sys path derm and ocular
1/257
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
258 Terms
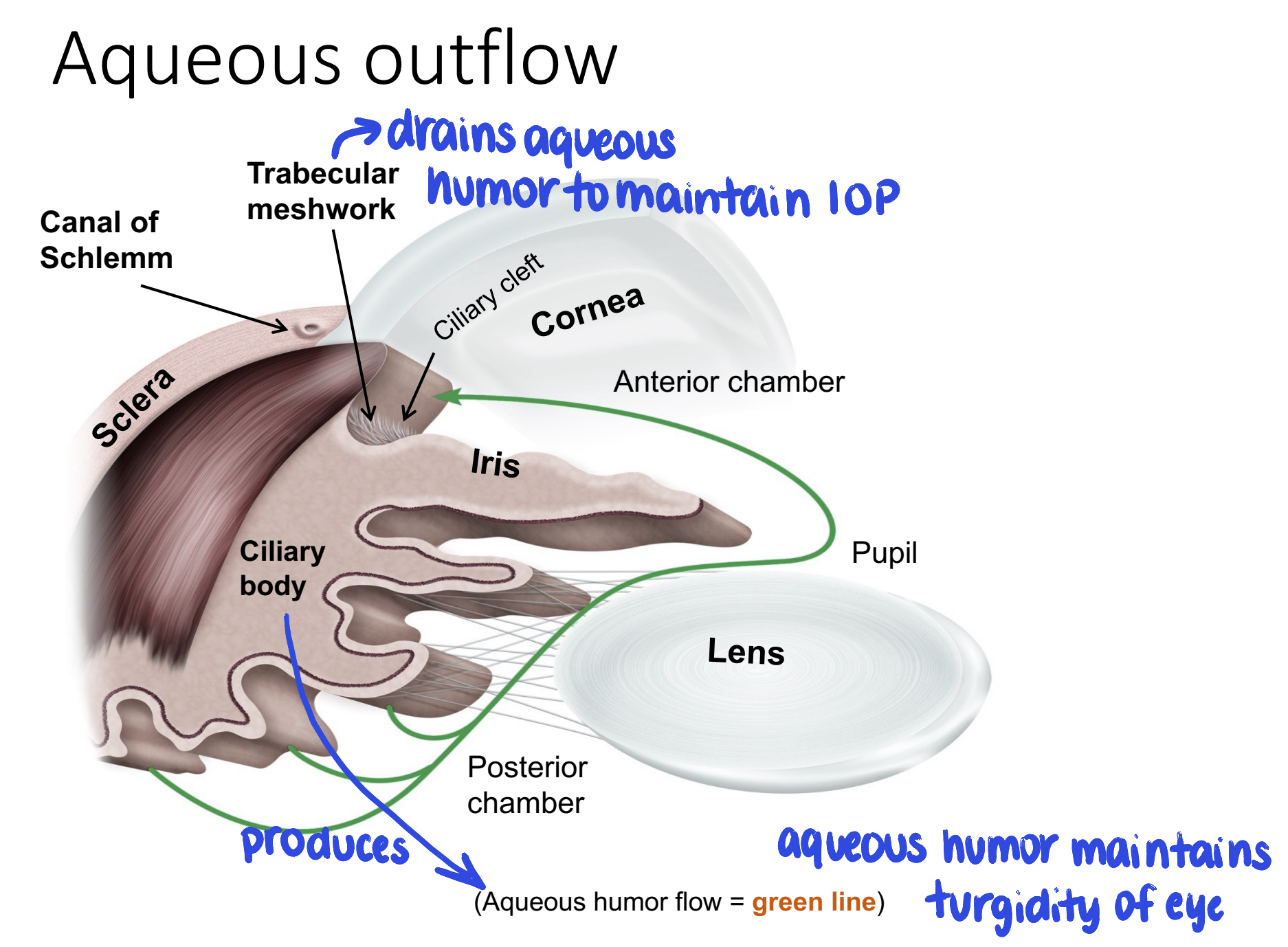
what structure produces aqueous humor? where does aqueous humor drain out of the eye?
aqueous humor is produced by the ciliary body
drains out of trabecular meshwork
how is corneal optical clarity maintaned?
avascular
nutrition provided by:
pre-corneal tear film
aqueous humor
dehydrated
maintained by:
hydrophobic corneal epithelium
corneal endothelial cation pump (Na/K ATPase) — pumps H2O out
regular array of collagen lamellae and keratocytes
steps in corneal epithelial wound healing
initial lag phase — 1 hr
sliding of epithelial cells at ulcer margin to cover defect
replication of germinal cells at limbus within 24 hrs
uncomplicated 2mm ulcer heals in a few days
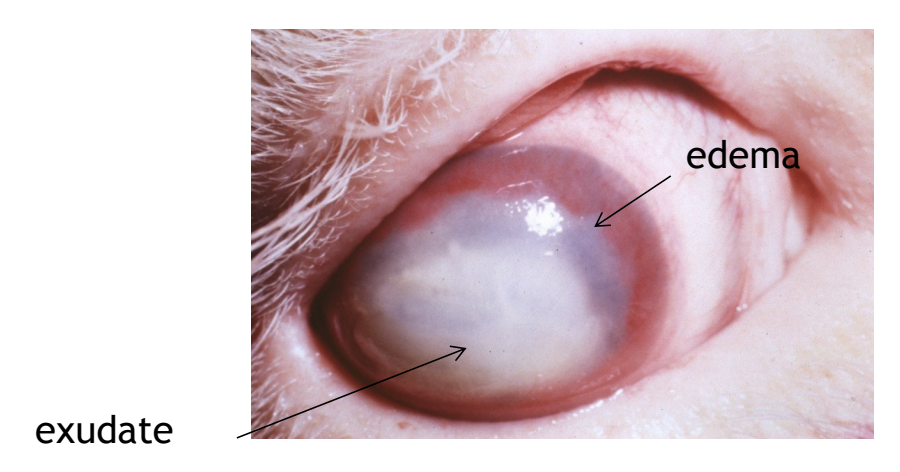
causes of corneal edema
epithelial defects (ulcers) — superficial stromal edema
stromal vascularization — leaky blood vessels
endothelial defects — deep stromal edema
endothelialitis
canine adenovirus-1 vaccine reaction (“blue eye”)
malignant catarrhal fever
endothelial degeneration — age-related
endothelial cell damage
clinical appearance of ulcerative keratitis (corneal ulcers)
corneal edema
superficial neovascularization
inflammatory cell infiltrates
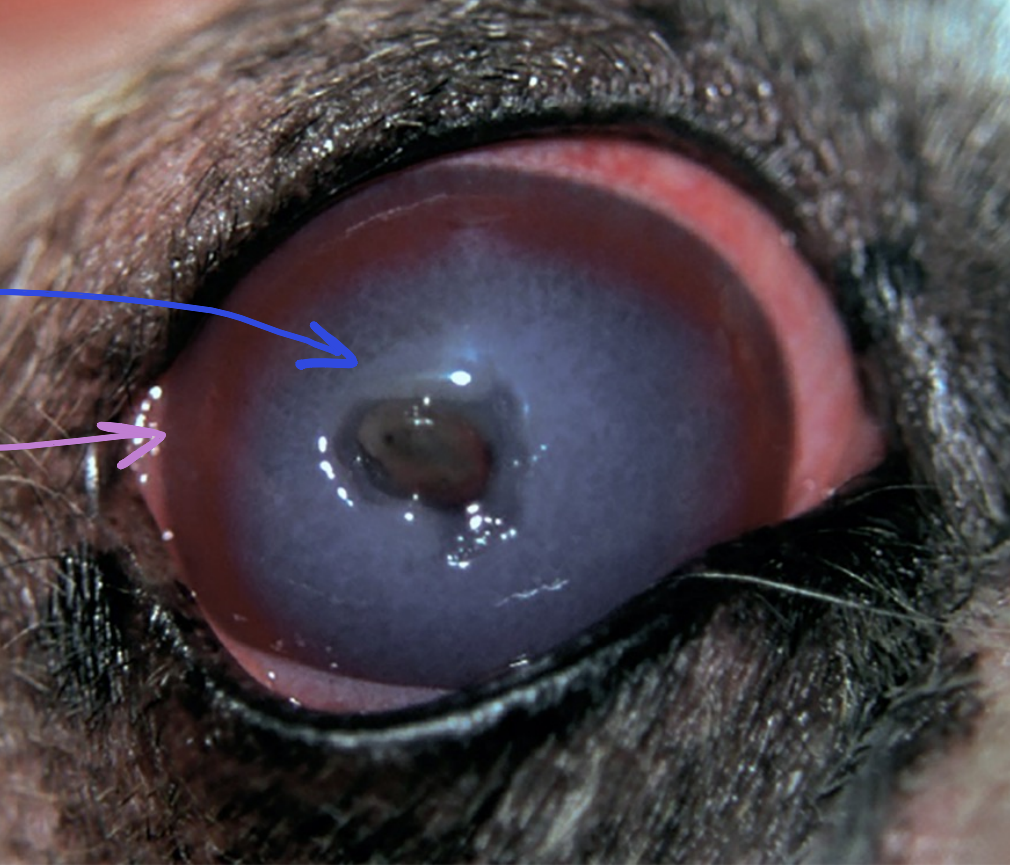
which stain can be used to diagnose corneal ulcers?
fluorescein stain — exposed stroma stains green
underlying causes of ulcerative keratitis
trauma
conformational lid defects (ex. entropion)
hair irritation
foreign body
exposure
lagophthalmos, exophthalmos
CN V or VII defects
keratoconjunctivitis sicca (dry eye)
primary or secondary infection
causative agent of infectious bovine keratoconjunctivitis
moraxella bovis (gram negative bacillus)
primary corneal pathogen with cytotoxic effects on neutrophils & corneal epithelial cells
highly contagious
predisposing factors to infectious bovine keratoconjunctivitis
summer months
corneal irritation (fly vectors, UV radiation, long grasses)
Hereford & Hereford-cross breeds
younger cattle (higher morbidity)
cattle housed inside have higher infection rate + longer duration but milder clinical disease
clinical appearance of infectious bovine keratoconjunctivitis
unilateral (initially) central corneal ulcer
intense inflammatory cell infiltrates may develop into a stromal abscess
superficial neovascularization
outcomes of infectious bovine keratoconjunctivitis
corneal healing with scarring
or
corneal perforation with iris prolapse (blindness)
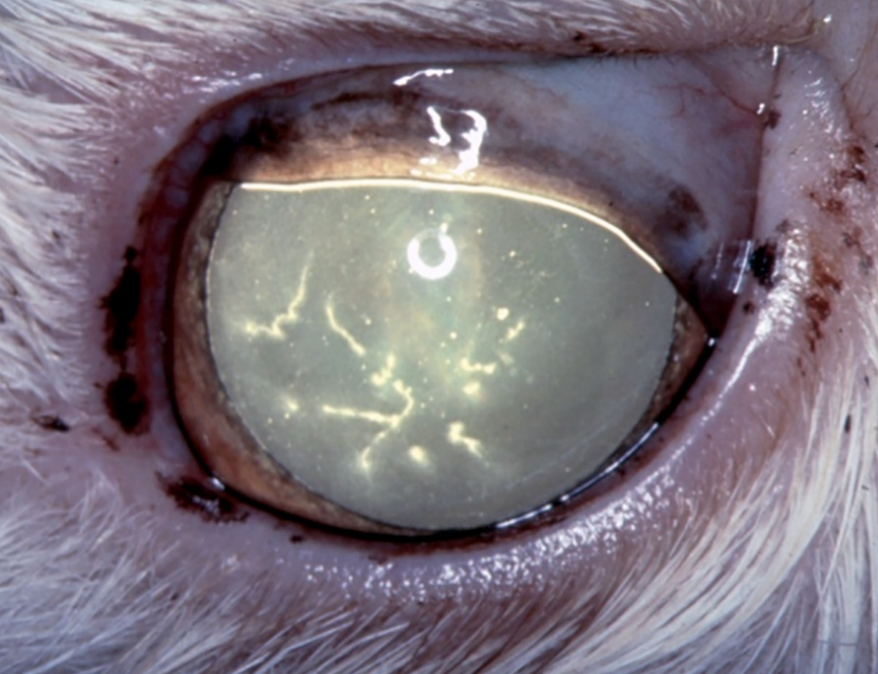
clinical appearance of feline herpesvirus-1 keratitis
formation of dendritic ulcers (linear ulcers) — characteristic
pathogenesis of feline herpesvirus-1 keratitis
virus reactivation and recrudescence
FHV-1 viral replication in corneal epithelial cells → cell death
may develop secondary bacterial infections
which stain is used to visualize FHV-1 keratitis?
rose bengal stain — epithelial defect may not extend to basement membrane; necessary to visualize epithelial lesions
causative agents of equine fungal keratitis
opportunistic pathogens — part of normal conjunctival flora of the horse
vary by geographic location
aspergillus
fusarium
lesions associated with equine fungal keratitis
ulcerative keratitis, frequently deep stromal
deep stromal abscesses
difficult to treat; may progress to corneal perforation & iris prolapse
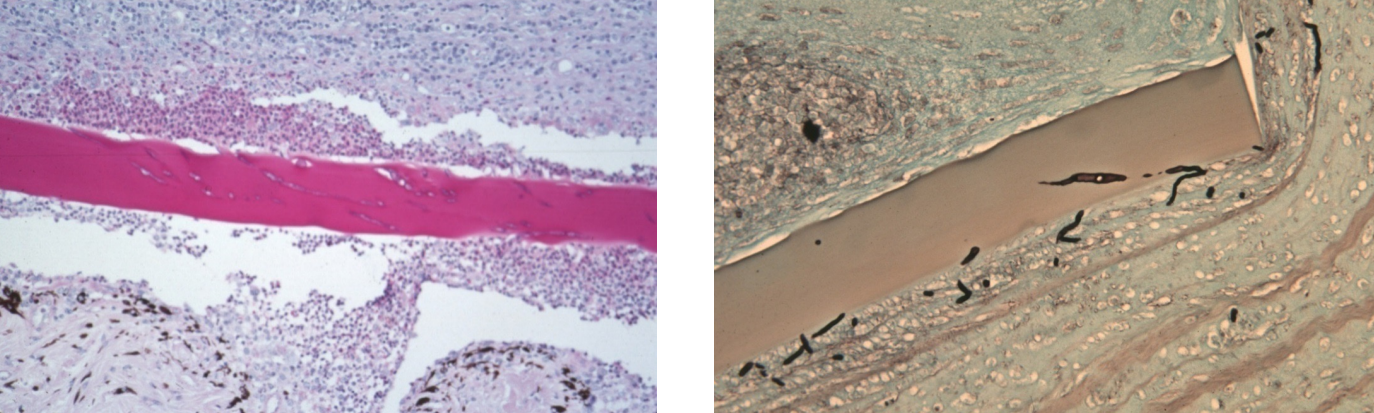
microscopic findings of equine fungal keratitis
involvement of posterior 1/3 of cornea
fungal hyphae found within descemet’s membrane (attracted to carbohydrates) & deep corneal stroma; rarely extend into anterior chamber
breaks in descemet’s membrane with pyogranulomatous inflammation; occasional multinucleated giant cells
often very little corneal neovascularization
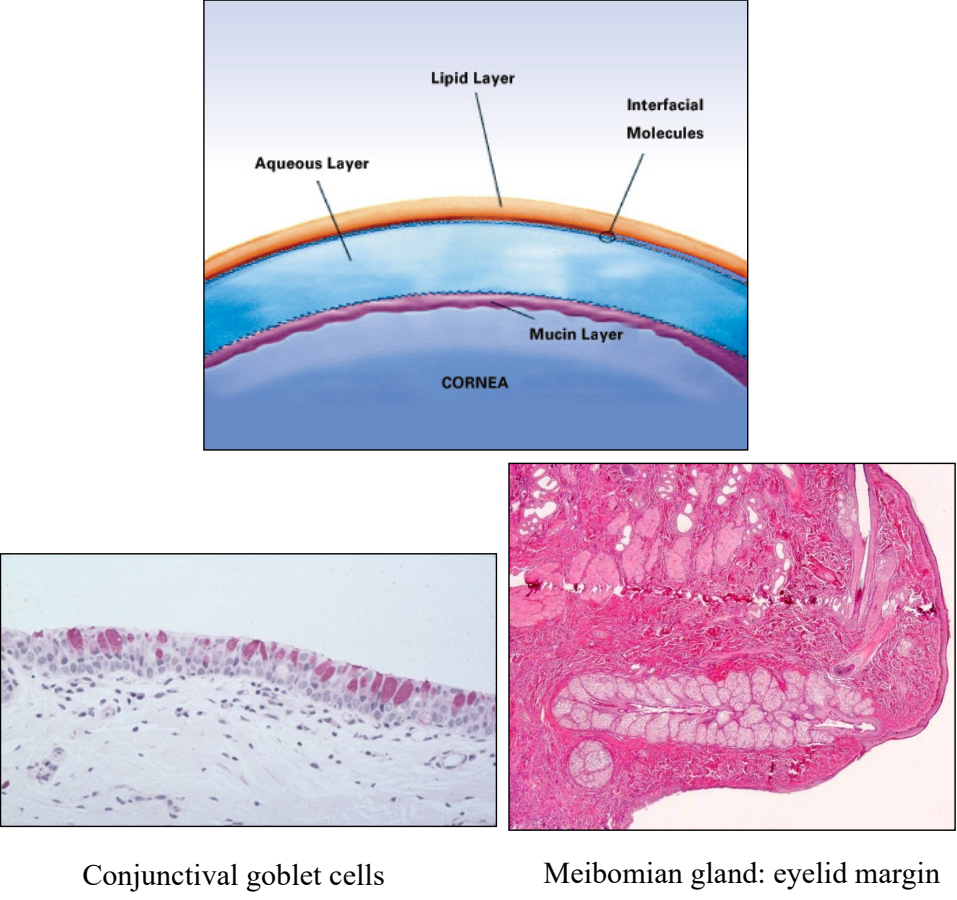
what are the components & functions of the tear film?
lipid layer (meibomian glands of the eyelids): forms optically smooth surface & prevents evaporation
aqueous layer (orbital lacrimal gland & gland of 3rd eyelid): provides nutrients & Ig to the avascular cornea
mucous layer (goblet cells of the conjunctival epithelium): adsorbs aqueous layer to corneal epithelium
production of what layer of the tear film is decreased with keratoconjunctivitis sicca (dry eye)?
aqueous portion
consequences of corneal desiccation
acute: corneal epithelium becomes ulcerated
chronic: corneal epithelium undergoes “epidermalization”
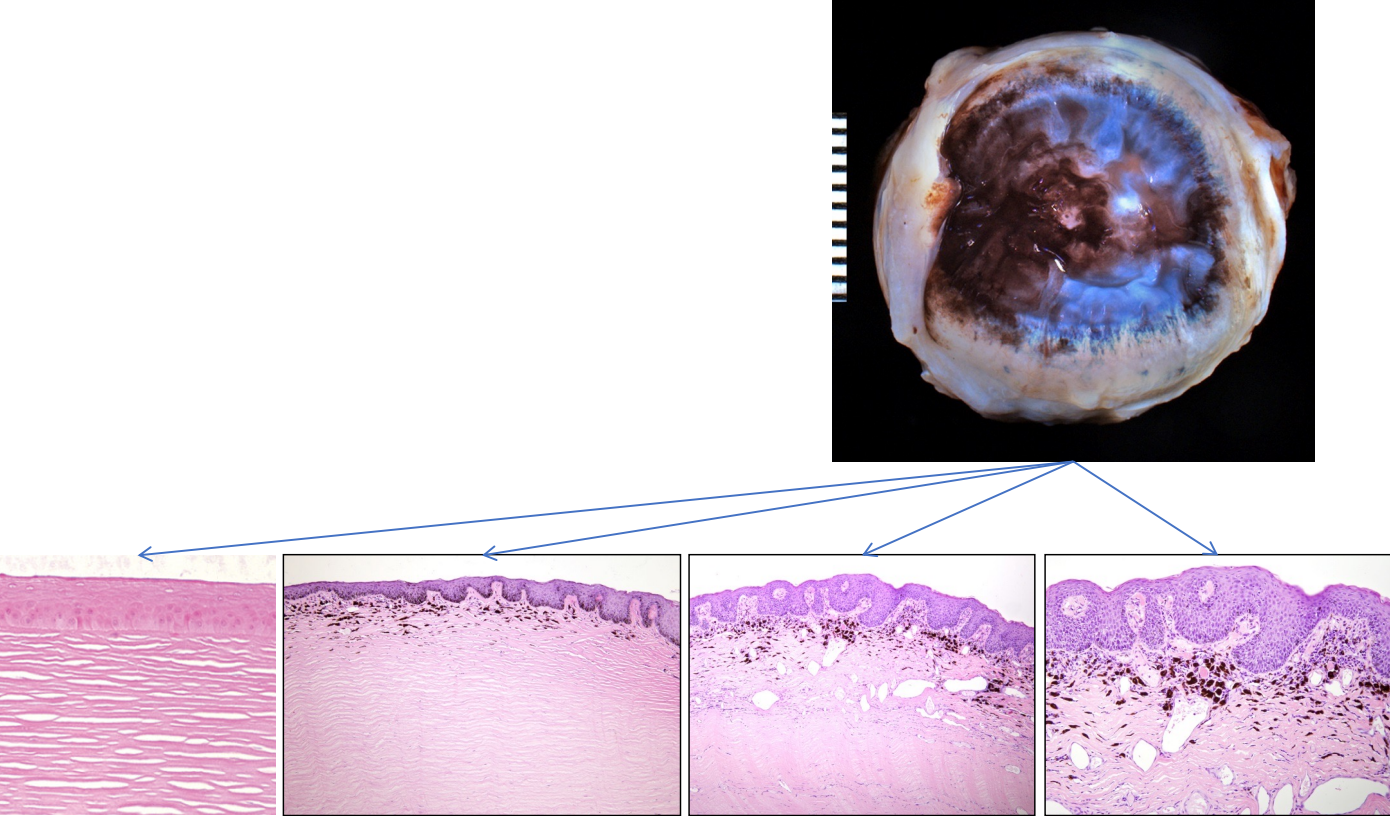
features of corneal “epidermalization”
not specific to KCS — any chronic, end-stage corneal disease
epithelial thickening with keratinization (↑ opacity)
rete ridge formation
pigmentation of the epithelium & superficial stroma
superficial neovascularization
superficial inflammation
goblet cell hyperplasia (conjunctiva)
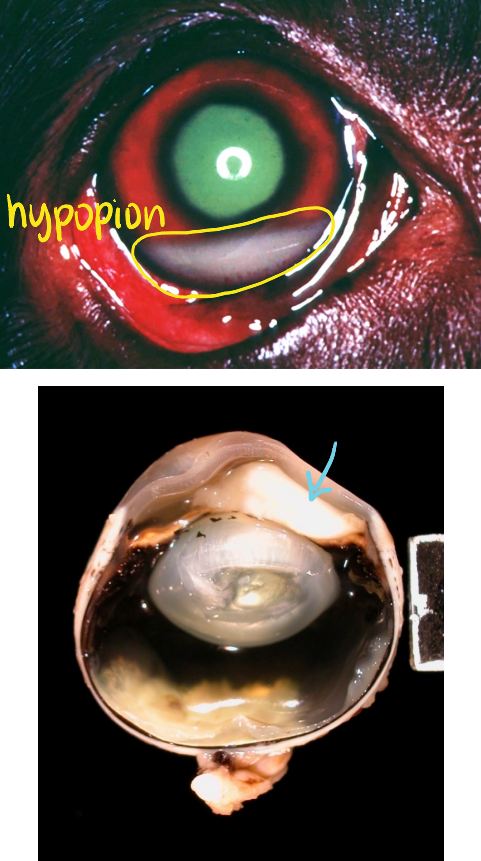
what is aqueous flare?
turbid aqueous humor, caused by the presence of:
fibrin & other proteins
inflammatory cells
seen with uveitis
hypopion
neutrophils (pus) in anterior chamber
what are consequences of intraocular inflammation?
corneal edema
effects on corneal endothelial cell function
pre-iridal fibrovascular membranes (PIFM)
synechia (adhesion)
block aqueous drainage
secondary glaucoma
retinal detachment
protein exudation ± inflammatory cells from choroidal vessels into subretinal space
how do preiridal fibrovascular membranes (PIFM) form?
angiogenic factors are released into the eye & stimulate iridal blood vessels to proliferate on the anterior surface of the iris
fragile vessels may bleed spontaneously; leaky!
hyphema = blood in anterior chamber
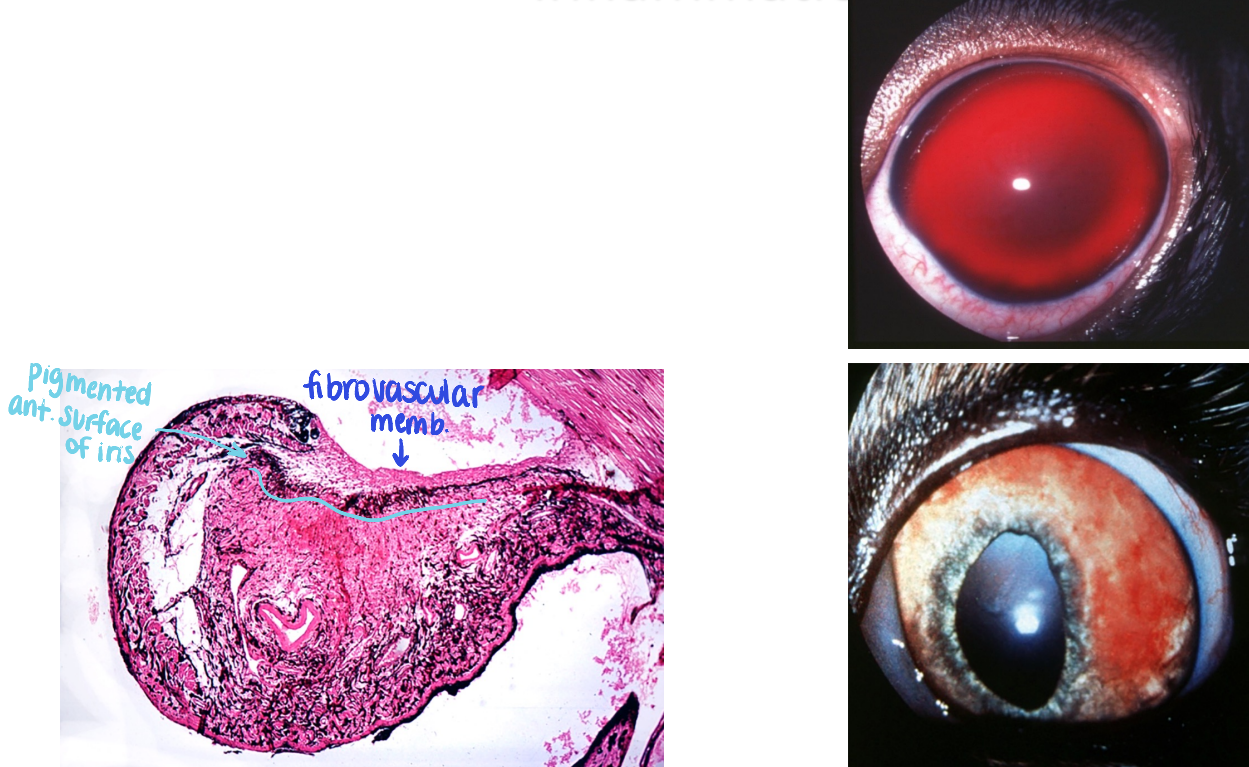
different types of synechia (adhesion)
anterior: adhesion to the cornea
posterior: adhesion of the lens
peripheral anterior: over the iridocorneal angle
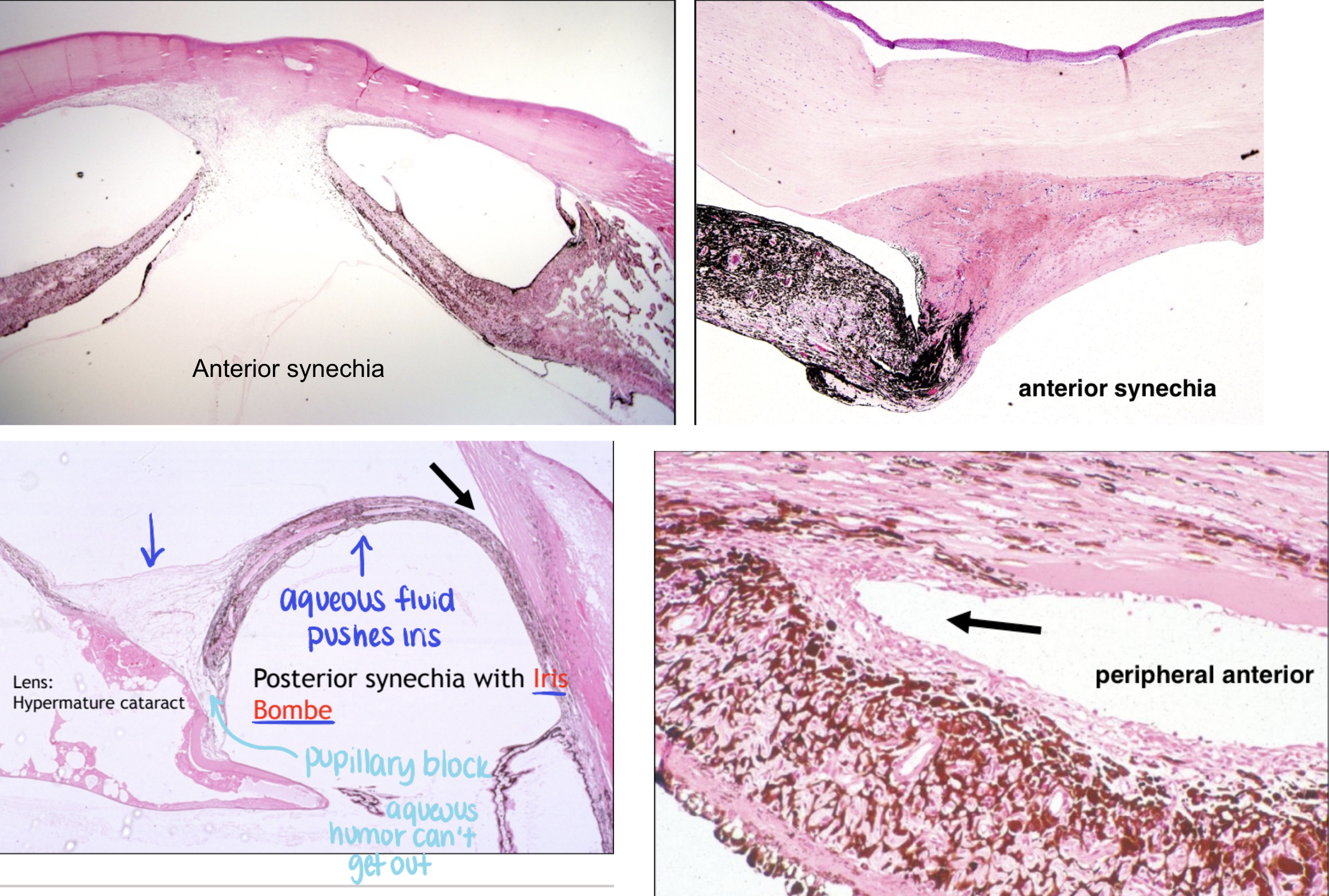
how can secondary glaucoma result from intraocular inflammation?
accumulation of inflammatory cells & fibrin in the iridocorneal angle may obstruct aqueous outflow
peripheral anterior synechia caused by preiridal fibrovascular membrane can close the angle
pupillary block: anterior or posterior synechia
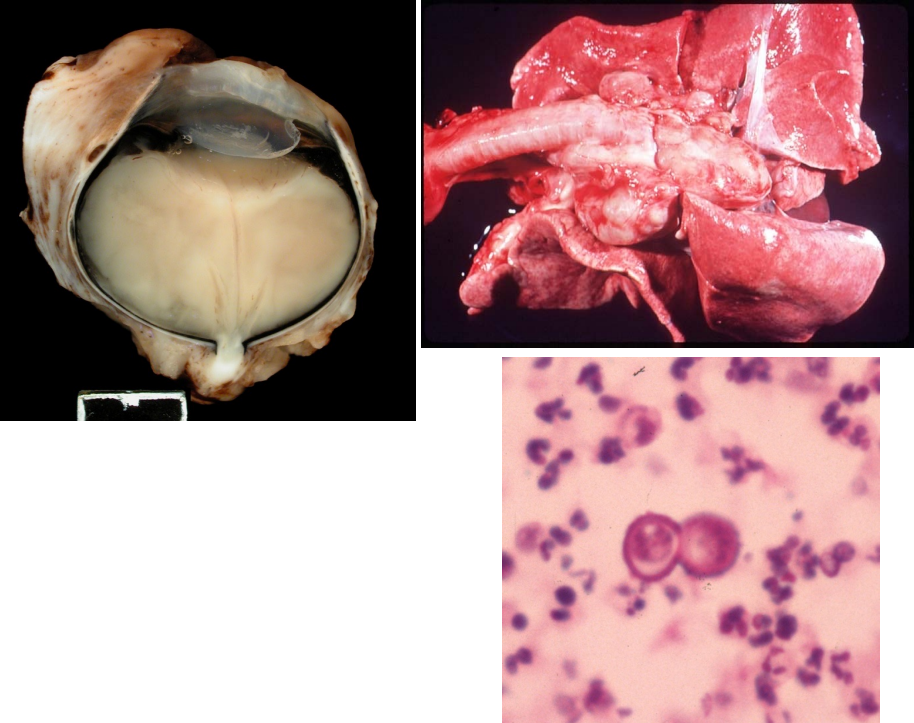
where is blastomycosis common? which species does it affect?
common in wisconsin
dogs > cats > people (not zoonotic)
pathologic findings of blastomycosis
pyogranulomatous endophthalmitis & chorioretinitis
retinal detachment
thick-walled yeast, broad-based budding
systemic disease: lungs, skin, bone, eye
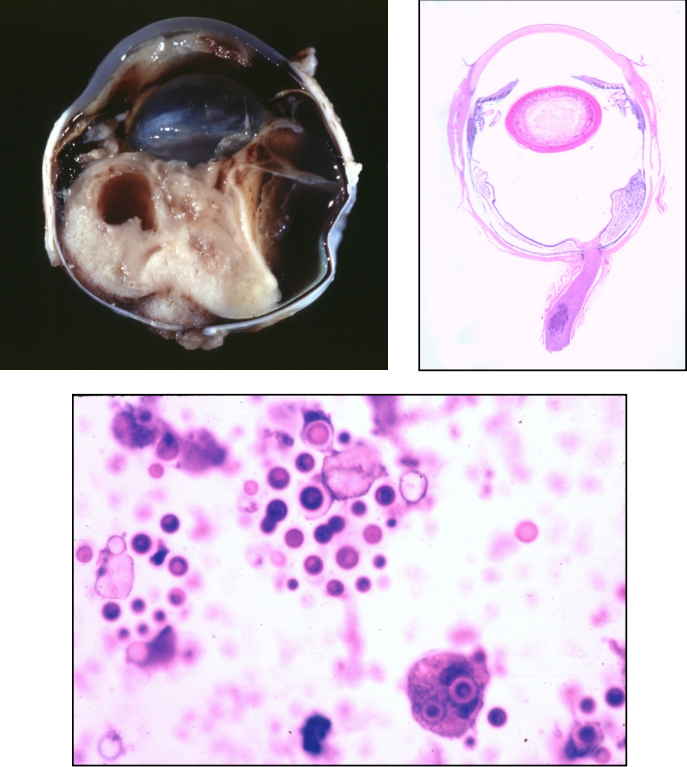
where is cryptococcosis common? which species does it affect?
more widespread than blastomycosis
associated with river systems, pigeon droppings
more common in cats than dogs
pathologic findings of cryptococcosis
pyogranulomatous chorioretinitis
also: nasal cavity, cutaneous involvement, brain
thin-walled yeast, narrow-based budding, thick capsule
where is coccidioidomycosis common?
valley fever (AZ, NM, CA)
pathologic findings of coccidioidomycosis
pyogranulomatous chorioretinitis
primarily pulmonary infections
may disseminate: long bones, heart, CNS, eyes
round spherules, filled with small endospores
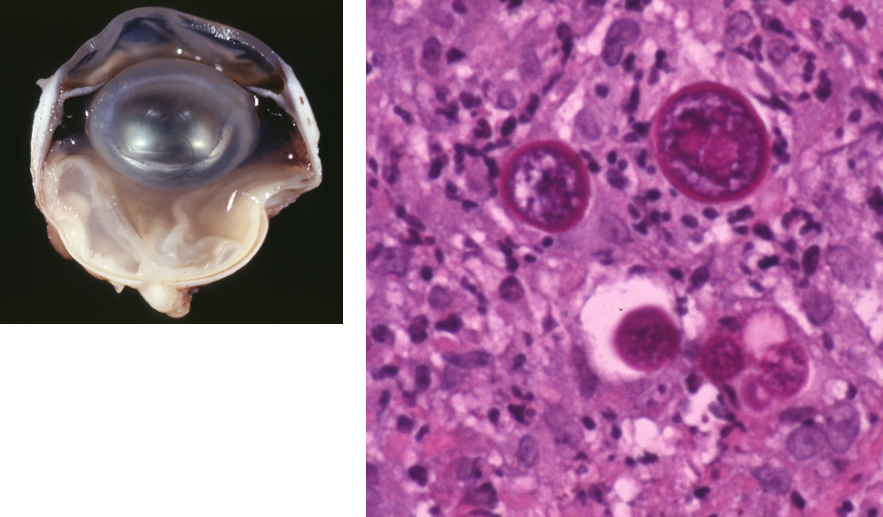
where is histoplasmosis common? what species does it affect?
widely distributed, esp. Ohio & Mississippi river valleys
soil fungus associated with bird & bat droppings
most common in dogs and cats
pathologic findings of histoplasmosis
pyogranulomatous chorioretinitis
primary pulmonary infection with dissemination to GI, liver
small round yeast, intracytoplasmic in macrophages
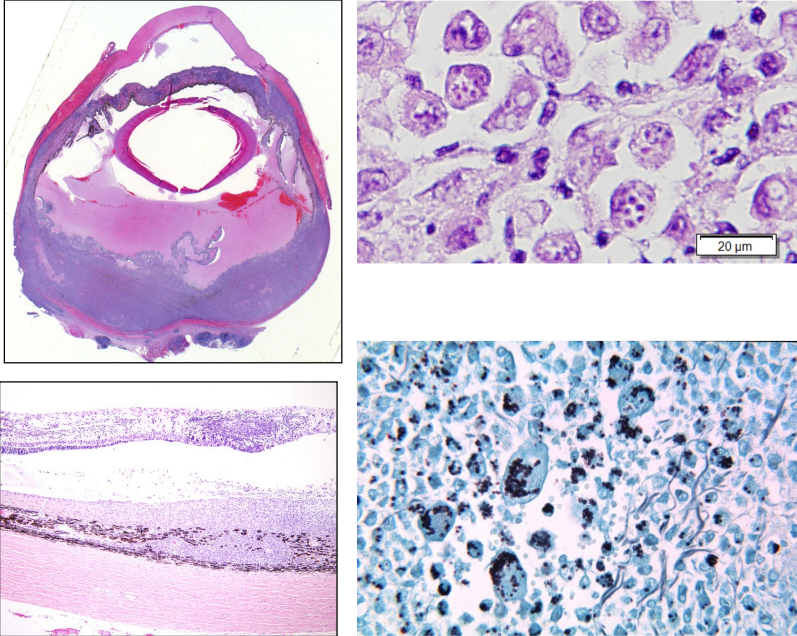
which species is affected by lymphoplasmacytic uveitis?
idiopathic disease of cats
clinical signs of lymphoplasmacytic uveitis
anterior uveitis (bilateral)
aqueous flare
hypopion (pus in anterior chamber)
rubeosis iridis (fibrovascular membrane)
anterior & posterior synechia
iridal nodules — lymphoid follicles
may obliterate ICA → secondary glaucoma
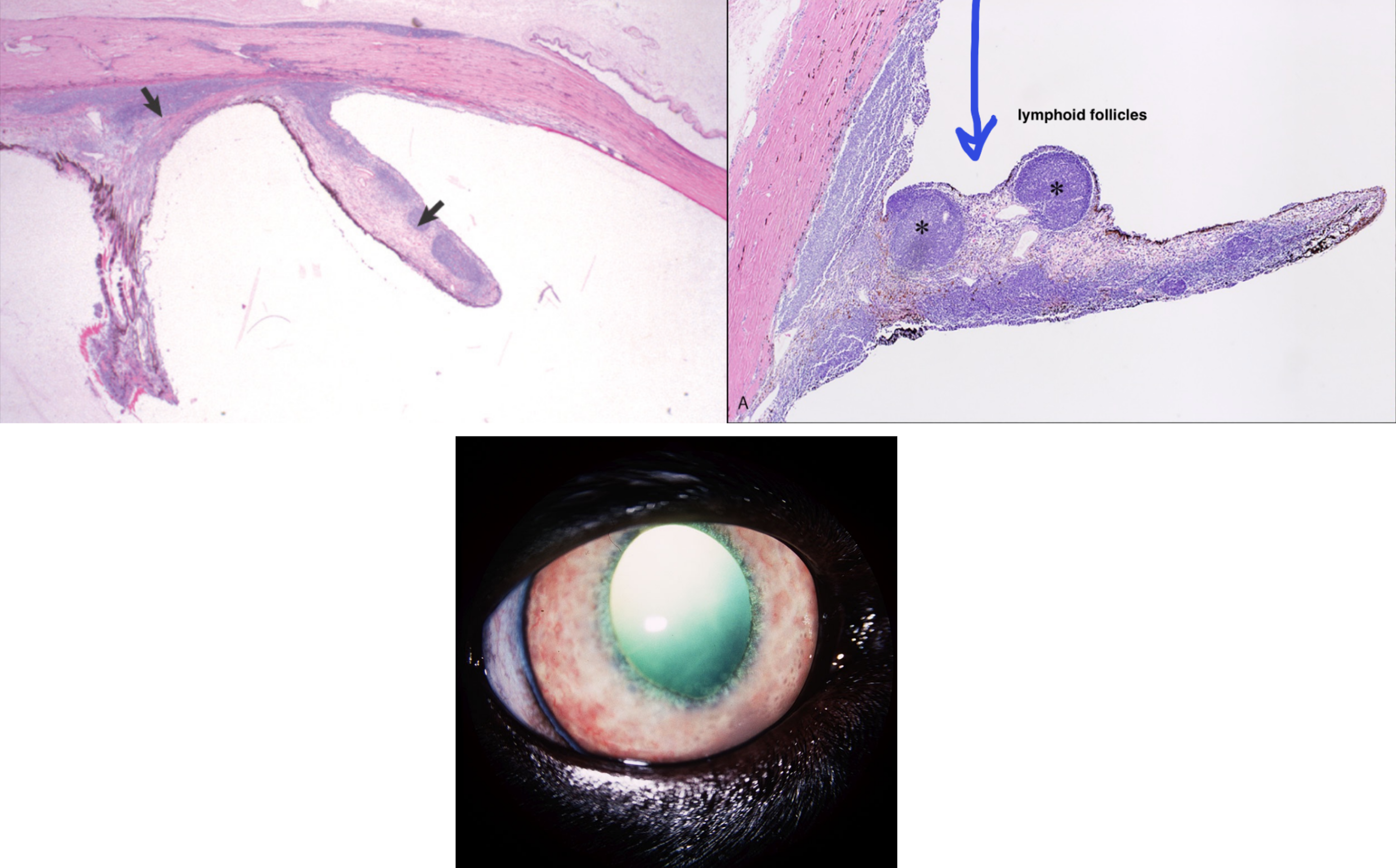
what is the most common cause of blindness in horses?
equine recurrent uveitis — recurrent episodes of anterior & posterior uveitis, typically progressive
equine recurrent uveitis pathogenesis
multifactorial, complex, controversial
inciting factors
most common: Leptospira infection → antigens cross react with equine cornea and lens antigens
immune mediated mechanisms result in recurrent bouts, even in the absence of the initiating factor
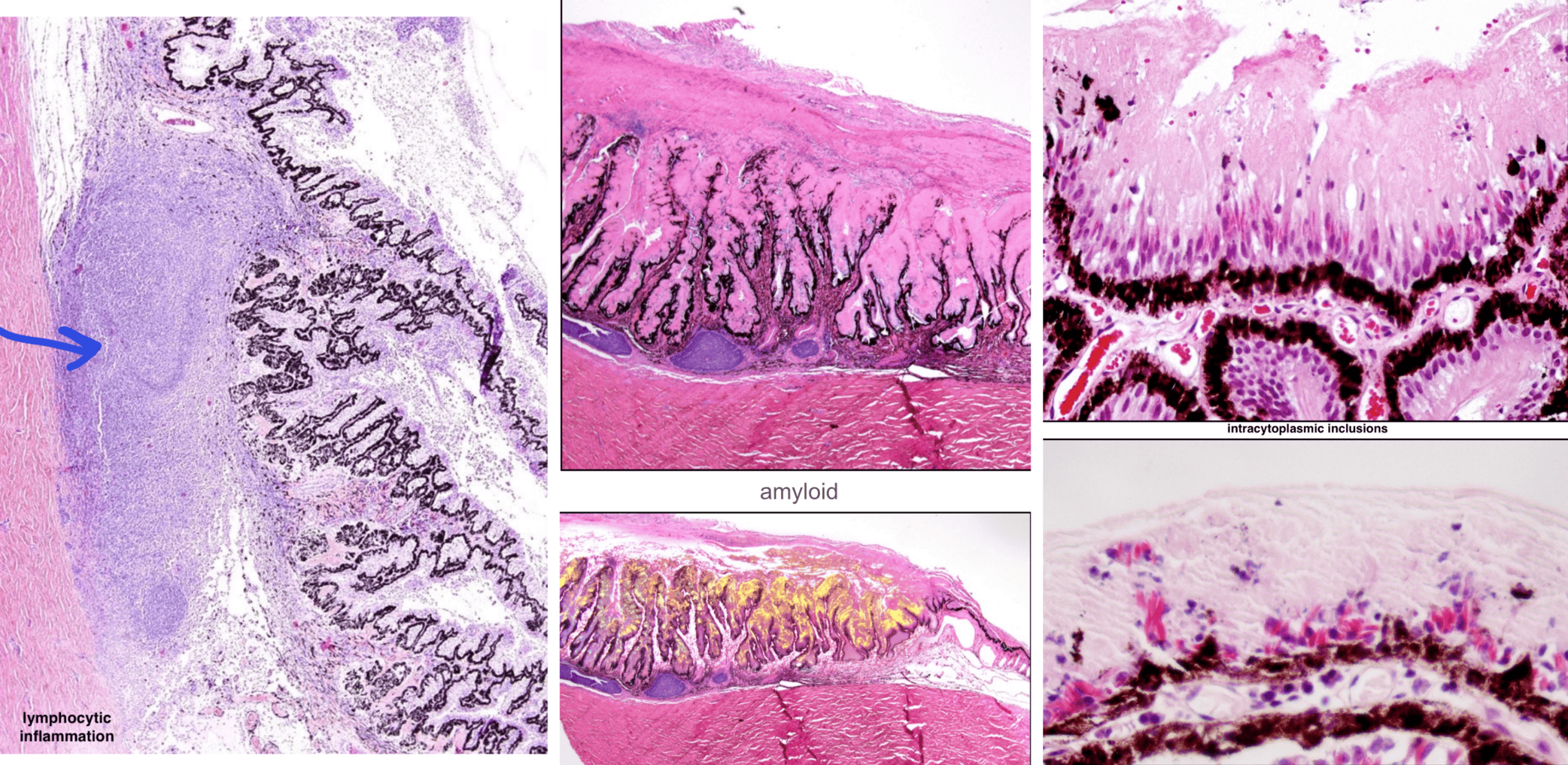
histopathologic findings of equine recurrent uveitis
lymphocytic inflammation in the uvea, sometimes follicular
eosinophilic (hyalinized) membrane coats the ciliary processes — amyloid
intracytoplasmic inclusions in non-pigmented ciliary body epithelium
causes of impaired aqueous outflow
fibrovascular membranes
synechia — anterior, posterior, peripheral anterior
pupillary block — iris bombe
clogging of trabecular meshwork with cells
obliteration of iridocorneal angle by neoplasia
goniodysgenesis
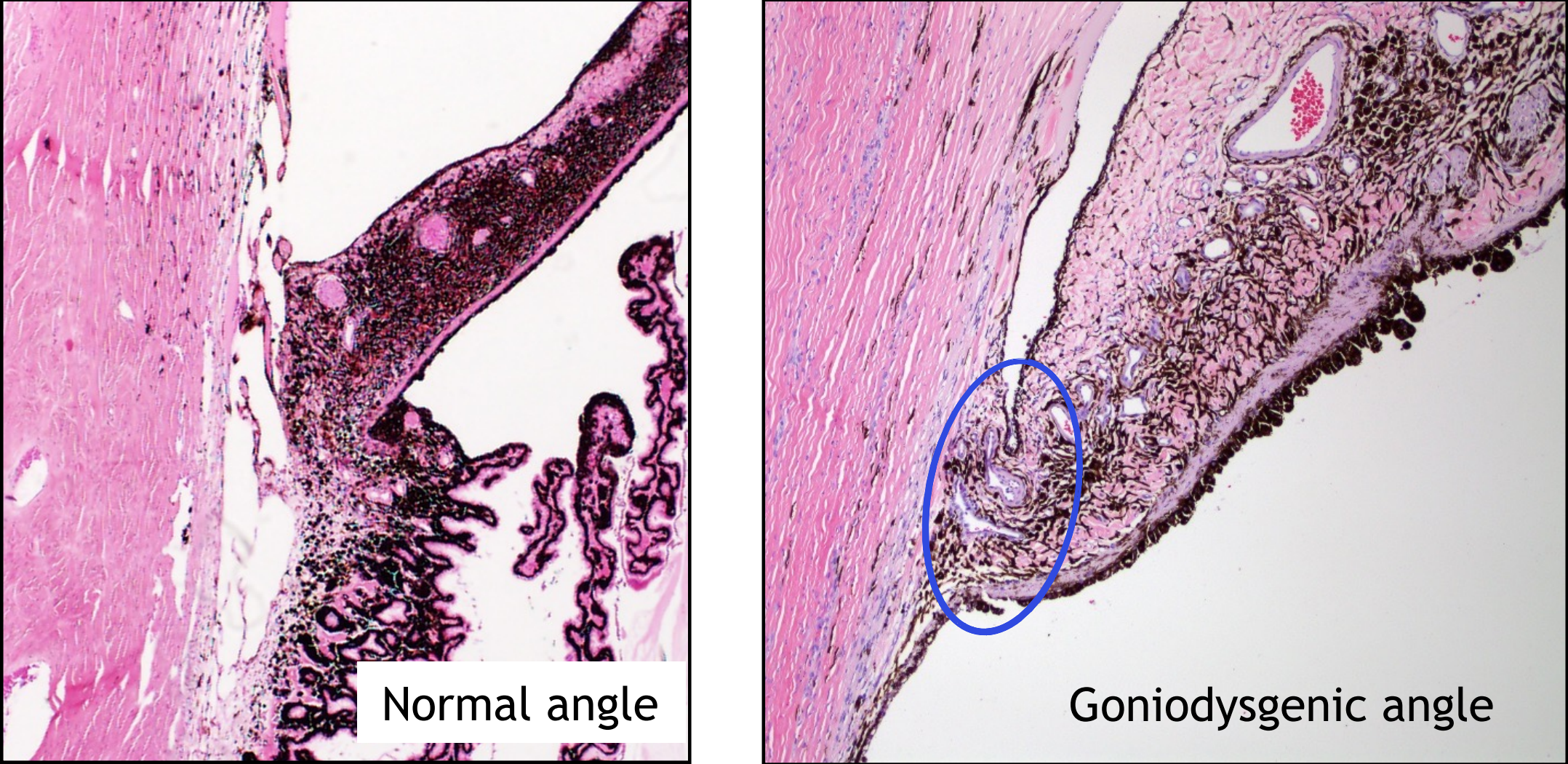
what is goniodysgenesis?
congenital abnormality — malformation of the iridocorneal angle structures
predisposes animal to development of glaucoma at any age
breed dispositions to goniodysgenesis
cocker spaniel, bassett hound, samoyed, great dane, chow chow, norwegian elkhound
(any breed can be affected)
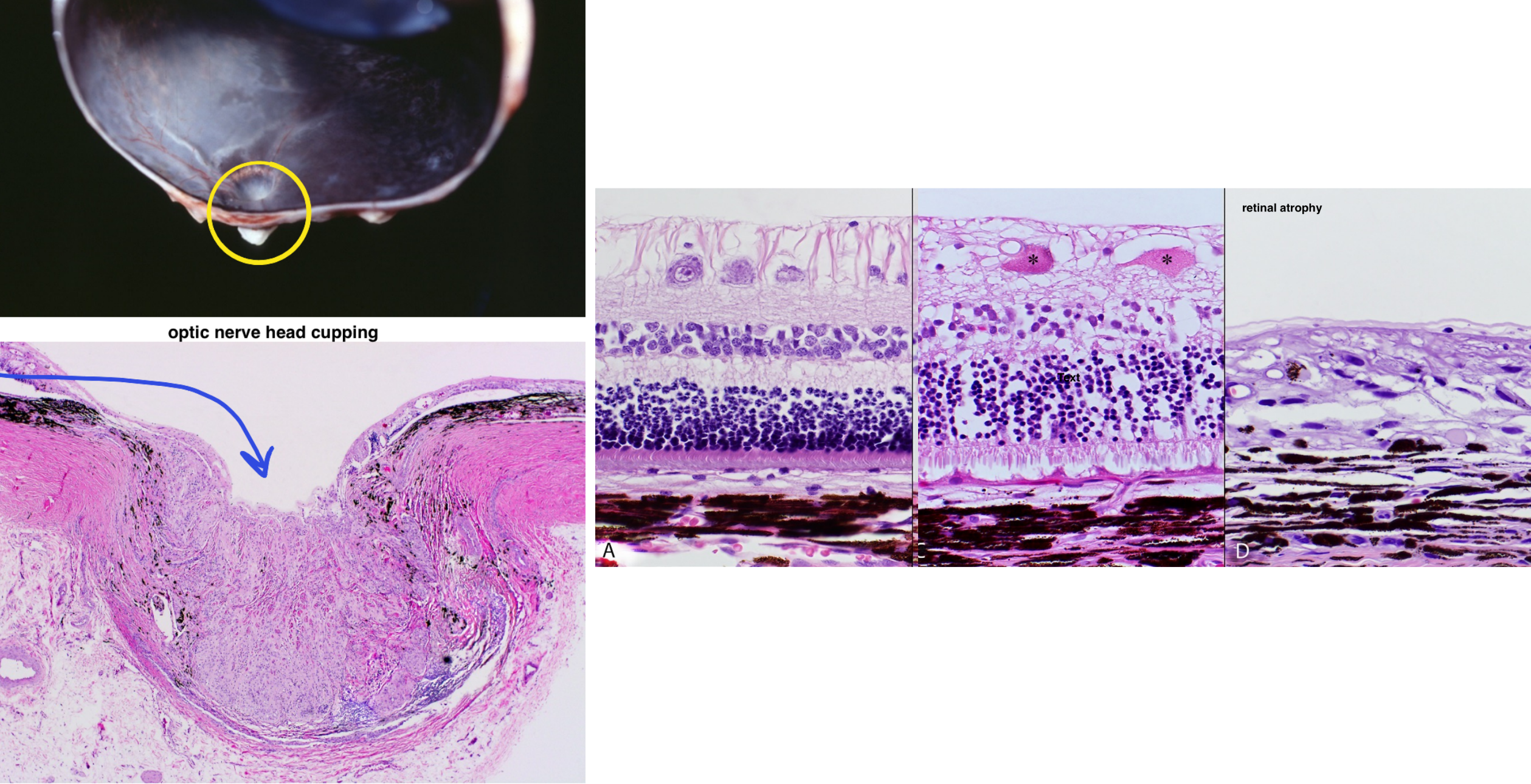
morphologic features of glaucoma
retinal atrophy
decreased numbers of retinal ganglion cells
atrophy & gliosis of the never fiber layer
full thickness retinal atrophy (in dogs only)
cupping of the optic nerve head
end stage changes
buphthalmia (bulging)
lens luxation
phthisis bulbi (eye shrinks, atrophies)
what is the most common intraocular tumor in the dog?
melanocytoma/malignant melanoma
distribution of melanocytoma/malignant melanoma
anterior uveal tract — most common
choroid
epibulbar/limbar
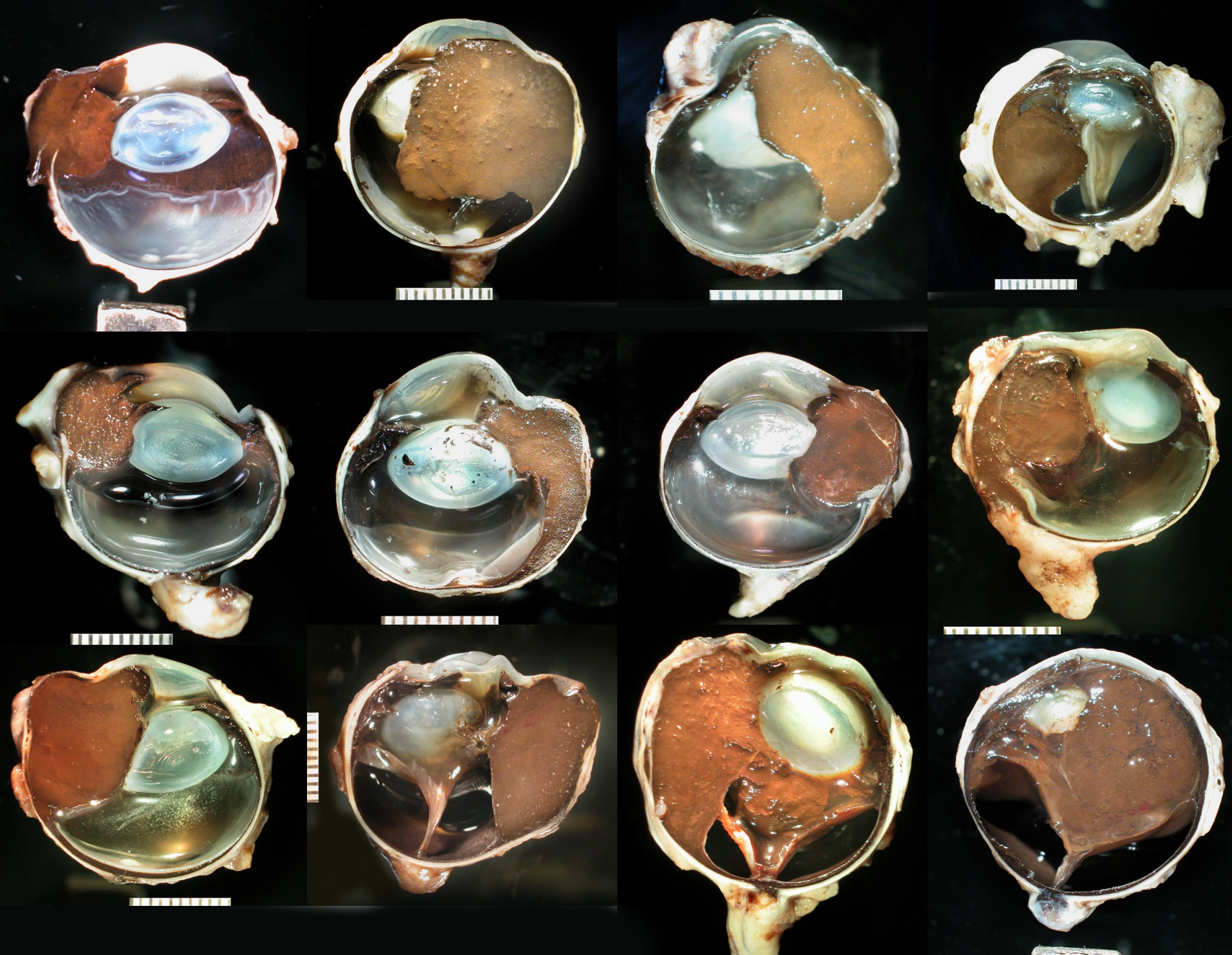
biologic behavior of melanocytoma/malignant melanoma
destruction of globe — can fill globe, extend through sclera
melanocytoma does not metastasize (benign)
malignant melanoma infrequently metastasizes
melanocytoma can transform to malignant melanoma
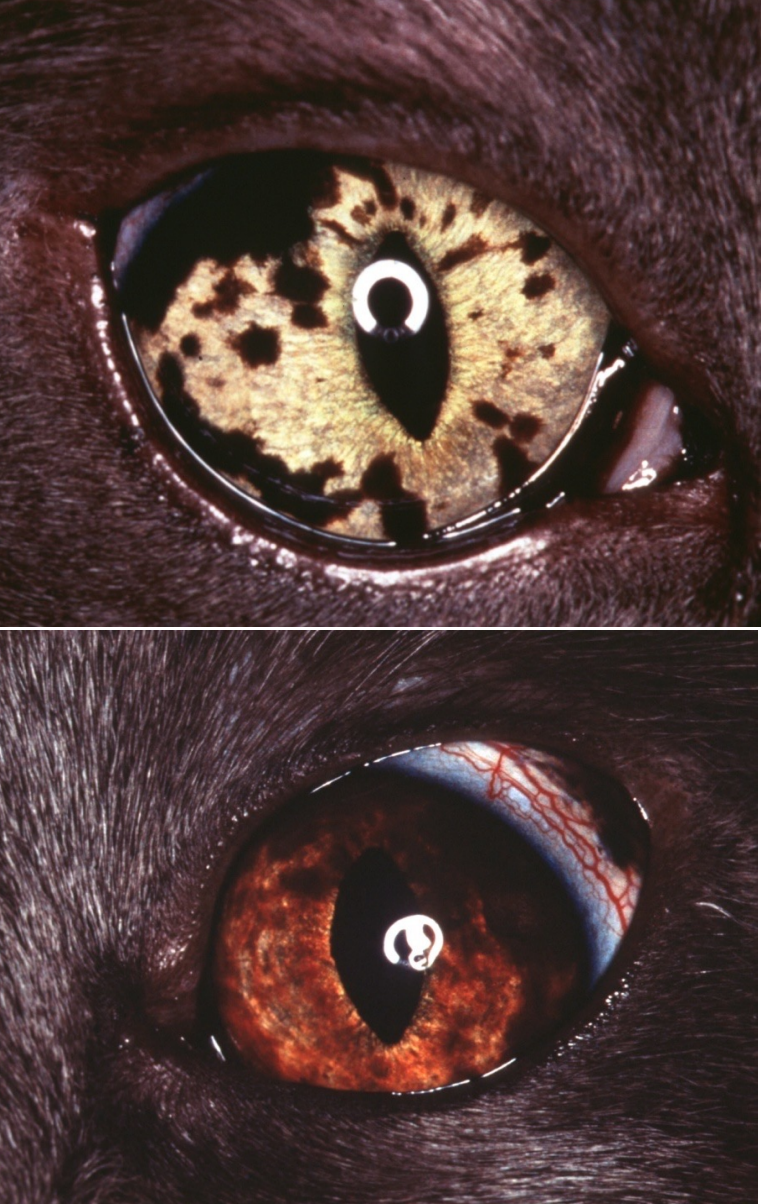
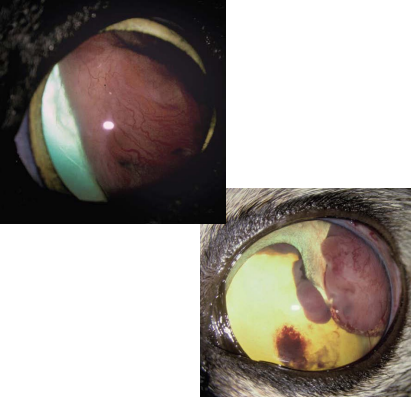
what is the most common intraocular tumor in the cat?
feline diffuse iris melanoma (FDIM)
gross appearance/biologic behavior of feline diffuse iris melanoma (FDIM)
gross appearance:
begins as hyperpigmented foci (freckles) on the iris, over months to years coalesce and form masses involving the iris, ciliary body, choroid
may invade the iridocorneal angle & cause secondary glaucoma
distant metastasis may occur infrequently
most commonly to liver, lung, kidney, and spleen
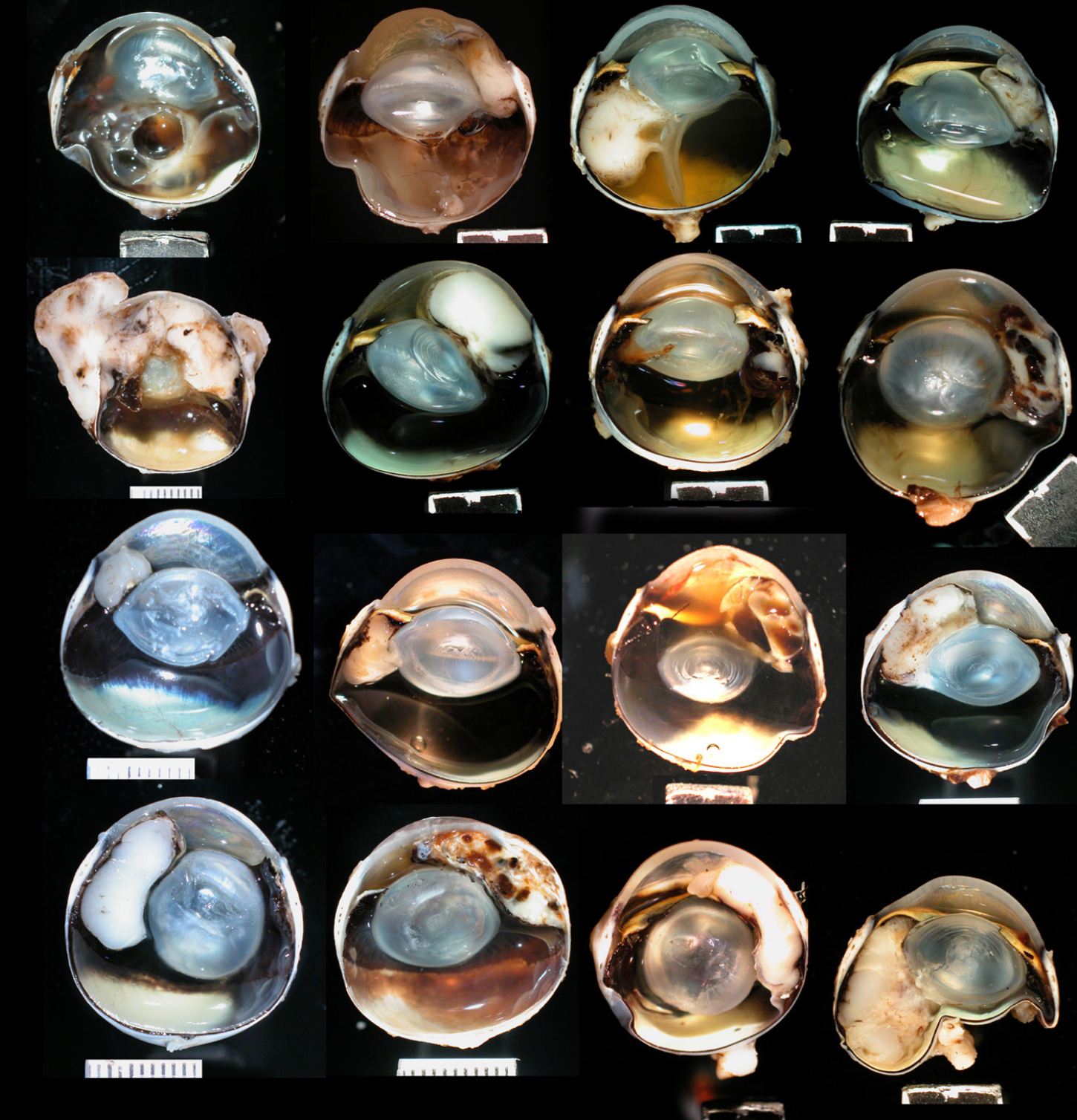
characteristics of iridociliary epithelial tumors (origin, species, biologic behavior)
cell of origin: pigmented or nonpigmented ciliary body epithelium
common in dogs, less common in cats
biologic behavior
vast majority benign = iridociliary adenomas
scleral invasion: iridociliary adenocarcinoma
gross appearance of iridociliary epithelial tumors
arises in posterior chamber
may extend through the pupil or invade the iris
often well vascularized
typically cream-colored mass; may be partial or completely pigmented
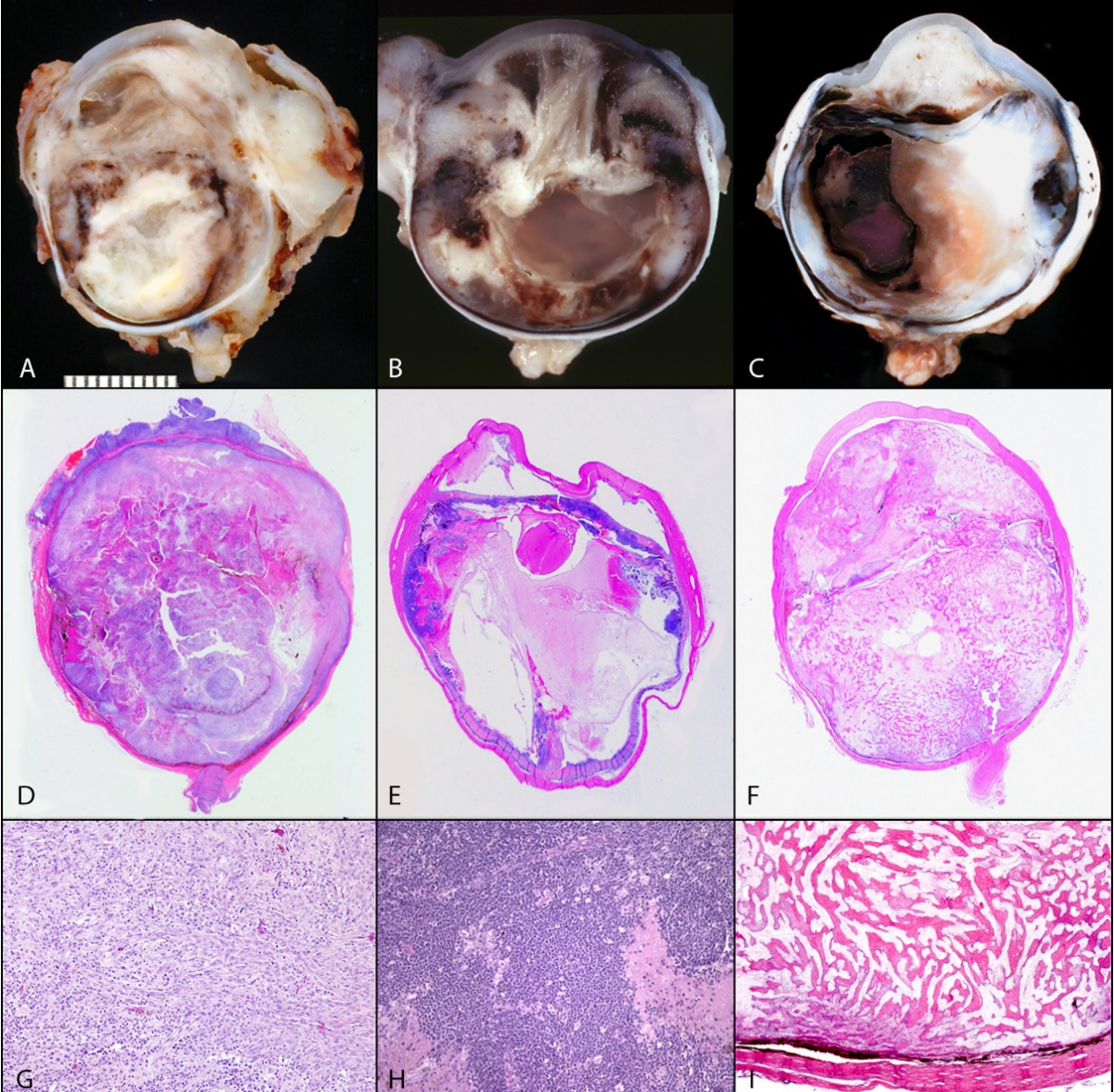
origin/associated risk factors of feline post-traumatic sarcoma
associated with previous ocular trauma & lens capsule rupture
development of tumor may occur months to years following the traumatic event
cell of origin: lens epithelial cells, released following lens capsule rupture
distribution of feline post-traumatic sarcoma
lines chambers of the eye, fills globe, extends through sclera, and can invade the optic nerve into the brain
may reoccur in orbit after enucleation
distant metastasis possible
which species are most commonly affected by squamous cell carcinoma of eyelids and conjunctiva?
cattle > horses > cats > dogs
how does squamous cell carcinoma develop? what are associated risk factors?
develops through pre-cancerous stage (plaques > papilloma) before malignant transformation over months or years
can be associated with viral infections: papilloma, herpes
associated with UV light exposure
tumors that commonly metastasize to the eye
lymphoma
histiocytic sarcoma
melanoma
hemangiosarcoma
mammary adenocarcinoma
other carcinomas
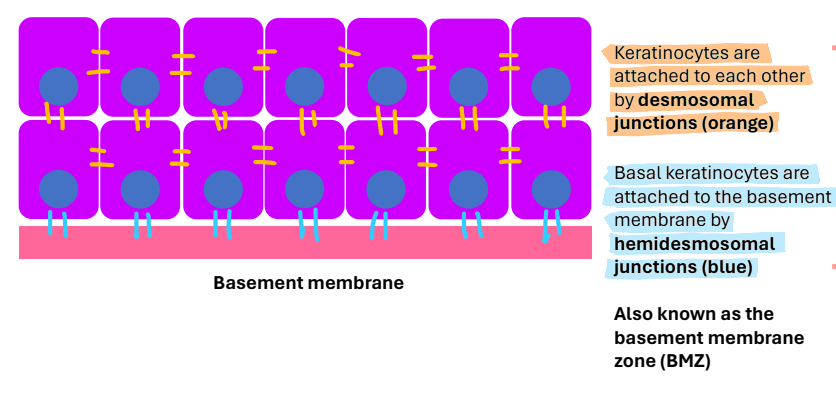
how are keratinocytes attached to each other?
desmosomal junctions
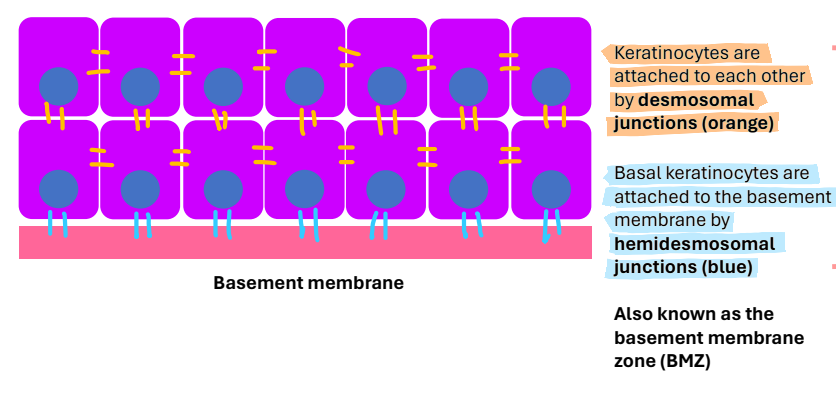
how are basal keratinocytes attached to the basement membrane?
hemidesmosomal junctions
what are the phases of the hair follicle cycle?
anagen = growth phase; longest (years)
most hairs are in anagen
catagen = transitional or involuting phase; short
telogen = resting phase
→ kenogen
→ anagen
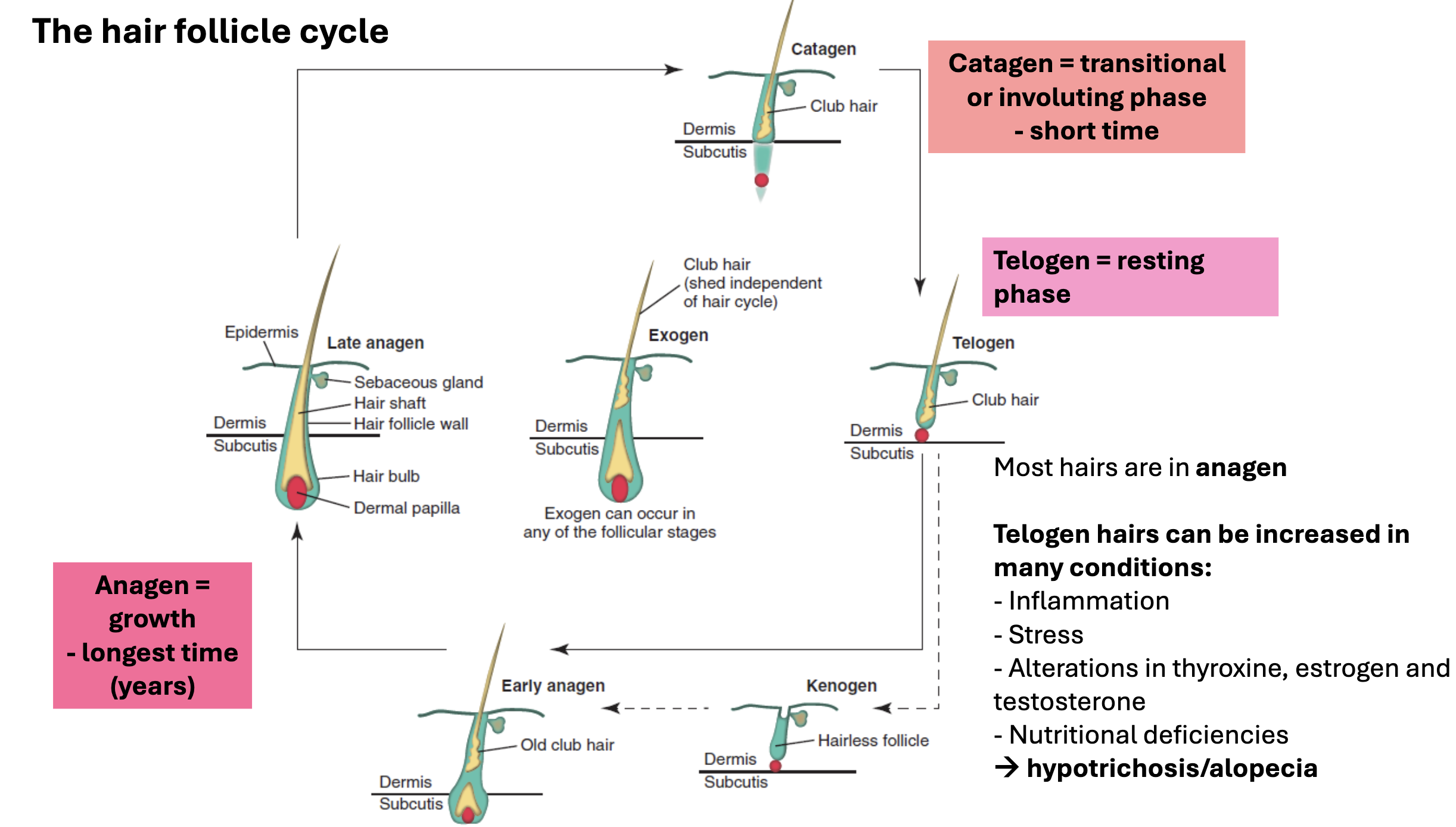
what can cause telogen hairs to be increased?
conditions such as:
inflammation
stress
alterations in thyroxine, estrogen, and testosterone
nutritional deficiencies
→ hypotrichosis (thinning hair) / alopecia (no hair)
what is the function of sebaceous glands?
produce sebum that coats the hair:
maintains normal hydration
acts as chemical barrier
gives coat a glossy sheen
acts as a pheromone
what are the different type of apocrine glands?
epitrichial — secrete to the primary hair follicles; sweat mixes with sebum to form the protective coating
thermoregulation in cattle/horses
eccrine — secrete directly to skin (paw pads)
anal glands vs anal sac
anal glands: specialized apocrine glands that open directly onto the anal skin via a duct at the rectoanal junction
similar apocrine glands line the anal sacs
anal sacs: squamous lined cystic cavity containing odiferous secretions
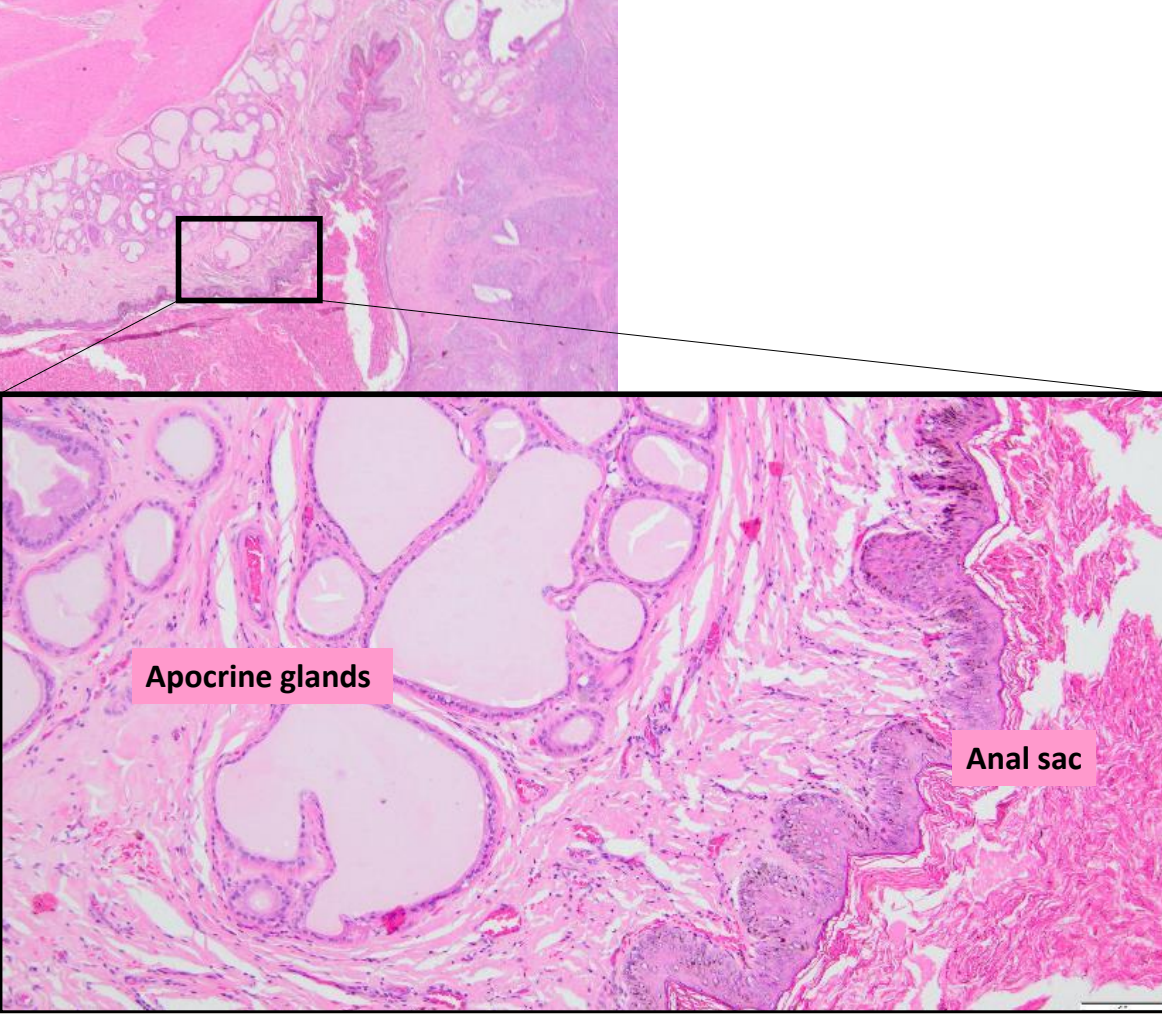
what are hepatoid glands? where are they located?
modified sebaceous glands around the anus, along the tail, prepuce, dorsolumbosacral areas
primary vs. secondary lesions
primary lesions occur directly
secondary lesions are often a result of self trauma (scratching or chewing)
papule
primary lesion
slightly raised, red, <1 cm
epidermal & dermal inflammation and edema
ex. insect bites
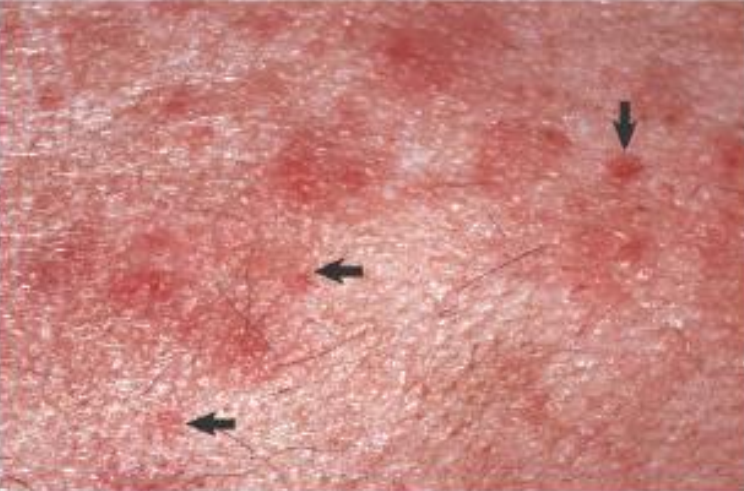
macule
primary lesion
flat, different color to adjacent skin
ex. hemorrhage, lentigo (increased pigment), vitiligo (loss of pigment)
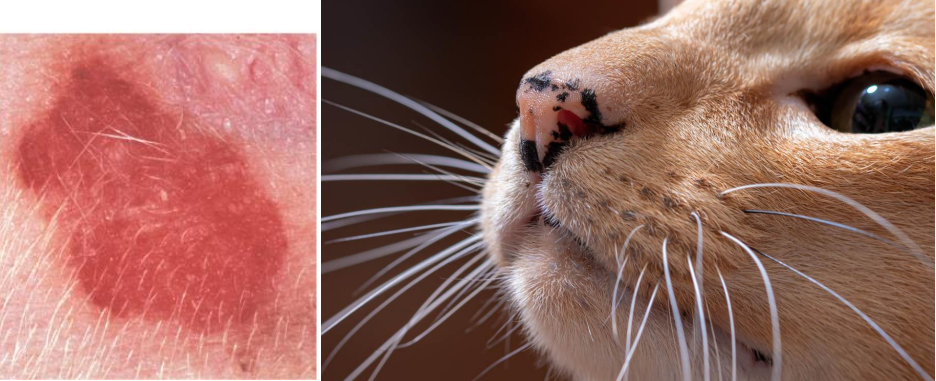
plaque
primary lesion
raised, flat-topped, semi-firm/firm
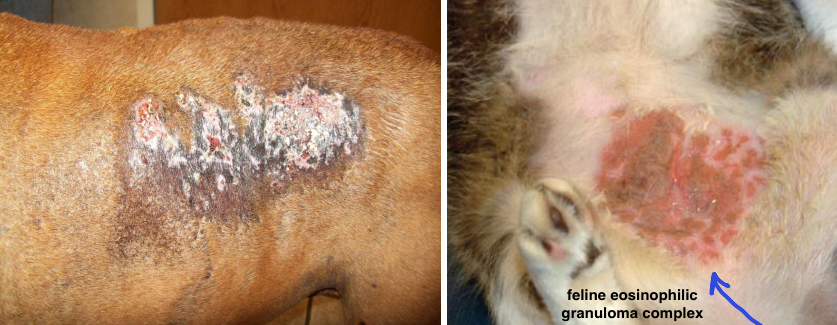
ex. calcium deposits (calcinosis cutis), thickened epidermis with inflammation - e.g. feline eosinophilic granuloma complex
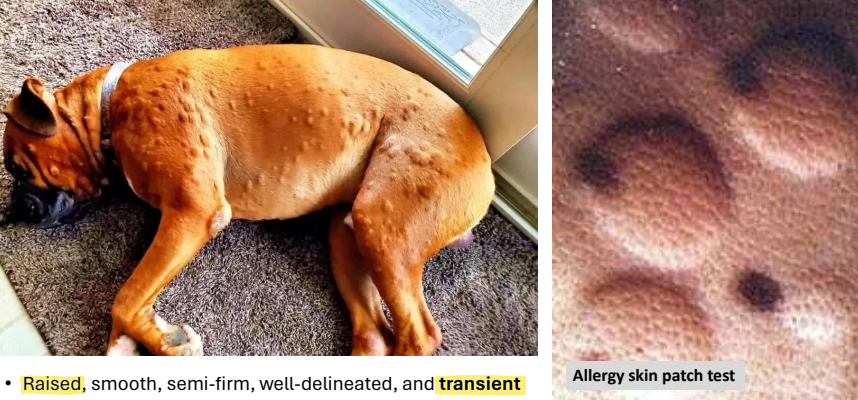
wheals (hives)
primary lesion
raised, smooth, semi-firm, well-delineated, and transient
edema → insect bites or allergy
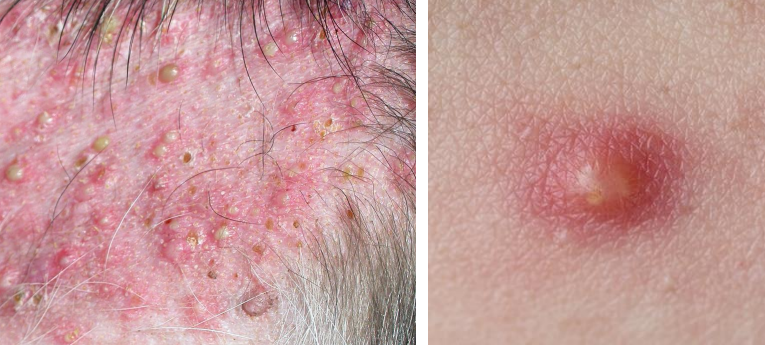
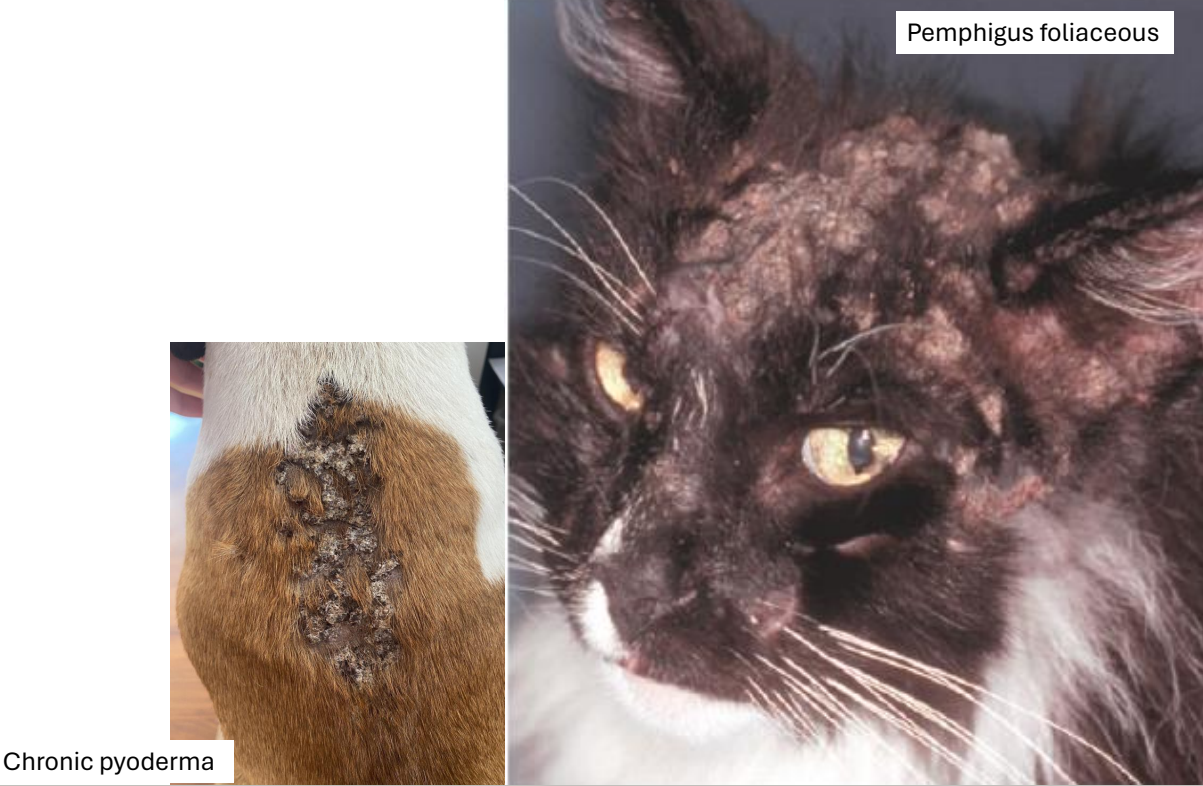
pustule
primary lesion
raised, smooth, soft, pale yellow to green (pus), red periphery
ex. bacterial infection (very common), pemphigus foliaceus (rare)
vesicle(s) & bulla(e)
primary lesion
vesicle <1 cm
bulla >1cm
fluid-filled pocket (blisters)
ex. burns, viral / immune-mediated / bacterial diseases
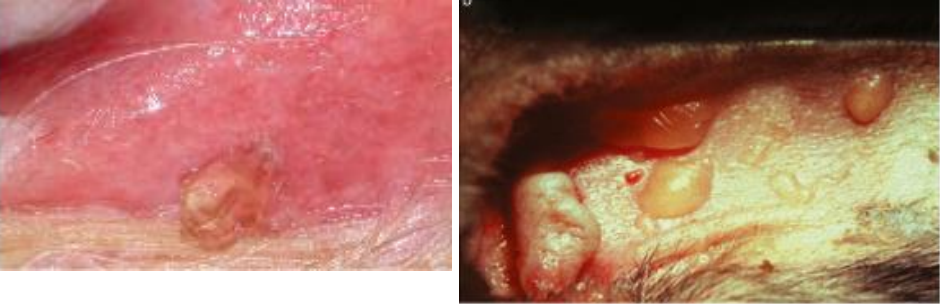
scale
secondary lesion
fragments of keratin on the surface; seen in many diseases
↑↑ stratum corneum
collarette
secondary lesion
flat to minimally elevated ring of scale that enlarges peripherally
remnant of an old pustule
ex. bacterial & fungal infections
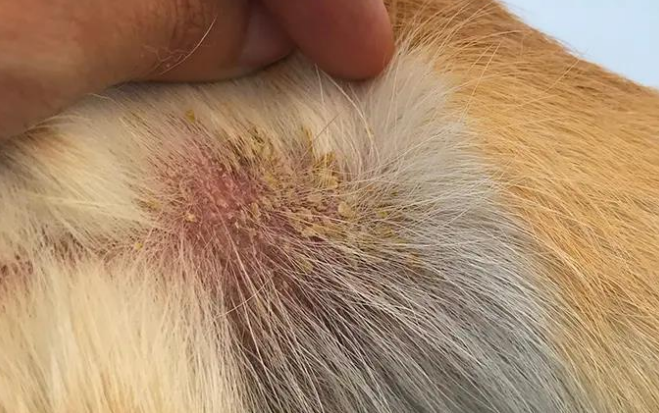
crust
secondary lesion
desiccated keratin with serum, blood, cellular debris, and inflammatory cells
seen in many diseases
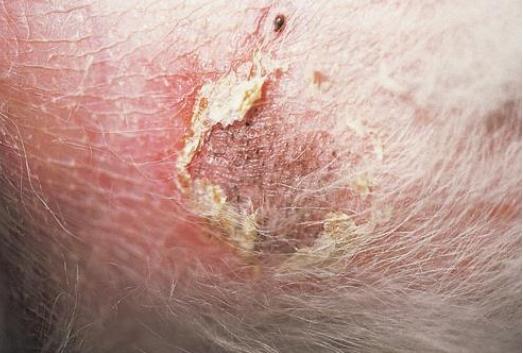
excoriation
secondary lesion
superficial loss of epidermal layers
secondary to physical trauma (scratching)
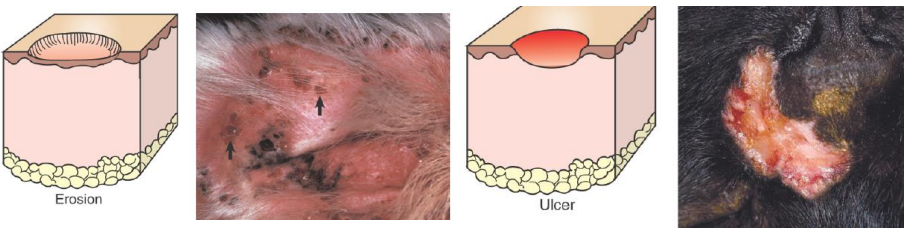
erosions & ulcers
secondary lesions
clinically look similar
erosion = partial thickness epidermal defect
ulcer = full thickness epidermal defect
general causes of hypo- and hyperpigmentation
secondary lesions
hyperpigmentation: chronic inflammatory diseases (common), some endocrine diseases (uncommon)
hypopigmentation: inflammation damaging pigment-containing epidermal cells or melanocytes
lichenification
secondary lesion
thickening of the skin, often with hyperpigmentation
accentuated creases
associated with chronic inflammation
rubbing, scratching, irritation
fissure
secondary lesion
linear crack
associated with chronic thickening of the skin
often seen on/near paw pads
comedo (blackheads)
secondary lesion
plugged follicle with keratin & sebum
ex. endocrine dermatoses (hyperadrenocorticism); pressure points
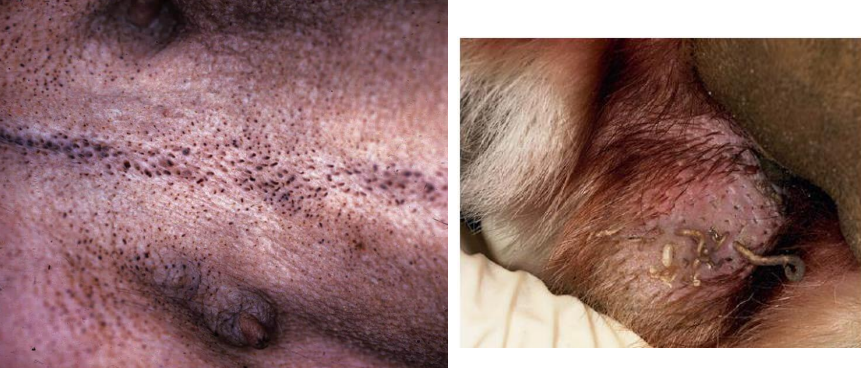
what is skin scraping best for?
mites — demodex spp.
what are acetate tape preparations/impression smears best for?
bacteria & fungi
what are fine needle aspirates best for?
nodules & tumors
when would a biopsy be appropriate to perform?
confirm a clinical diagnosis if the chosen treatment has significant side effects
a nodular lesion, ulcer, or non-healing wound (i.e., possible tumor)
lesions develop suddenly, are severe, or unusual
lesions develop during therapy (possible drug reaction)
multiple clinical differentials and routine exam & ancillary tests do not differentiate
skin disorder fails to respond to therapy or disorder recurs when therapy is stopped
how do you perform a skin biopsy?
collect multiple sites representative of the range of lesions
collect some samples at the junction of normal & abnormal
include crusts, if present
do not surgically prepare the site (including clipping) if lesion is confined to epidermis or superficial dermis
use largest punch biopsy possible (6-8mm) or elliptical full thickness biopsies
fix in 10% neutral buffered formalin (1:10 ratio of tissue to formalin)
consider saving a frozen section or submit for cultures
sample before therapy (or with appropriate withdrawal period)
clinical presentation of perivascular dermatitis
erythema & edema
most common → all inflammation starts perivascularly
least diagnostic
what is hyperkeratosis?
excess of surface layer (stratum corneum) of the epidermis
primary only in congenital ichthyosis
common secondary finding in many diseases
increased rate of keratinocyte proliferation OR delayed shedding of keratin
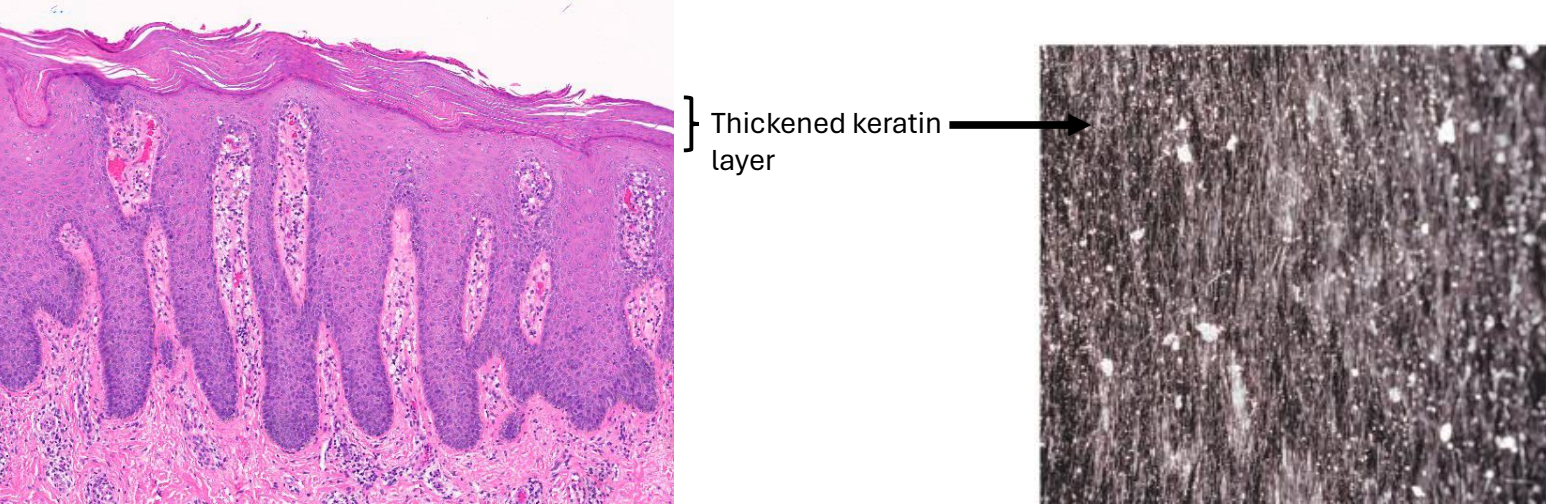
causes of hyperkeratosis (i.e., scale & crust)
focal: skin reaction to any inflammation (common)
bacteria, fungi, yeasts, ectoparasites
widespread: underlying metabolic disorders (uncommon/rare)
superficial necrolytic dermatitis
zinc-responsive dermatosis
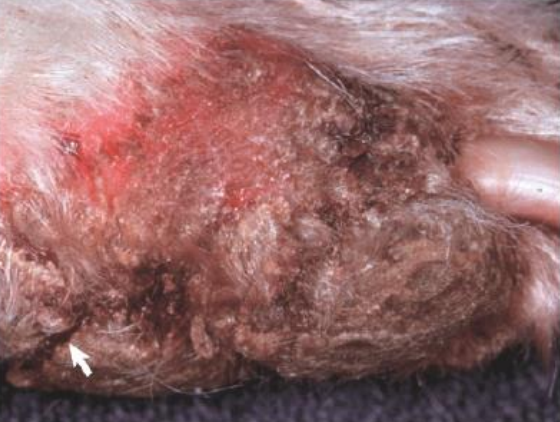
clinical presentation/distribution of dermatophilosis (“rain scald”)
crusts, scale, alopecia
distribution of lesions: dorsum + lower limbs
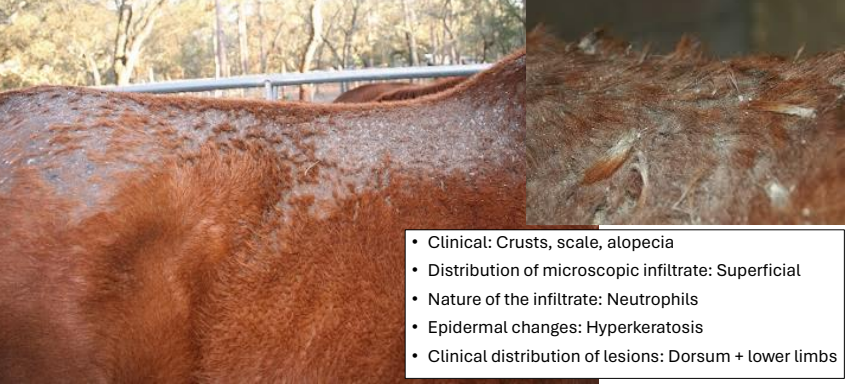
dermatophilosis infiltrate & epidermal changes
distribution of infiltrate: superficial
neutrophilic infiltrate (bacterial agent)
epidermal changes: hyperkeratosis
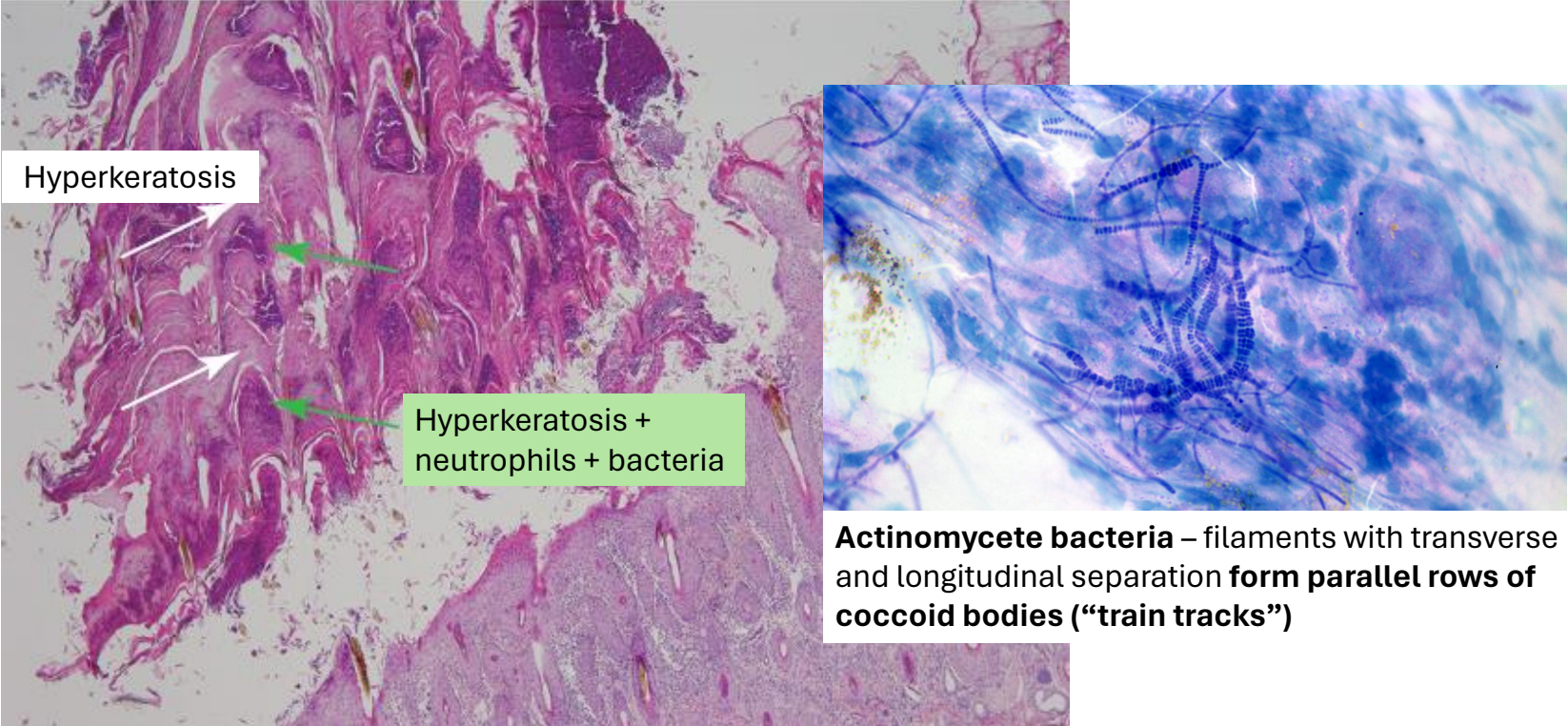
dermatophilosis pathogenesis
trauma to the skin & prolonged wetting → zoospores enter via breaks in the skin surface → invasion with neutrophils → epidermal regeneration → cycle starts again → thick laminar & parakeratotic crusts = characteristic
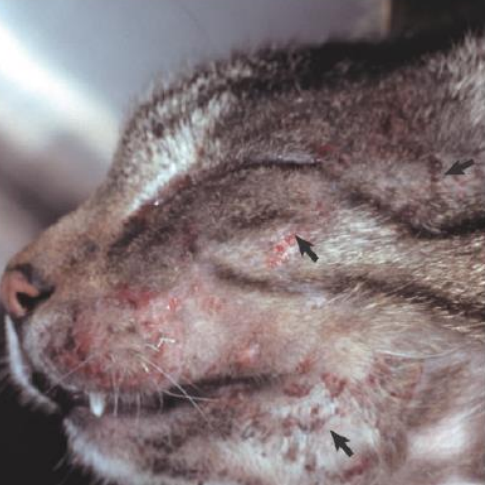
clinical presentation/distribution of dermatophytosis (“ringworm”)
crusts, scale, alopecia
distribution of lesions: face
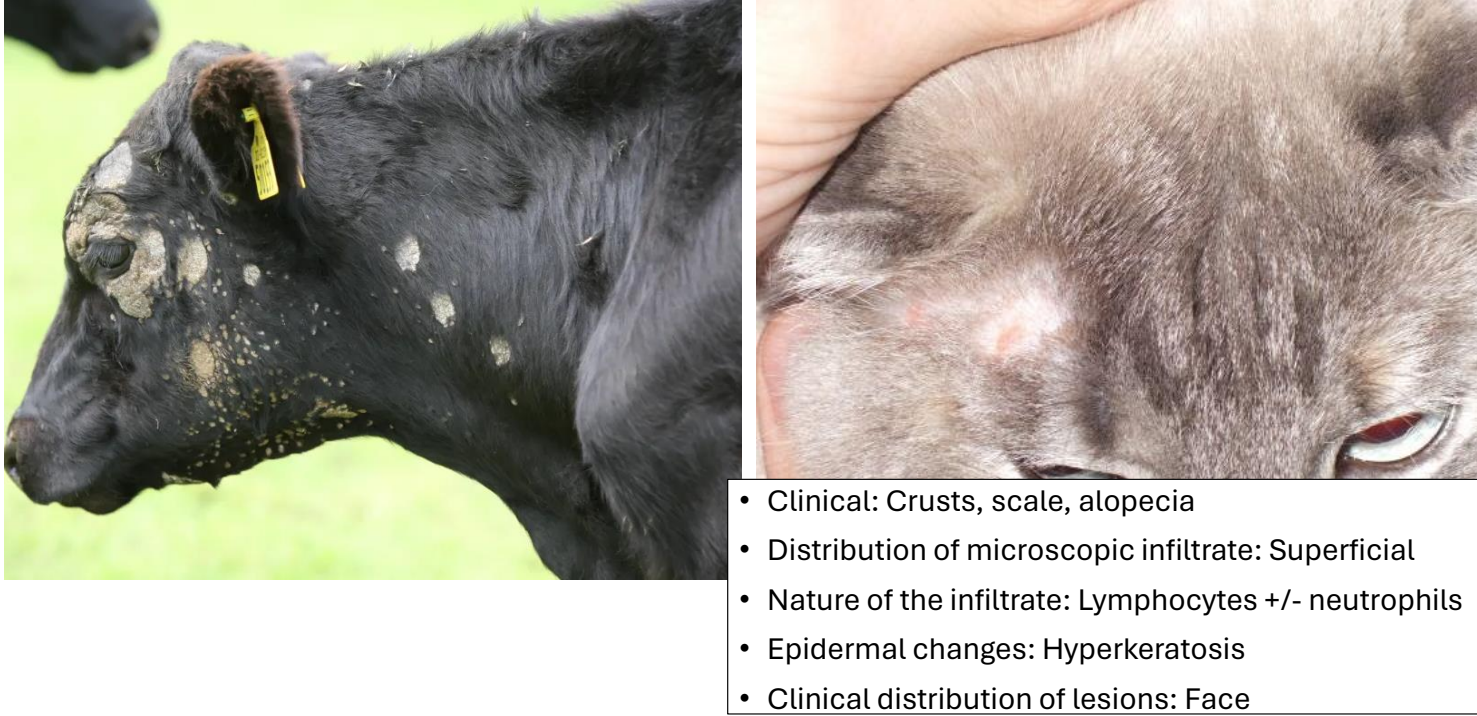
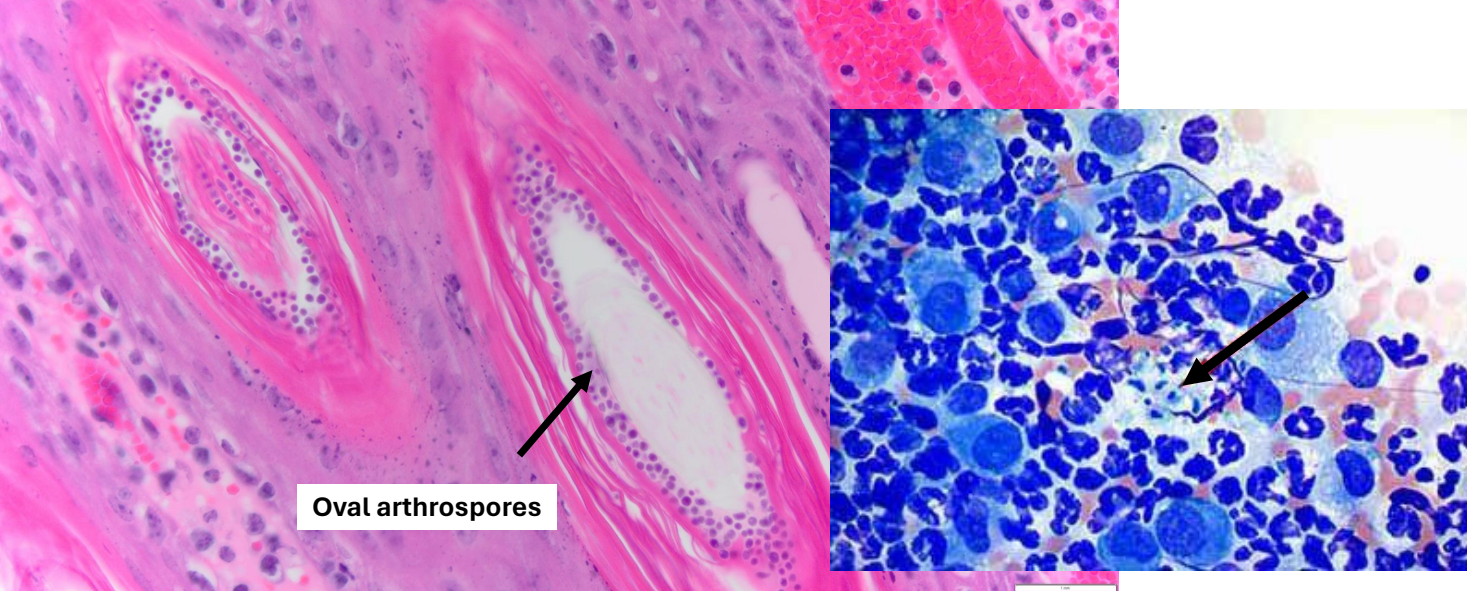
dermatophytosis infiltrate & epidermal changes
distribution of infiltrate: superficial
lymphocytic ± neutrophilic infiltrate (fungal agent)
epidermal changes: hyperkeratosis
dermatophytosis pathogenesis
breaks in stratum corneum facilitate invasion by the fungus → adhere to keratinocytes & migrate to follicular orifice
dermatophytes produce keratinolytic enzymes that hydrolyze keratin and enable them to invade the hair shaft → invade to the keratogenous zone of the hair bulb
stop here as cannot survive in viable tissues
as hair enters telogen, hair is sloughed and fungal growth stops
overall, self-limiting disease
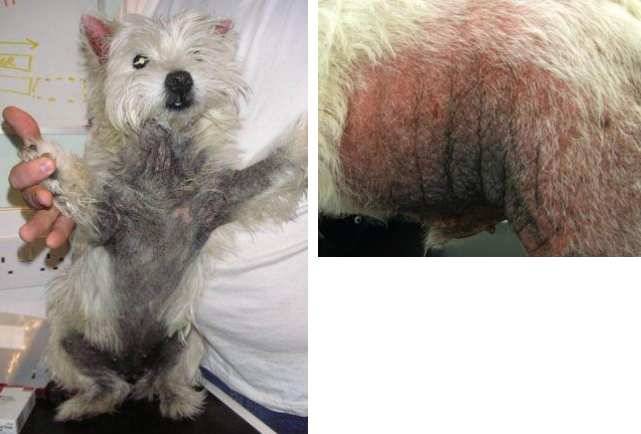
which signalments are affected by zinc-responsive dermatosis?
syndrome 1: siberian huskies & alaskan malamutes (+ other large breeds) — inherited
syndrome 2: puppies of any breed with a relative deficiency in Zn — probably secondary to excessively high calcium and/or phytates in diet (compete with zinc)
clinical presentation/distribution of zinc-responsive dermatosis
crusts, scale, alopecia
distribution of lesions: face
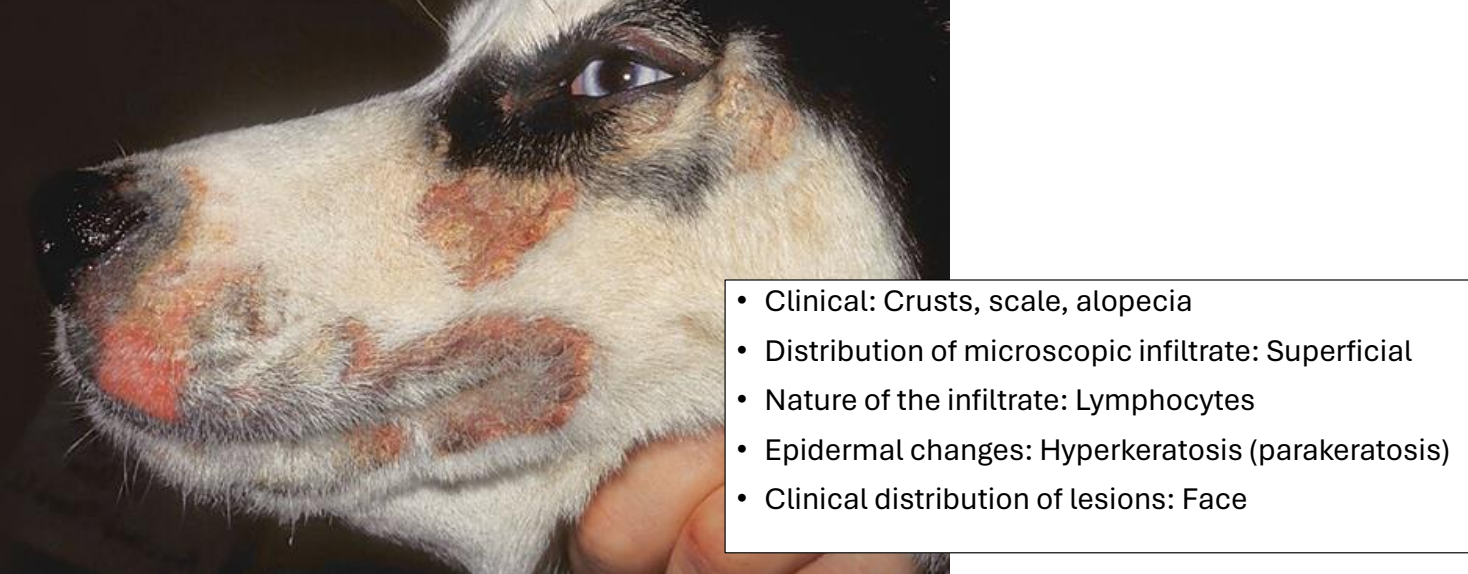
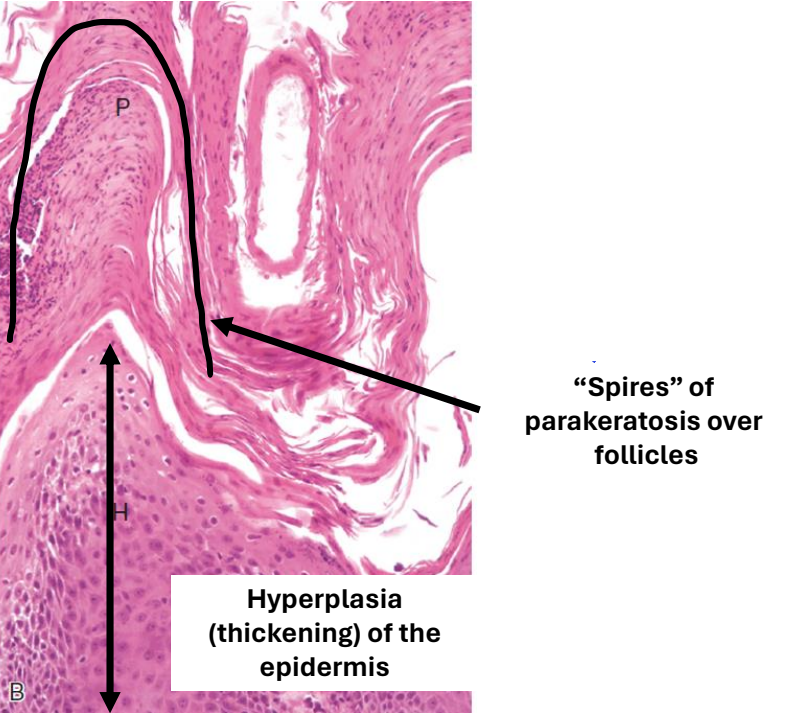
zinc-responsive dermatosis infiltrate & epidermal changes
distribution of infiltrate: superficial
lymphocytic infiltrate
epidermal changes: hyperkeratosis (parakeratosis)
clinical presentation/distribution of superficial necrolytic dermatitis
crusts & ulcers
distribution of lesions: face, feet ± mucocutaneous junctions
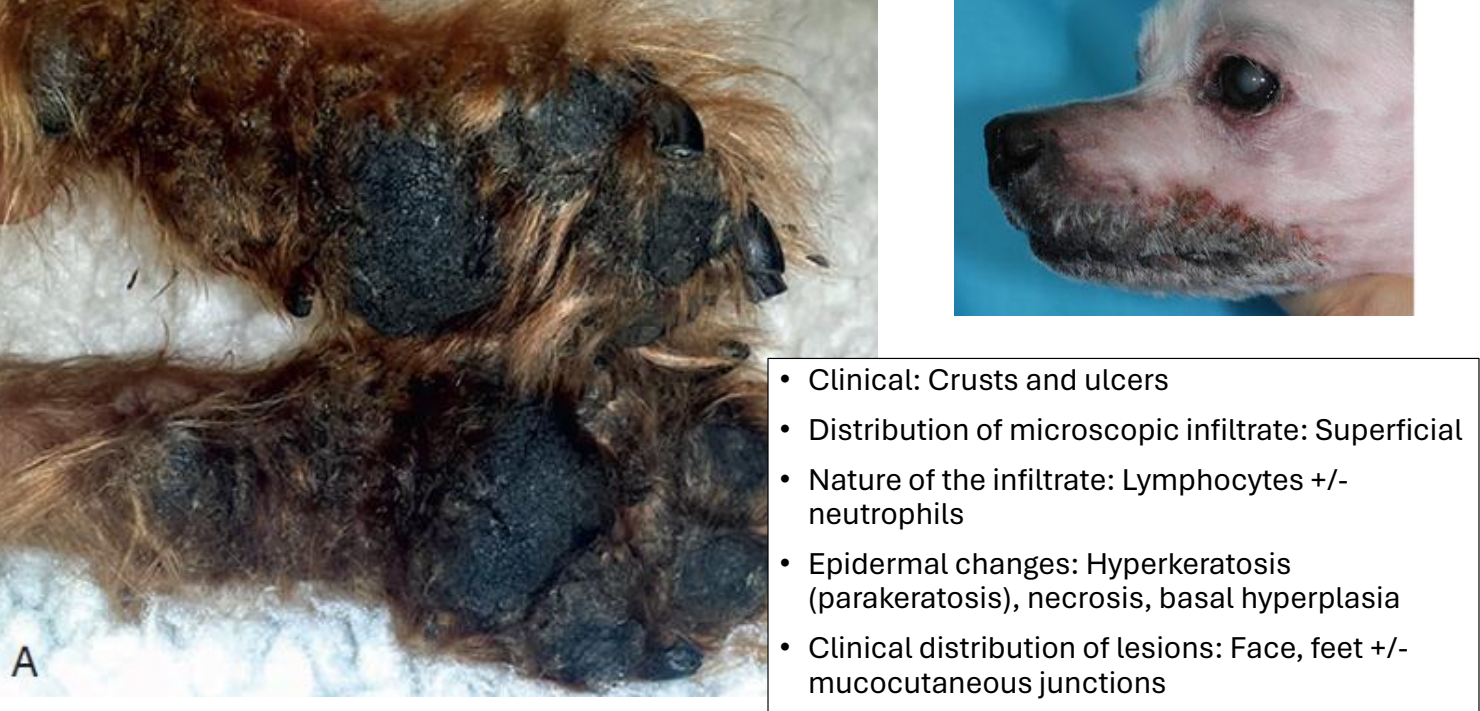
superficial necrolytic dermatitis infiltrate & epidermal changes
distribution of infiltrate: superficial
lymphocytic ± neutrophilic infiltrate
epidermal changes: hyperkeratosis (parakeratosis), necrosis, basal hyperplasia
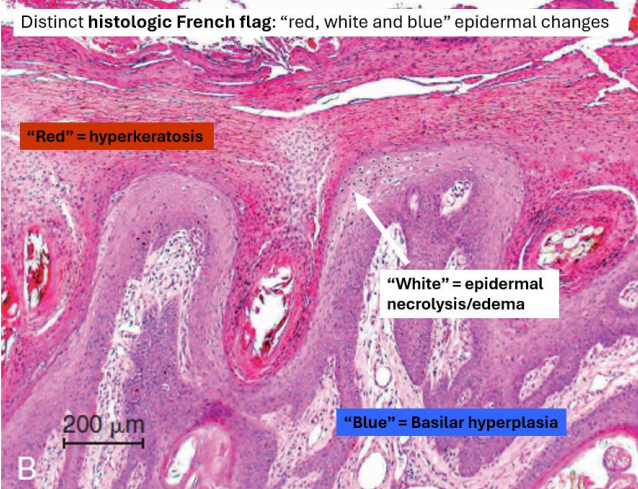
superficial necrolytic dermatitis pathogenesis
related to hepatic dysfunction & derangement of glucose and amino acid metabolism
↑ glucagon
↓ amino acids → ↓ epidermal proteins → epidermal necrosis
abnormal zinc & fatty acid metabolism
dogs: associated with glucagon-secreting pancreatic tumors, hyperglucagonemia, diabetes mellitus, & liver disease
rare manifestation of liver disease