Lab 8: Nucleophilic Substitution of Alkyl Halides
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
In free-radical reactions, molecules break through the process of _____________?
Homolysis, which involves each atom in the bond getting an equal amount of electrons
In ionic reactions, molecules break through the process of _______________?
Heterolysis, in which one atom gets all of the electrons from the bond and the other gets none
What is the geometry of a carbocation?
Trigonal planar, with an empty p orbital
What is the relative order of stability of carbocations?
From most to least stable: tertiary, secondary, primary, and methyl
How do alkyl groups stabilize carbocations?
Help displace the resulting positive charge by displacing or releasing electrons
What is a nucleophile?
A species with an unshared pair of electrons that wants to attack a molecule so that it can share this pair of electrons
What are the two types of nucleophiles that can exist?
1. One with a full negative charge (such as a halide)
2. One that does not carry a full charge, but contains an unshared pair of electrons (such as water)
Not all leaving groups are equally good. What might be the reason for this?
Not all of them can easily stabilize a negative charge. For instance, fluorine is a very small molecule and does not have as much space to "spread out" its electron density
Iodine, on the other hand, would be a better leaving group because it is larger and can better stabilize a negative charge
The rate equation for an Sn2 reaction is dependent on what?
The concentration of the nucleophile and the concentration of the alkyl halide
How many steps are present in an Sn2 reaction?
One
What type of molecule would be most likely to undergo an Sn2 reaction?
Primary alkyl halide
What does Sn2 stand for?
Substitution nucleophilic bimolecular
When an Sn2 reaction takes place, what is the chirality of the resulting molecule?
Opposite chirality of the starting molecule
This is because the starting molecule does not form an intermediate but proceeds in one step, which means that the nucleophile can only attack the alkyl halide from one direction
What does the rate of an Sn1 reaction depend on?
The concentration of the alkyl halide only
What does Sn1 stand for?
Substitution nucleophilic unimolecular
In an Sn1 rection, what is th chirality of the product?
50:50 (racemic mixture) because the nucleophile can attack the carbocation from either side
The carbocation that is formed in the first step is planar, which allows the nucleophile to attack from either side
What type of molecule is most likely to undergo an Sn1 reaction?
Tertiary alkyl halide
What type of molecule could undergo either Sn1 or Sn2 reactions?
A secondary alkyl halide
What is the primary determinant of whether a molecule will undergo Sn1 or Sn2 reactions and what is the definition of this term?
Steric hindrance, which refers to the ability of a nucleophile to come within bonding distance of the alkyl halide
Bulky molecules are less likely to undergo Sn2 reactions because they are more sterically hindered
What effect do polar protic solvents have on the nucleophile in Sn2 reactions?
They can interact with the nucleophile and slow down the reaction between the nucleophile and the alkyl halide
What effect do polar aprotic solvents have on the nucleophile in Sn2 reactions?
The solvent does not interact with the nucleophile and the reaction can proceed faster
What effect do polar protic solvents have on Sn1 ractions?
They stabilize the transition state, thus lowering the activation energy and increases the reaction rate
What effect do polar aprotic solvents have on Sn1 reactions?
They do not stabilize the transition state and thus make the Sn1 reaction proceed slower
Which substitution mechanism, Sn1 or Sn2, do vinylic and aryl halides go through?
Neither, because both have resonance stabilization
Moreover, the aryl halides have a bulky benzene ring that causes an increase in steric hindrance
What is the effect of increasing the temperature of a reaction have on the rate of the reaction?
Increases the rate of the reaction
What would be the effect of carrying out the sodium iodide in acetone reaction with the alkyl halides using an iodide solution half as concentrated?
Since this solution favors an Sn2 reaction, both the concentration of the alkyl halide and the concentration of the nucleophile contribute to the rate. SN2 reactions are first order with respect to the nucleophile and first-order with respect to the alkyl halide.
Thus, halving the concentration of the sodium iodide solution would half the rate of the reaction.
The addition of sodium or potassium iodide catalyzes many SN2 reactions of alkyl chlorides or bromides. Explain.
Since Iodine is a strong nucleophile, it will rapidly replace chlorine or bromine to form an alkyl iodide.
Moreover, because iodine is also a good leaving group, it can be displaced from an alkyl halide fairly readily, causing the original iodine ion to be regenerated.
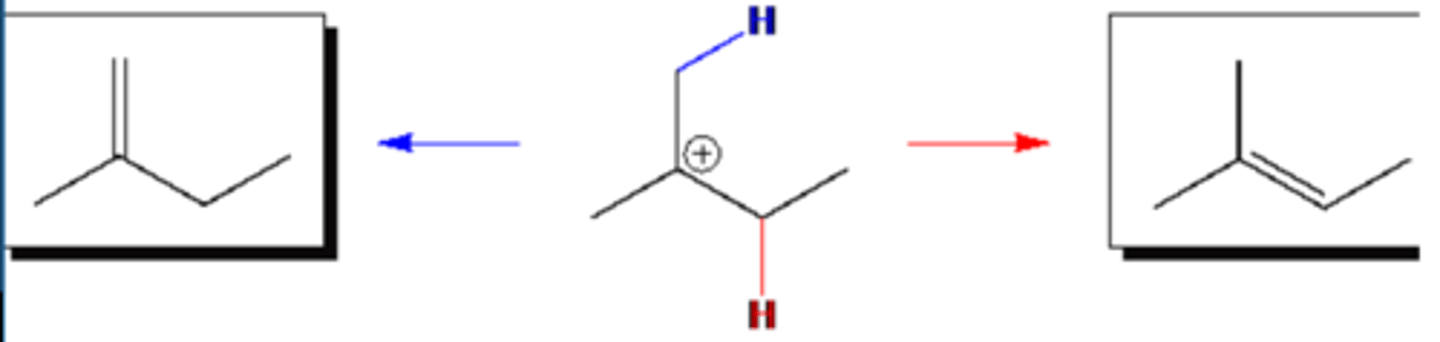
In SN1 reactions, the intermediate carbocations can eliminate a proton to yield alkenes or react with the solvent to yield ethers. Draw the structures of the by-products of this type that would be derived from the reaction of the carbocation derived from 2-bromo-2-methylbutane in ethanol.