Carboxylic Acids & Polymers
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
physical properties of carboxylic acids
shorter chains dissolve in water due to H bonds, forms H bonds with other COOH molecules
biodiesel
veg oil + CH3OH + KOH (catalyst) → biodiesel
forms methyl ester (biodiesel) & glycerol
CH3COO(CH2)xCH3
soap
made from fat/oil + NaOH → sodium salt (soap) + glycerol
veg oil
is an ester
has unsaturated (double bonds) fatty acid chains - do not pack closely together
weaker van der waals - lower m.p.
can show E/Z isomerism
animal fat
is an ester
solid at room temp - saturated fatty acid chains, chains pack closely → stronger van der waals
ester hydrolysis - base and acid
acid: produces alcohol & COOH doesn't go to completion, eqm. established
base: sodium salt + alcohol eqm. disrupted goes to completion
acid anhydride + water
2x carboxylic acid
acid anhydride + ammonia
amide + carboxylic acid
acid anhydride + alcohol
ester + carboxylic acid
acid anhydride + 1o amine
N substituted amide + carboxylic acid
acyl chloride + water
carboxylic acid + white HCl fumes (vigorous reaction)
acyl chloride + ammonia
amide + white HCl fumes (vigorous reaction)
acyl chloride + alcohol
ester + white HCl fumes (vigorous reaction)
acyl chloride 1o amine
N substituted amide + white HCl fumes (vigorous reaction)
why are acid anhydrides used instead of acyl chlorides in industry?
less corrosive
doesn’t produce HCl fumes
doesn’t react as vigorously with water
is cheaper
what is heating under reflux used for?
heating volatile liquids without loosing the products
reaction to make aspirin
ethanoic anhydride + salicylic acid → aspirin + ethanoic acid
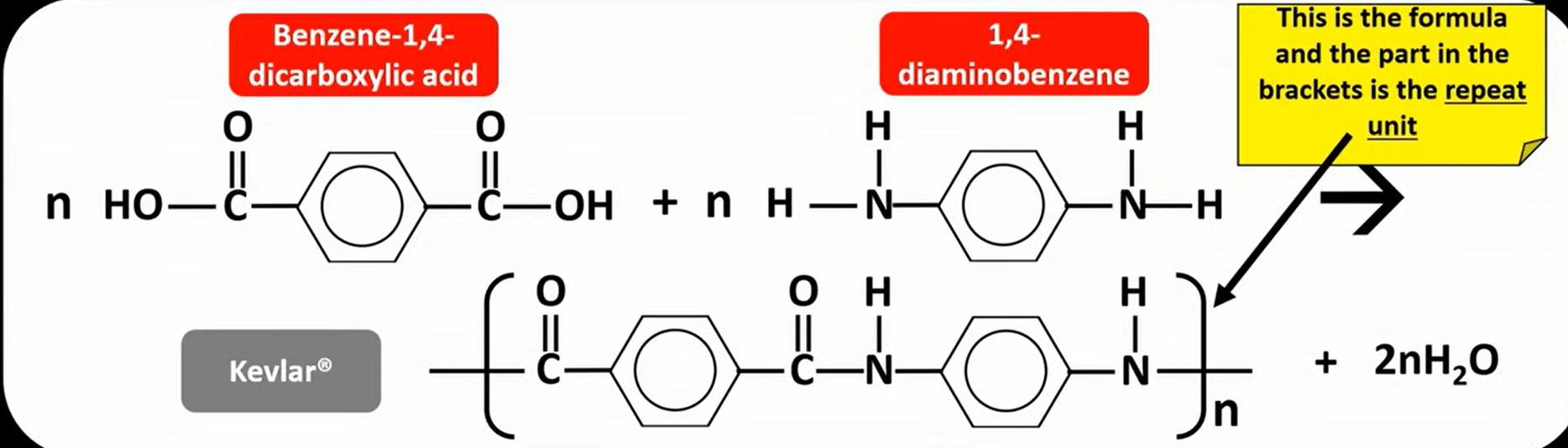
Kevlar
is a polyamide with an amide link/bond
made from benzene -1,4- dicarboxylic acid 1,4-diaminobenzene
used for bulletproof vests, car tyres - lightweight but very strong
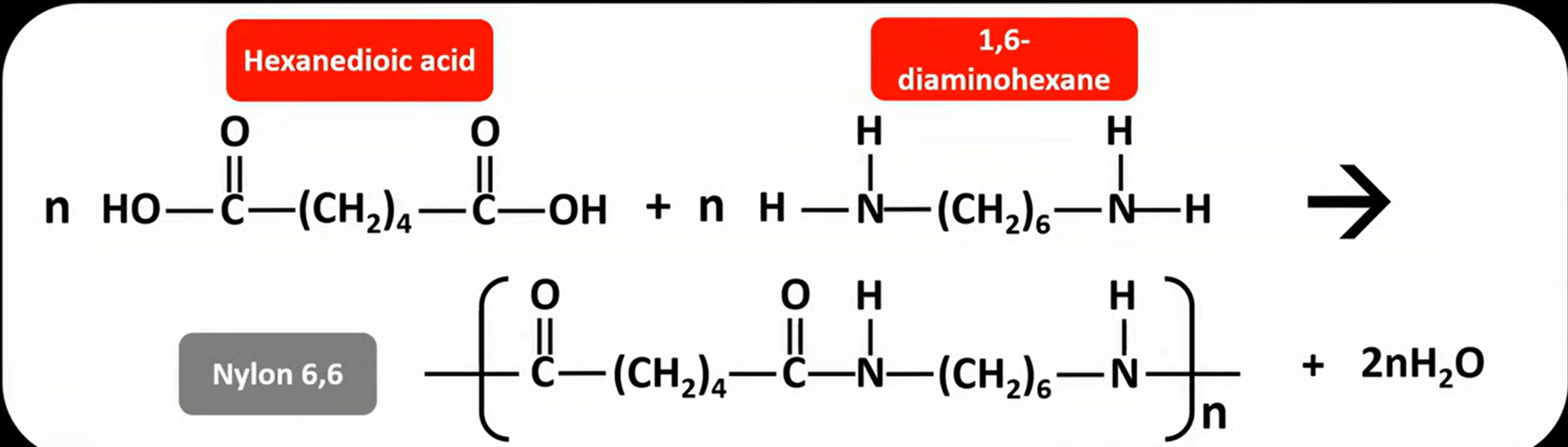
nylon 6,6
is a polyamide
hexanedioic acid + hexane-1,6-diamine
used in ropes, fabric, carpet & parachute clothing
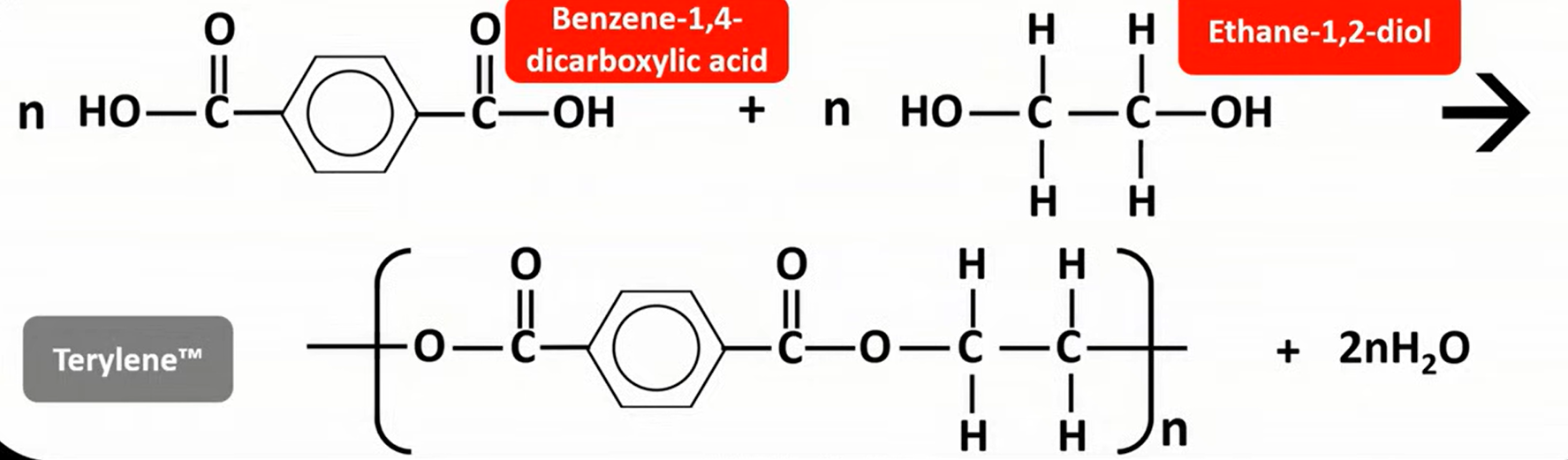
Terylene
is a polyester
benzene-1,4-dicarboxylic acid + ethane-1,2-diol
used in plastic drink bottles, sheeting & clothes
condensation vs addition polymers
condensation polymers have polar bonds - they can interact between chains forming H bonds
condensation have higher m.p. due to H bonds & pdpd
condensation are susceptible to attack from nucleophiles → biodegradable & water soluble
addition polymers (polyalkenes) - saturated, usually non-polar & unreactive
addition do not biodegrade quickly
why are addition polymers non-biodegradable?
non-polar, saturated
disposal: landfill
used for plastics that:
are difficult to recycle
difficult to separate from other materials
not enough plastic to extract to make extraction economically viable
→ landfill emits greenhouse gases, is expensive, not sustainable
→ risk of water contamination due to waste leaching
disposal: incineration
energy released can be used as power
release toxic fumes (PVC → HCl fumes)
flue gases added to neutralise acidity
disposal: recycling
remoulding plastics
cracking plastics using monomers to make organic feedstock
recycling pros & cons
pros: cheaper than making from scratch, less CO2 compared to incineration, reduces landfill use, preserves non renewable raw materials
cons: plastics can be contaminated, wide variety of plastics can be difficult to recycle, difficult to remake original plastic from recycled material, sorting & processing plastic more expensive than incineration
why does a polyester disintegrate when KOH is added?
delta positive C in polyester reacts with OH- ion
by nucleophilic addition-elimination