chem231 - ch.5 - stereochemistry
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
constitutional isomers
same molecular formula, different atom connectivity
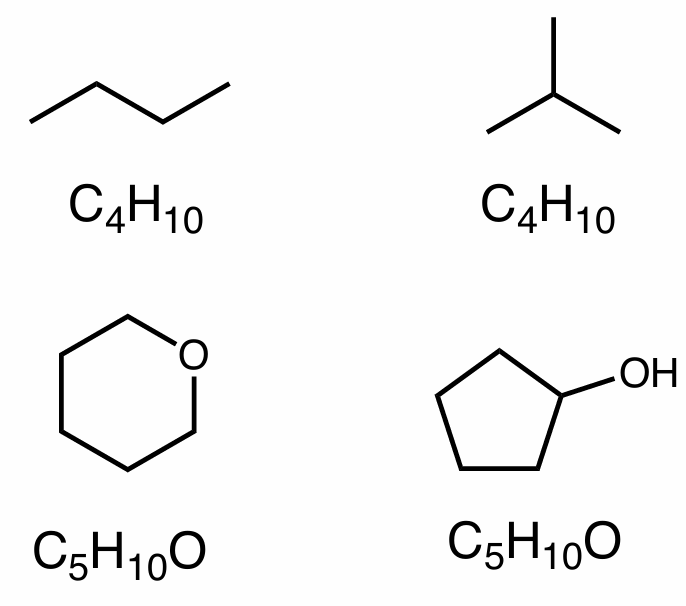
isomer
different compounds of the same molecular formula
stereoisomers
same molecular formula, different orientation in space
enantiomers
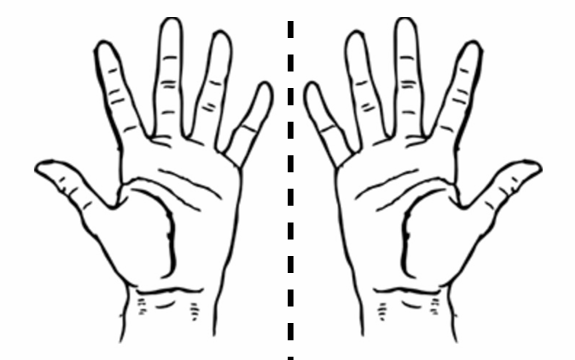
non-superimposable mirror images (mirror images)
exist in pairs
is chiral
ex. hands
can’t move which C it’s on
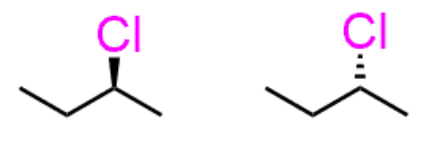
chiral
a chiral object (molecule) is chiral if it has an enantiomer
has:
chiral center
no plane of symmetry
axial chirality, planar chirality, inherent chirality
achiral
not chiral
identical, superimposable
no chiral center
chirality
the phenomenon where objects are non-superimposable mirror images
diastereomers
stereoisomers that are not enantiomers
may/may not be chiral
not mirror images
stereocenter
a point where switching 2 groups would lead to a different molecule
many diff types, we focus on chiral centers
chiral center
an atom (usually C) bearing four different groups
chiral centers are stereocenters
sp2 carbons NEVER chiral centers
if any 2 substituents have switched → opposite configurations
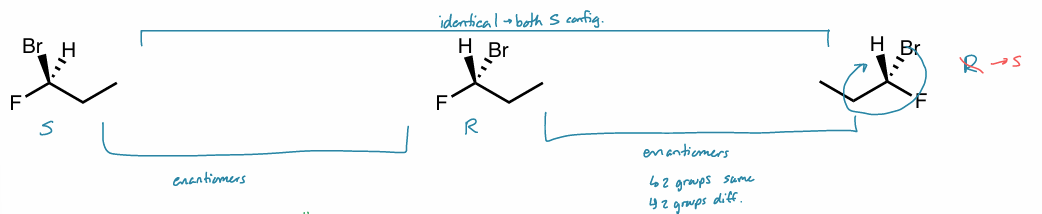
enantiomer methods
same molecule, switched dash & wedge
mirror images
R/S configuration
when a bond is dash/wedge, assume hydrogen (if there) is the opposite wedge/dash
enantiomers: method 1
molecules are exact same, but all wedges & dashes switched

enantiomers: method 2
molecule are mirror images
not flipped
if flipped, wedge → dash, vice versa
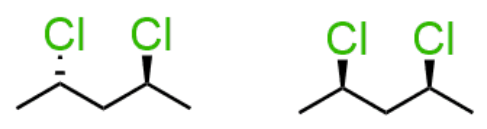
enantiomers: method 3
all chiral centers have opposite R/S configuration
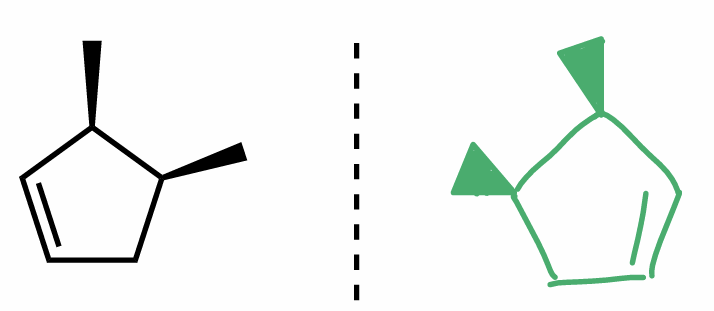
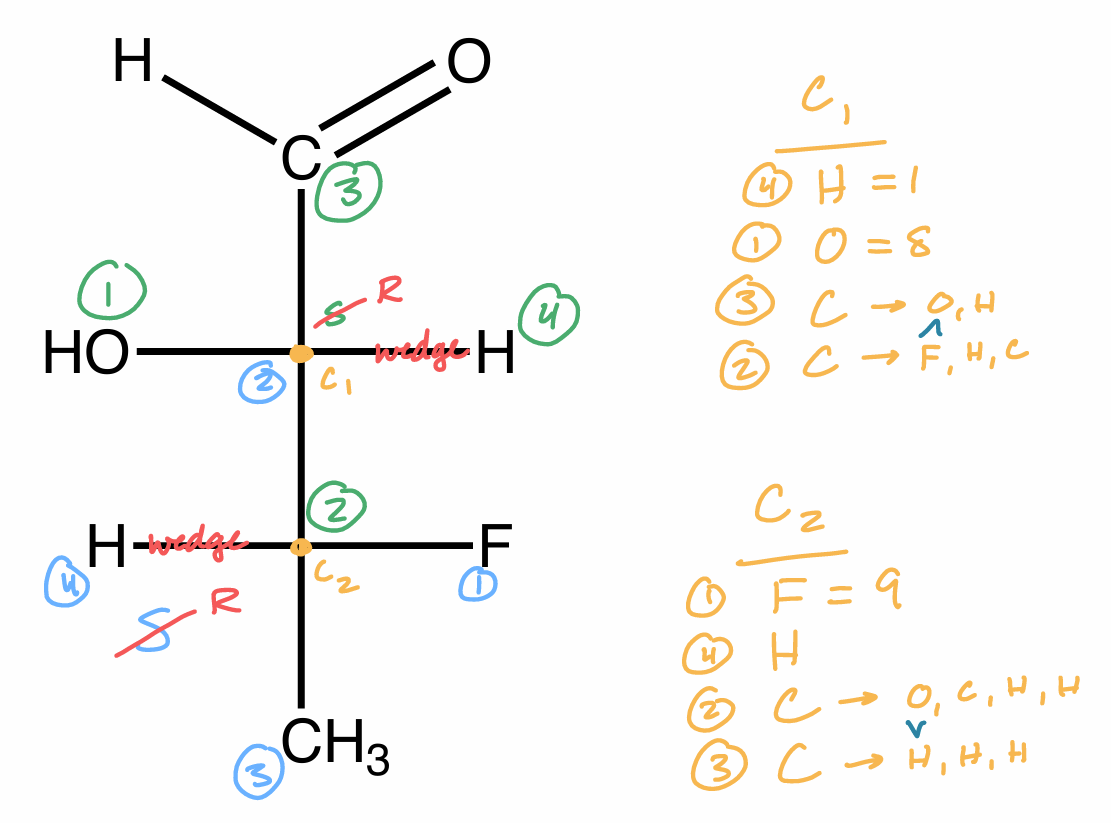
R/S steps
assign Cahn-Ingold-Prelog priorities of all atoms directly bonded to chiral center according to atom #
1 high → 4 low
lowest priority substituent (H) in back & connect 1 → 2 → 3
if in front, do it normal but switch R & S
if in plane, consider 3D orientation
label chiral center R or S
R = clockwise
S - counterclockwise
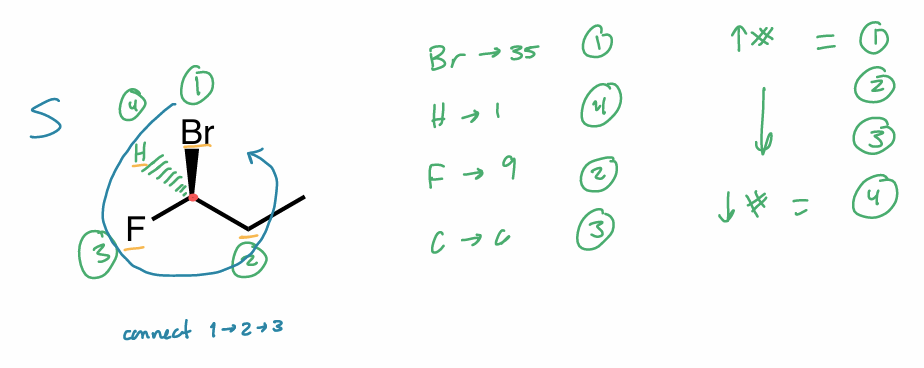
R/S 4th sub in plane
consider 3D orientation
look down bond from C w/ H behind it (NP)
rotate bond
“switch” method
R/S: method 1
look down bond from C w/ H directly behind chiral center
like Newman projection
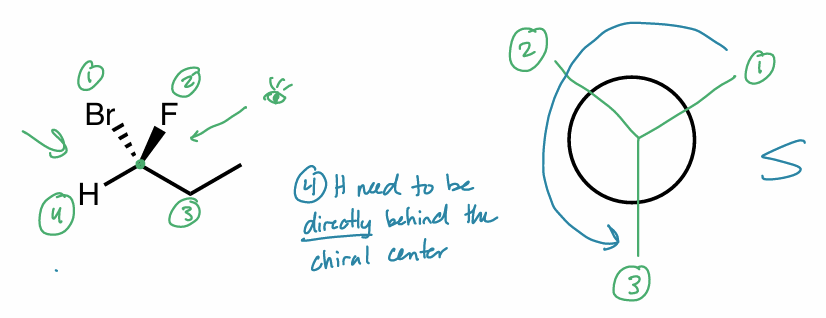
R/S: method 2
rotate bond so 4th priority subst. (H) isn’t in plane anymore
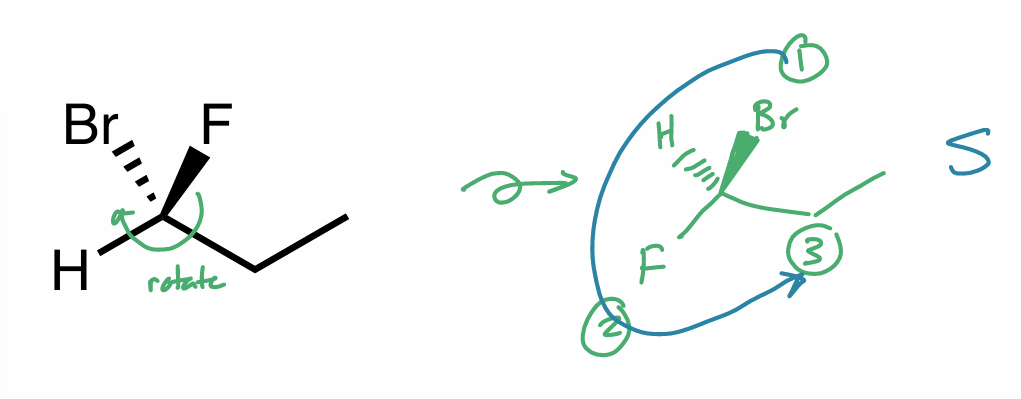
R/S: method 3
“switch” method
assign priorities
switch 2 substituents & switch other 2 substituents
any 2 groups
want H in back (dash)
assign R/S as usual
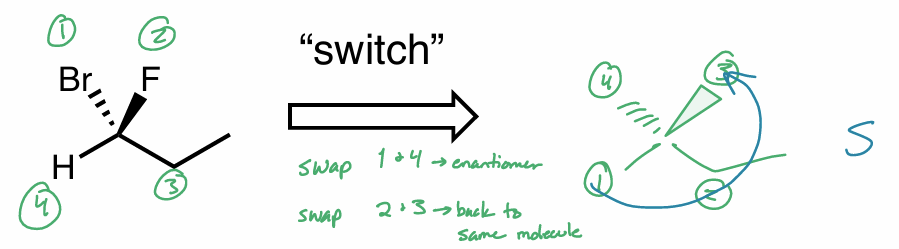
racemic mixture
when there are no wedges or dashes drawn, molecule represents racemic mixture (50:50) of enantiomers
half R
half S

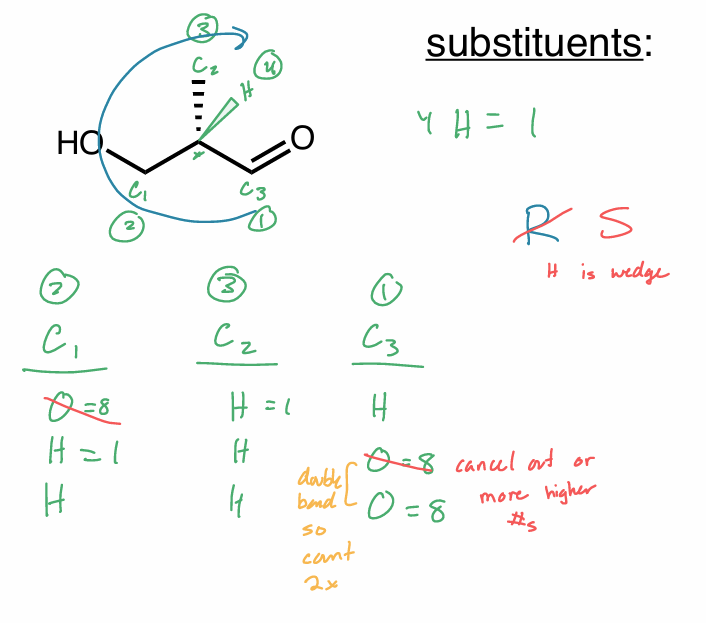
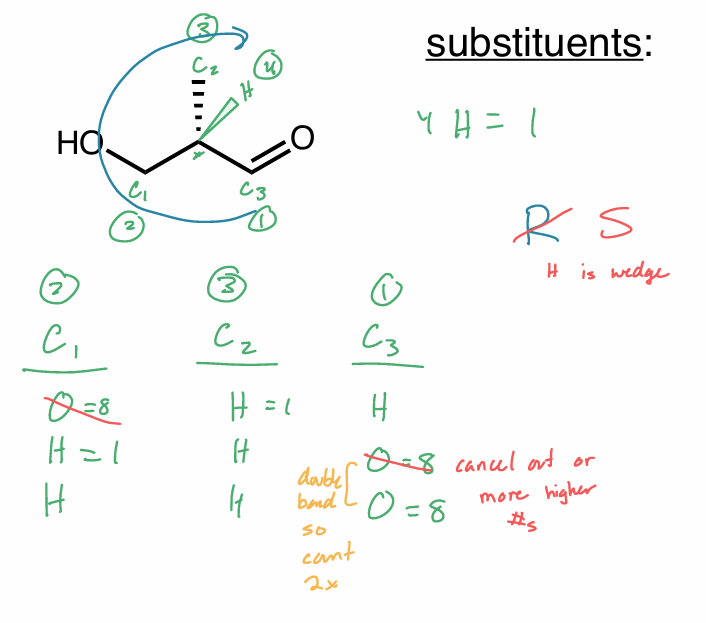
R/S 2 same atom
continue along the chain until you get a point of difference
double bonds count twice
triple bonds count thrice
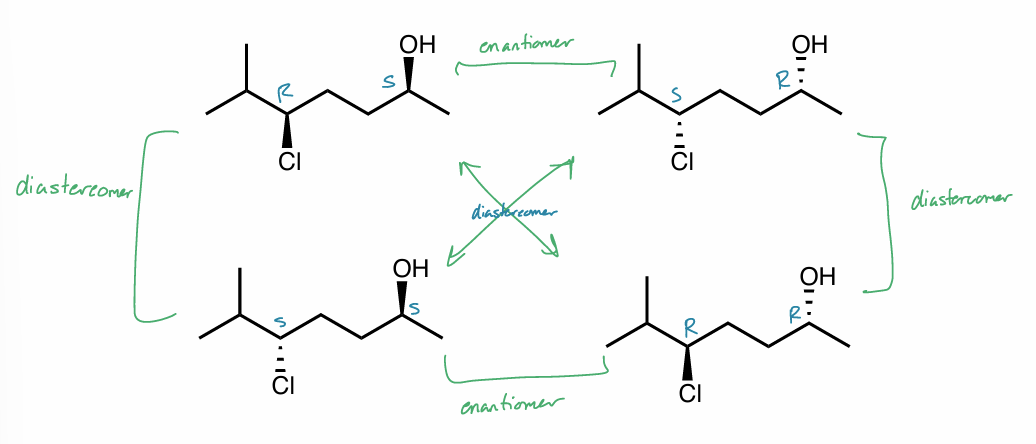
diastereomers
a stereoisomer that is not an enantiomer
must have multiple chiral centers OR
an alkene

diastereomers: multiple chiral centers
identical → same configurations
enantiomer → opposite at chiral centers (dash/wedge)
diastereomer → at least 1, but not all chiral centers opposite (dash/wedge)
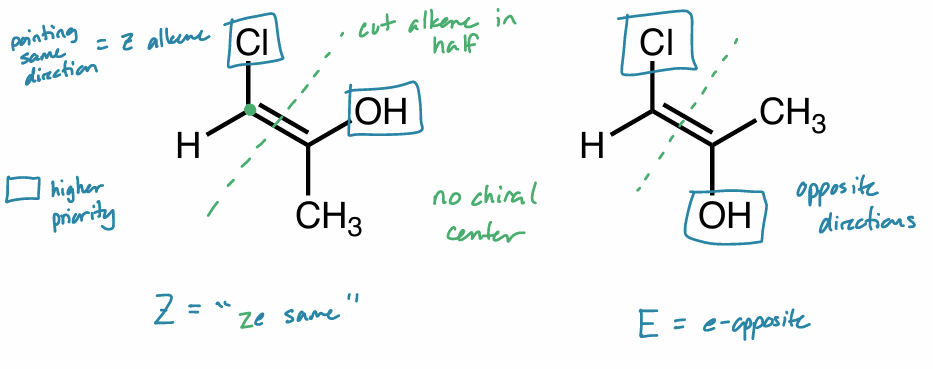
diastereomers: alkene
designate as E, Z or neither
Z → same “ze same”
E → opposite “e-opposite”

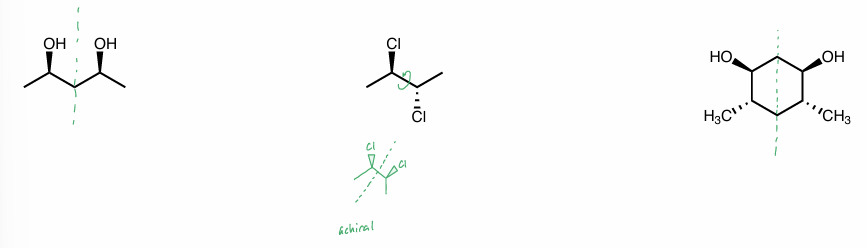
meso compound
has a superimposable mirror image & are achiral
has
chiral center (but not chiral)
plane of symmetry
2 chiral centers, but molecule itself is not chiral b/c internal plane of symmetry
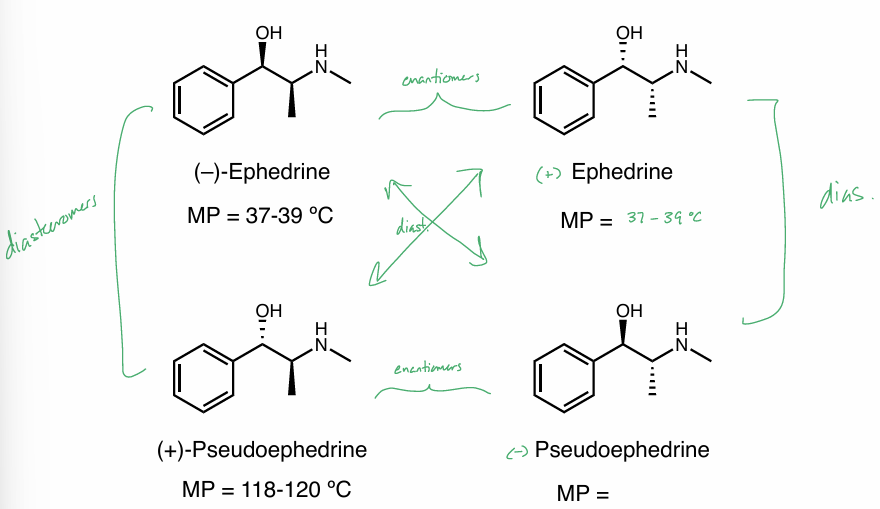
enantiomer properties
identical physical & chemical properties except they rotate plane polarized light in opposite directions (optical rotation)
behave differently in chiral environments (enzymes)
diastereomer properties
different physical properties (similar / drastically different)
utilize these diff physical properties to separate diastereomers
boiling point
melting point
solubility
etc
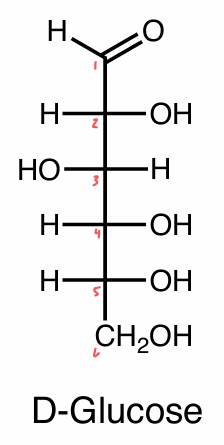
Fischer projections
Emil Fischer (1902)
a way to look at carbohydrates/sugars in the highest E conformation
skeletal → lowest E conformation
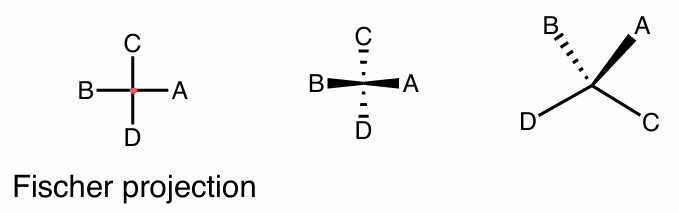
reading FP
| horizontal → wedge
— vertical → dash
converting FP
turn counterclockwise
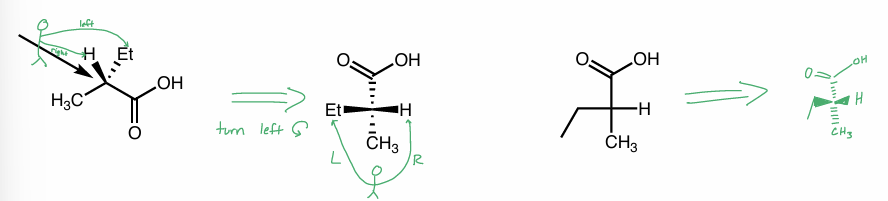
converting FP to skeletal
to skeletal
vertical axis → plane (solid line)
1 side is dashed, the other is wedged
“tip” on its side
want carbons in zig zag formation
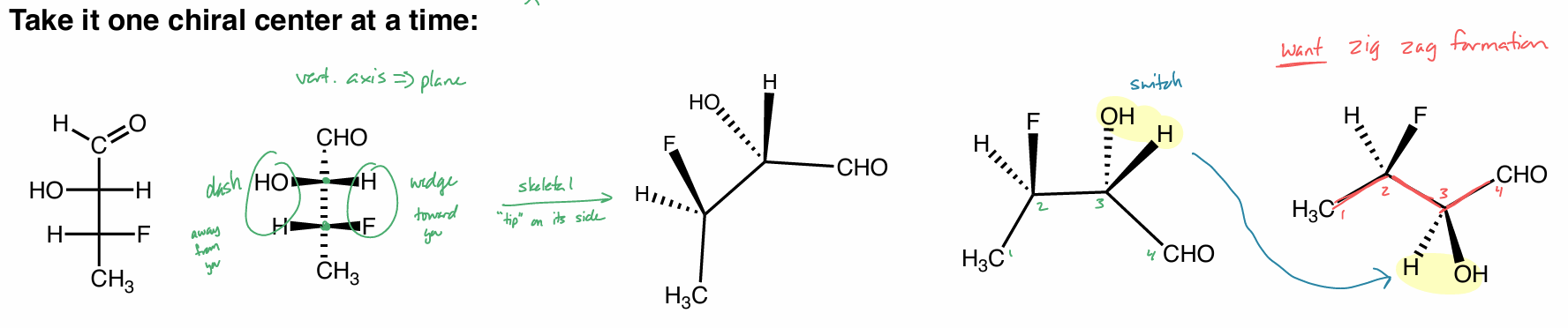
converting NP to FP
make everything eclipsed w/ FP
push from/back carbon to top/bottom so sides point towards you
point ↑ → push ↑
point ↓ → push ↓
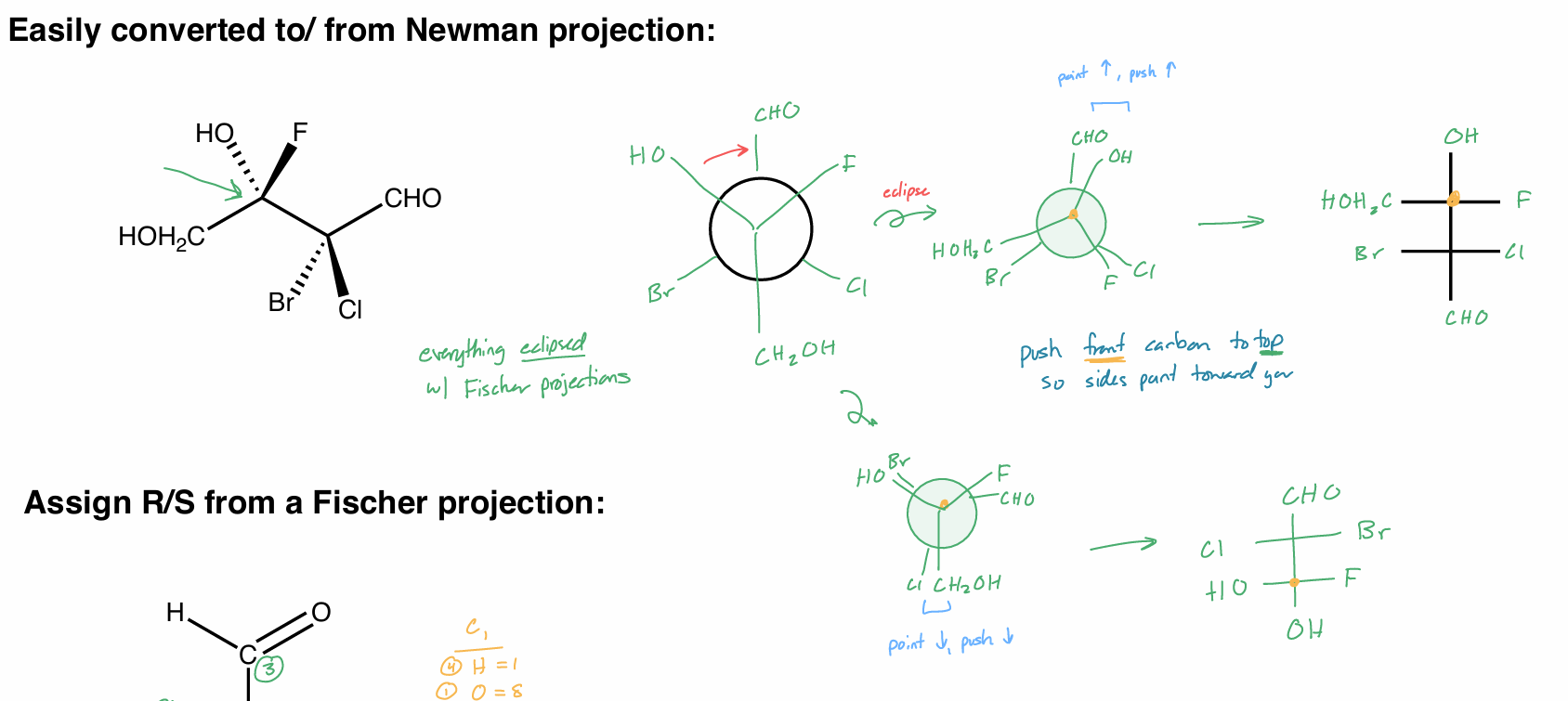
FP R/S
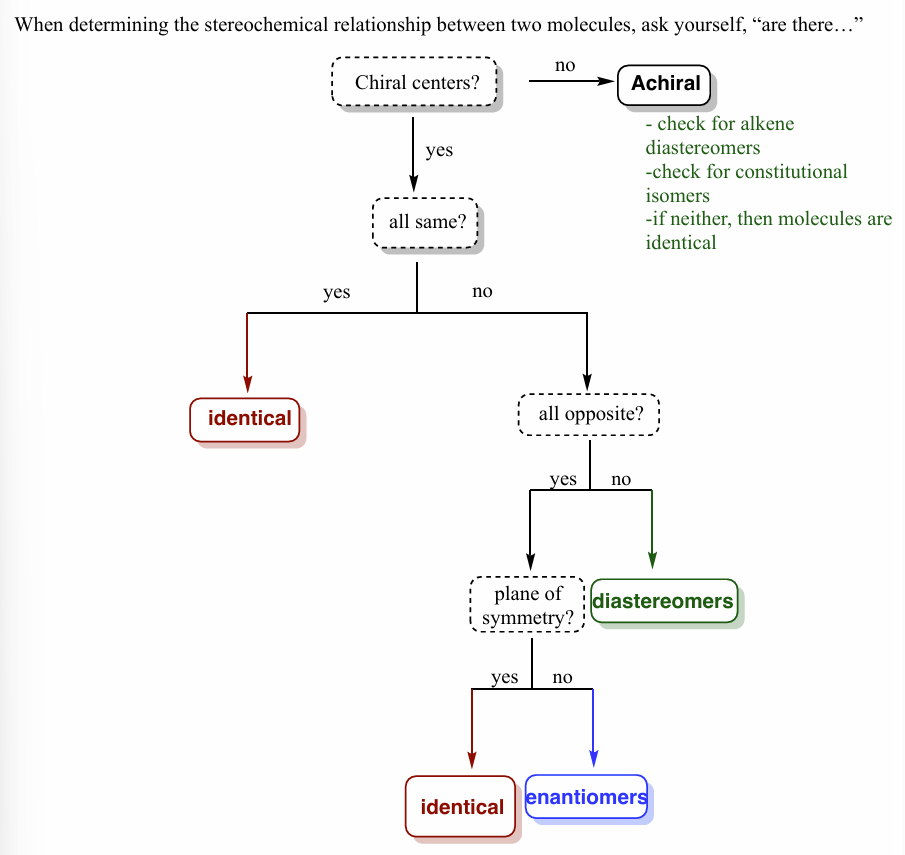
stereochemistry flow chart