHNETIUM

RHENIUM

IRON

Ruthenium

Osmium

Cobalt

Rhodium

Iridium

Nickel

Palladium

Platinum

Copper

Silver

Gold

Zinc

Cadmium

Mercury

Lanthanides

Actinides

doubly-magic
A term used in nuclear physics to describe a nucleus that has both a neutron and a proton number that are both equal to magic numbers, resulting in increased stability.
what is the equivalent for the octet rule for trans metals?
18 electron rule (since theres 18 elements in the row)
coordination compound
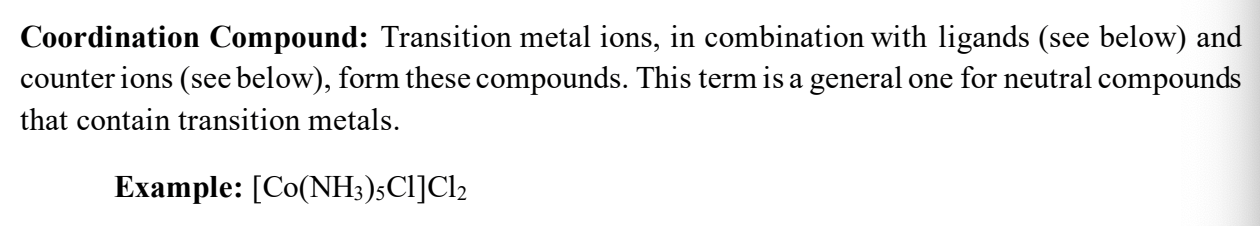
complex ions

Counter Ions
Cl2 here

Ligands

Coordination Number (CN)
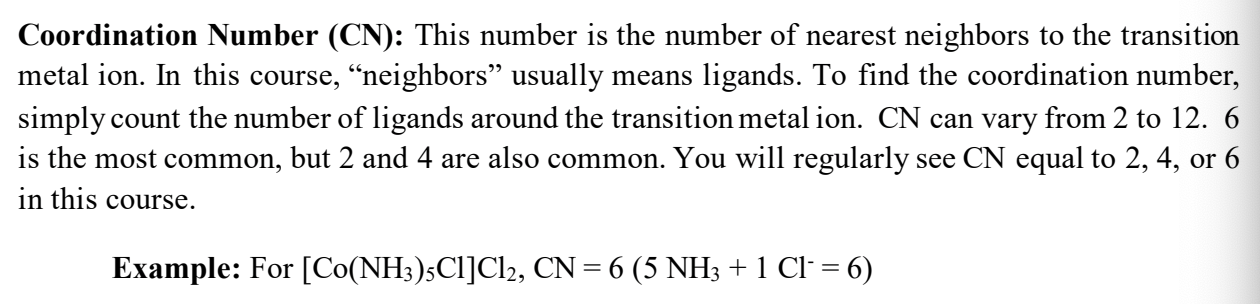
Complex
General term for any species involving ligands connected to a transition metal ion.
oxidation state
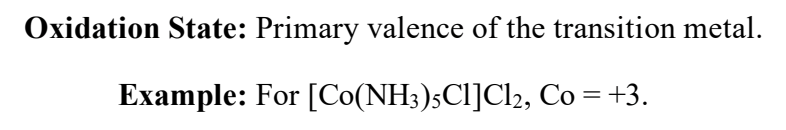
monodentate

polydentate (also, what’s another term for it?)

LIST OF LIGANDS we needa know(mono dentates)
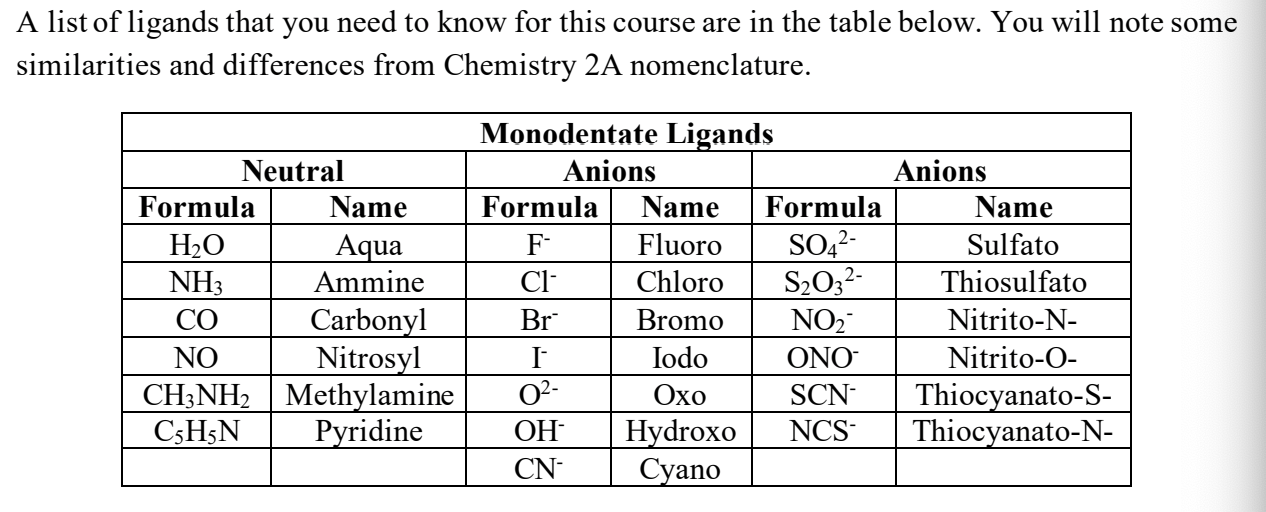
ligands we needa know (polydentates)
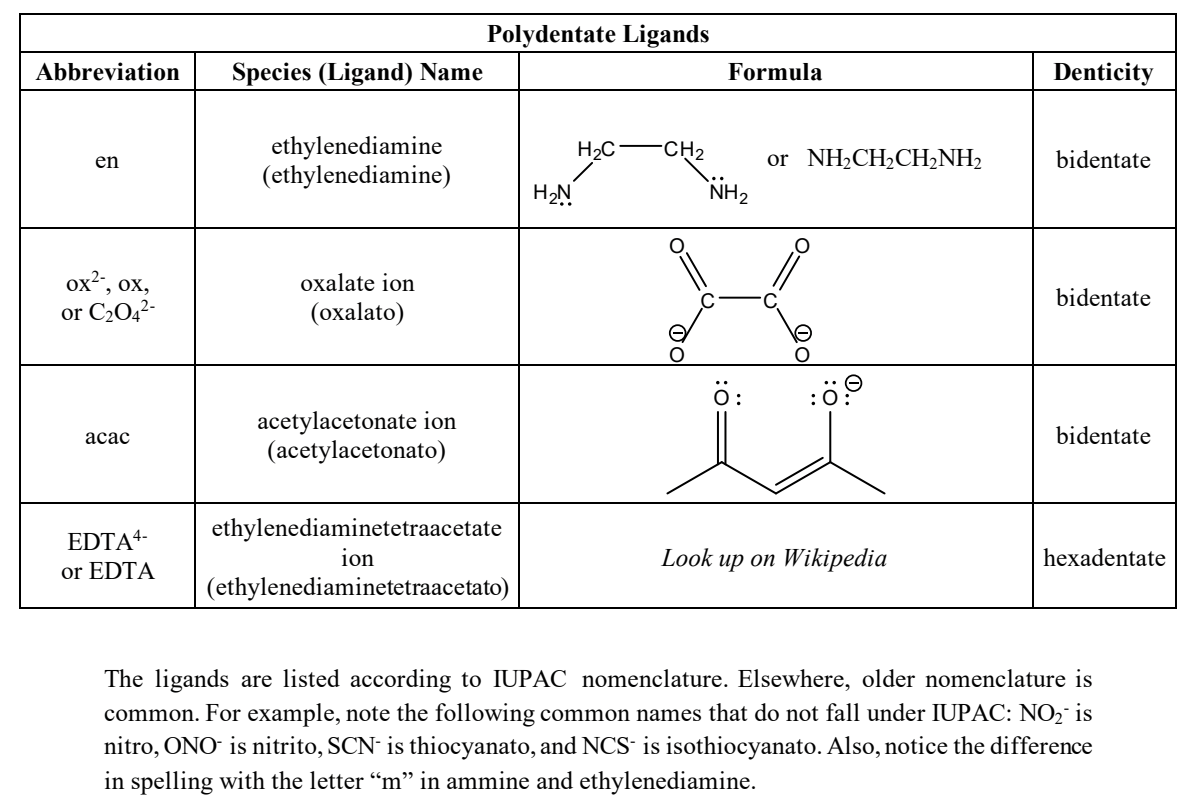
composite ligand

when writing the formula from the chemical name, list the ligands in WHAT order
alphabetical order
ligand naming