AQA A level Chem 3.1 Introduction to Organic Chemistry
1/7
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Define the term stereoisomers. (2)
(Different molecules/compounds with the) same (molecular and) structural formula (1)
Different spatial arrangement of atoms (1)
Under suitable conditions, 2-bromobutane reacts with sodium hydroxide to
produce a mixture of five products, A, B, C, D and E.
Products A, B and C are alkenes.
A is a structural isomer of B and C.
A does not exhibit stereoisomerism.
B and C are a pair of stereoisomers.
Products D and E are alcohols.
D and E are a pair of enantiomers.
Deduce the name of isomer A. Explain why A does not exhibit stereoisomerism. (2)
A = but-1-ene (1)
two groups/atoms/Hs the same on one of the C=C carbons (1)
Under suitable conditions, 2-bromobutane reacts with sodium hydroxide to
produce a mixture of five products, A, B, C, D and E.
Products A, B and C are alkenes.
A is a structural isomer of B and C.
A does not exhibit stereoisomerism.
B and C are a pair of stereoisomers.
Products D and E are alcohols.
D and E are a pair of enantiomers.
Deduce the name of isomer B and the name of isomer C. Explain the origin of the stereoisomerism in B and C. (2)
Z-but-2-ene AND E-but-2-ene (1)
lack of/restricted/no (free) rotation around C=C/double bond (1)
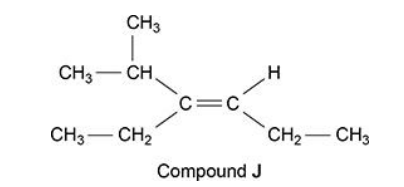
Compound J is one of a pair of stereoisomers
Explain:
• what stereoisomers are
• how E-Z stereoisomerism occurs
• how the Cahn-Ingold-Prelog rules can be used to decide whether compound J is an E or Z isomer. (6)
Stereoisomers are compounds with the same structural formula but a different arrangement of atoms in space.
E/Z isomerism arises due to restricted rotation around the C=C bond
When each C of C=C is attached to two different groups
CH3CH2 is higher priority than H on RHS C
as atomic number of C is higher than H or C = 6 v H = 1
(CH3)2CH is higher priority than CH3CH2 on LHS C
as atomic numbers of atoms joined to C (joined to C=C) are higher for (CH3)2CH than CH3CH2
highest priority groups on opposite sides (of C=C)
Therefore compound J is a E isomer (6)
1-chloropropane can also be produced by the reaction between propane and chlorine in the presence of ultraviolet light. State why ultraviolet light is needed for this reaction to occur. (1)
provides energy to break (covalent) bond in chlorine
OR
to form chlorine free radicals (1)
Explain the differences between structural isomerism and stereoisomerism. Use examples to show how compounds with the molecular formula C4H8 exhibit stereoisomerism and the three types of structural isomerism. (6)
Stereoisomers are compounds with the same structural formula but have different arrangement of atoms in space
This occurs as there is a restricted rotation around the C=C bond
Structure of E and Z-but-2-ene
correct identity of E and Z isomers
Structural isomers are compounds with the same molecular formula but have a different structural formula
Structural isomers
different C chain, e.g. methylpropene & but-1-ene / but-2-ene
different position of functional group e.g. but-1-ene & but-2-ene
different functional group, e.g. cyclobutane & but-1-ene / but-2-ene / methylpropene
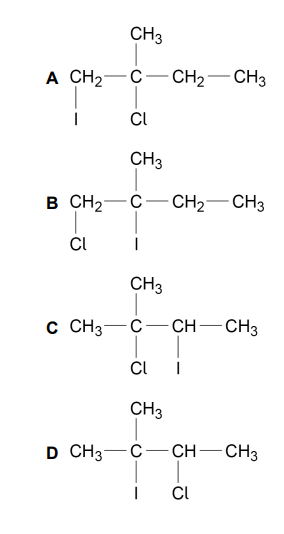
Which is the major product of the reaction between 2-methylbut-2-ene and iodine monochloride (ICl)? (1)
C (1)
This is because ICl is polarised as Iδ⁺—Clδ⁻
How many structural isomers with an unbranched carbon chain have the molecular formula C4H8Br2?
A 4
B 5
C 6
D 7 (1)
C (1)
1,1-dibromobutane
1,2-dibromobutane
1,3-dibromobutane
1,4-dibromobutane
2,2-dibromobutane
2,3-dibromobutane