Differential Extraction
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
Define differential extraction
A method used to separate compounds based on their different solubilities in two immiscible liquids, usually water and an organic solvent.
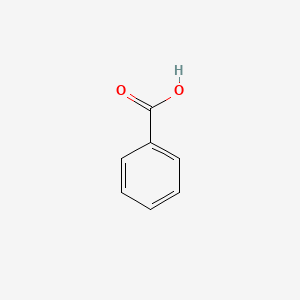
Structures for benzoic acid
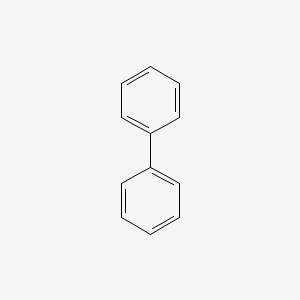
Structures for biphenyl
Know what compound will be in the aqueous layer and ether layer
Benzoic acid goes into the aqueous layer after it reacts with base and becomes water soluble.
Biphenyl stays in the ether layer because it is nonpolar
Know which solvent is best for polar compounds and used for wide range of compounds
Water is best for polar compounds.
Ether is a common organic solvent used for many nonpolar compounds.
Purpose of acids like HCl in a reaction
HCl is used to protonate a compound, often to turn a water soluble salt back into its neutral form so it precipitates.
What is sodium sulfate used for in this experiment
It is a drying agent. It removes water from the organic layer.
Melting points, what happens when impurities are present
Impurities lower the melting point and make the melting range wider.
Define recrystallization
A purification method where a solid is dissolved in hot solvent and then slowly cooled so pure crystals form.
Why cool crystals to room temp before using an ice bath
This allows slow crystal formation first. If cooled too fast, impurities can get trapped in the crystals.