Topics 9 & 19
1/33
Earn XP
Description and Tags
Redox Processes
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
oxidation
the loss of one or more electrons from a substance
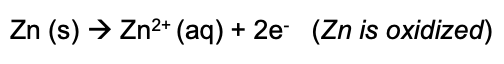
Also, in its simplest level, can be considered as a reaction in which a substance combines with oxygen (gains oxygen) or removes hydrogen
reduction
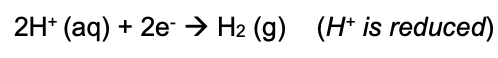
the gain of one or more electrons
In its simplest level, can also be considered as a reaction in which a substance removes oxygen or add hydrogen
What do oxidation and reduction always do?
They always occur together (OIL RIG - Oxidation Is Loss & Reduction Is Gain)
oxidation number
a concept providing a way to keep track of electrons in redox reaction according to certain rules
Covalent compounds - more electronegative element forming negative ion
Oxygen always has oxidtaion number of -2 except in following two situations:
H2O2 (oxygen is assigned -1 in peroxides)
OF2/F2O (oxygen is assigned +2 in this compound)
Hydrogen always has an oxidation number of +1 except when it’s part of a metal hydride:
Ex: NaH, CaH2; (hydrogen is assigned -1)
Fluorine always has an oxidation number of -1
carbonate
CO3-2
sulfate
SO4-2
What three elements always have the same charge (and what are their charges), even though they are in the d-block?
Zn (+2), Cd (+2), Ag (+1)
phosphate
PO43-
nitrate
NO31-
hydroxide
OH-1
chlorate
ClO3-1
ammonium
NH4+1
How to balance redox reactions in acidic solution?
Don’t do the thing with OH-
How to balance redox reactions in basic solution?
H+ ions appearing in final equations must be neutralized by adding an equivalent number OH-1 ions (forming H2O molecules) to each side of the equation
oxidizing agent
a substance that is able to oxidize other substances (undergoes reduction itself)
reducing agent
a substance that is able to reduce other substances (undergoes oxidation itself)
voltaic cell
a device used to obtain electrical energy from a spontaneous chemical reaction
batteries are examples of voltaic cells
also known as a galvanic cell
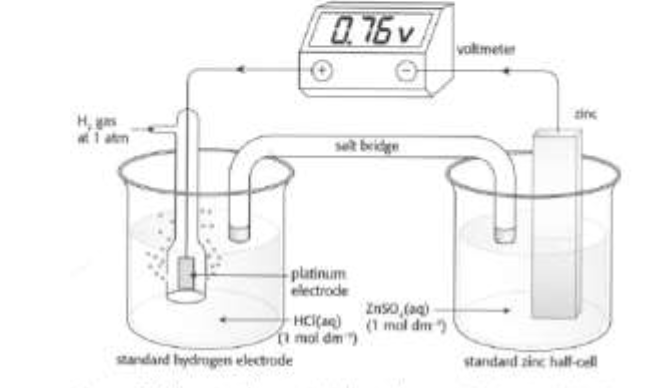
Voltaic cell diagram
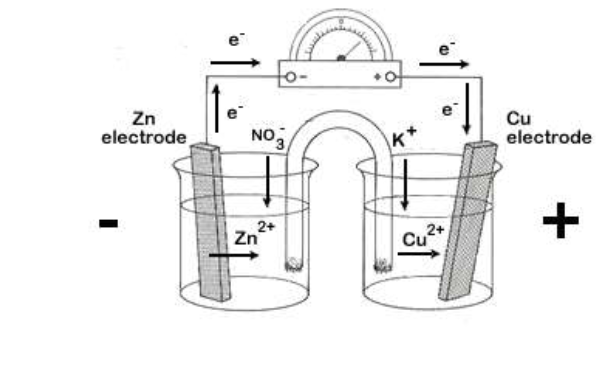
each beaker is referred to as a half-cell
oxidation occurs in the half-cell on the left side of the diagram
reduction occurs in the half-cell on the right side of the diagram
electrodes: the solid metals that serve to transfer electrons from one half-cell to another (a conducting wire connects the electrodes)
anode: the negative electrode in the half-cell where oxidation occurs
cathode: the positive electrode in the half-cell where reduction occurs
salt bridge: completes the circuit by preventing the build-up of charge by allowing ions to flow from one solution to another.
consists of a glass tube filled with a saturated solution of KNO3 gel with a porous plug at each end.
voltmeter: measures the potential difference in volts (V)
electrolytic cell
a device that uses electrical energy from an external source to make a non-spontaneous chemical reaction take place
uses include:
recharging batteries
producing metals like sodium, potassium and aluminum
electroplating materials
producing hydrogen and oxygen gas from water
unlike a voltaic cell, an electrolytic cell doesn’t contain two separate half-cells or a salt bridge
an electrolytic cell requires an external power source because the reaction is non spontaneous
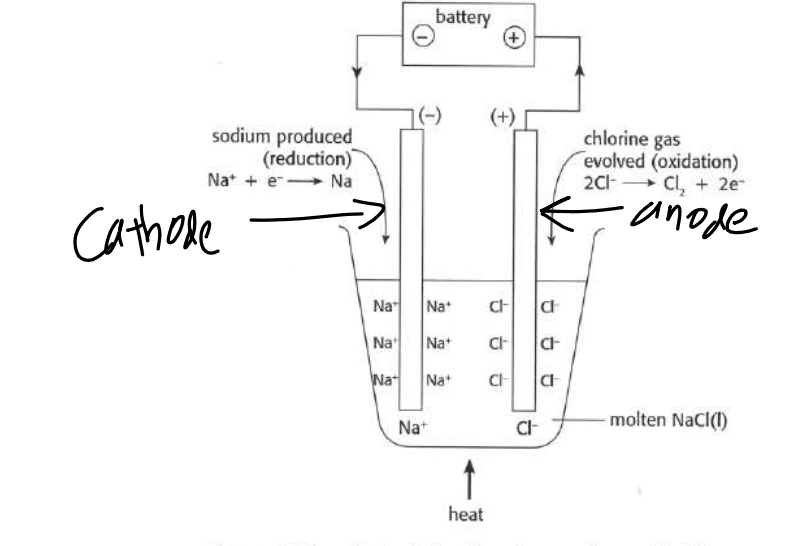
How do the anodes and cathodes of a electrolytic cell work?
similar to a voltaic cell in that oxidation still occurs at the anode and reduction still occurs at the cathode
different than a voltiac cell in that the anode is positively charged and the cathode is negatively charged in an electrolytic cell
electrolyte
a substance that conducts electricity in an electrolytic cell
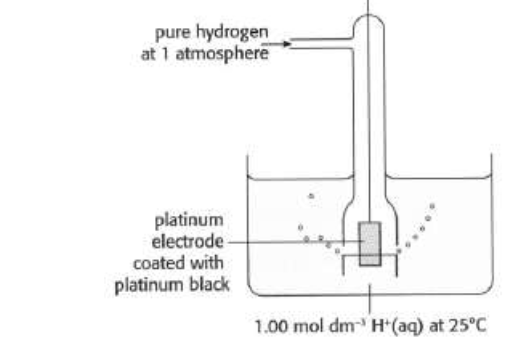
what’s the standard hydrogen electrode?
consists of a platinum electrode (inert) surrounded by hydrogen gas at 100 kPa (~1 atm) of pressures
the platinum electrode is immersed in an aqueous solution of acid in which the concentration of H+ ions is exactly 1 M (1 mol*dm3)
the temp is maintained at 298 K
assigned a standard electrode potential of 0.00 V as a reference point (like C-12 isotope is the basis of comparison for all atomic masses)
How are the standard electrode potentials of other half-cells determined?
It’s determined by connecting the half-cell to the standard hydrogen electrode using a salt bridge and connecting wire under standard conditions
what is standard electrode potential (Eθ)
the potential (“pull“ on the electrons from the oxidizing agent) of a half-reaction under standard state conditions, as measured against the potential of the standard hydrogen electrode.
is an intensive property that doesn’t change even when the reaction is multipllied by an integer for balancing
but, if reaction is reversed, the sign of Eθ is also reversed
How to calculate cell potentials using standard electrode potentials?
the reaction with the more positive Eθ will undergo reduction
the reaction with the more negative Eθ will undergo oxidation
When will a reaction be spontaneous using standard electrode potentials?
a reaction will be spontaneous if Eθcell is positive
Use cell potential to calculate the standard change in Gibbs energy for a redox reaction
ΔGθ=−nFEcellθ
n = amount, in moles of electrons transferred in balanced equation
F = Faraday’s constant (96,500 Coulombs)/(1 mol e-)
Ecellθ = standard cell potential
1 Volt Coulomb (VC) = 1 Joule (J)
Predict and explain the products of electrolysis of dilute aqueous solutions
if the electrolyte is a dilute aqueous solution, the water present can also be oxidized or reduced
2H2O (l) + 2e- → H2 (g) + 2OH-1 (aq) Eθ = -0.83 V
this reduction reaction will occur at the cathode (-) unless the electrolyte contains the ions of a metal with an Eθ > -0.83 V.
2H2O (l) → O2 (g) + 4H+ (aq) + 4e- Eθ = -1.23 V
this oxidation reaction will occur at the anode (+) unless the electrolyte contains the ions of a nonmetal with an Eθ > -1.23 V
sulfates (SO4-2) tend not to oxidize:
S2O8-2 (aq) + 2e- → 2SO42- (aq) Eθ = +2.01 V
Describe the use of electrolysis in electroplating
Electroplating is achieved by passing an electrical current through a solution containing dissolved metal ions and the metal object to be plated
Uses include:
gold plating has decorative and practical uses. Electrical connectors are often gold plated to provide a corrosion resistant conductive surface
rhodium plating is used to produce “white gold“
zinc plating is used with “galvanized“ nails. Iron is coated or plated with zinc. Zinc is more easily oxidized than iron. A protective coating of zinc oxide is formed when the zinc oxidizes, thus protecting the underlying iron.
tin plating is used to protect steel food cans from reacting with their contents (tin is less easily oxiidized than iron).
Describe the differences between a primary, secondary and fuel cell
primary (voltaic cell): an electrochemical device that converts chemical energy from spontaneous redox reactions into electrical energy
secondary cell: the chemical reactions are reversible, so the battery can be recharged
fuel cell: an electrochemical device that converts the chemical potential energy in a fuel directly into electrical energy
fuel cells differ from voltaic cells in that fuel cells require a continuous supply of oxidant/fuel, while voltaic cells contain a finite amount of reactants contained within them
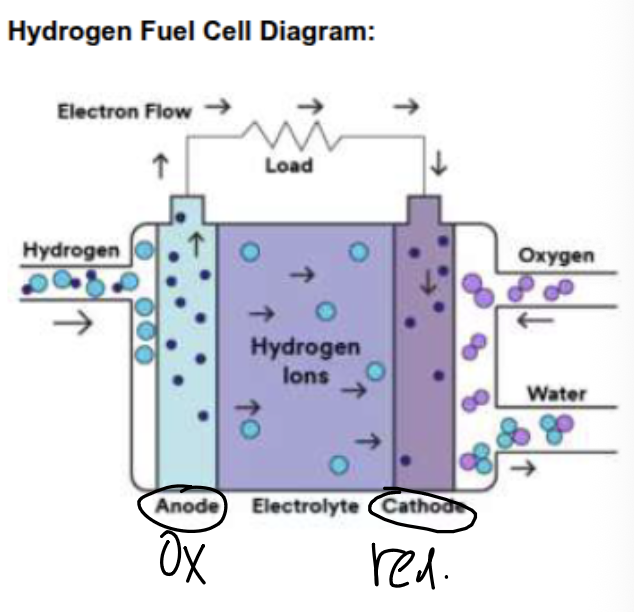
Describe the main features of a hydrogen fuel cell
The hydrogen fuel cell consists of:
a reaction chamber with separate inlets for hydrogen (H2) and oxygen (O2) gas
an outlet for the product, H2O
an electrolyte of aqueous sodium hydroxide
a semi-permeable membrane (PEM) that separates the hydrogen and oxygen gases (OH-1/H+ can pass through)
hydrogen gas is oxidized at the anode: H2 (g) → 2H+ (aq) + 2e-
the electrons cannot travel through the PEM, so they leave through the external circuit
oxygen gas is reduced at the cathode: O2 (g) + 4e- + 4H+ (aq) → 2H2O (l)
What are benefits of hydrogen fuel cells (compared to combustion reactions)
water is the only reaction product so no greenhouse gases are produced
the reaction takes place at room temp
there are no harmful oxides of nitrogen produced
What are the risks/problems of hydrogen fuel cells?
hydrogen is a highly flammable gas so production and storage are difficult
most hydrogen is produced as a by-product of the crude oil industry
What are the main features of a direct methanol fuel cell
A direct methanol fuel cell has the same components as the hydrogen fuel cell except it uses methanol to provide the H+ ions at the anode rather than hydrogen
At the anode the methanol and water react to form H+ and electrons which flow to the cathode where oxygen and the H+ react to form water
since this reaction requires water, a dilute solution of the methanol can be used (approx. 1 mol*dm-3)
This lowers the energy density, but it is still higher than hydrogen as a source in fuel cells
in most cells pure methanol is continuously fed into the system while water is recirculated, so the concentration of methanol remains constant
This type of fuel cell is not as “clean“ because it produces CO2