Alkene and Halogenoalkanes
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms

Mechanisms:
What does each colour on the boat represent?
What are the conditions for each reaction mechanism?
note- free radicals- X2= any halogen
Note- Electrophillic addition- H= hydrogen. X= halogen (X2 or HX happens at room temperature). Also note that HX stands for different electronegativities causing a permanent dipole.
Orange: Free radical substitution
Brown: Cracking
pink: Electrophillic addition
purple: Oxidation
Red: Nucleophillic substitution
green: Elimination

Polymer section is flashcards 13-39
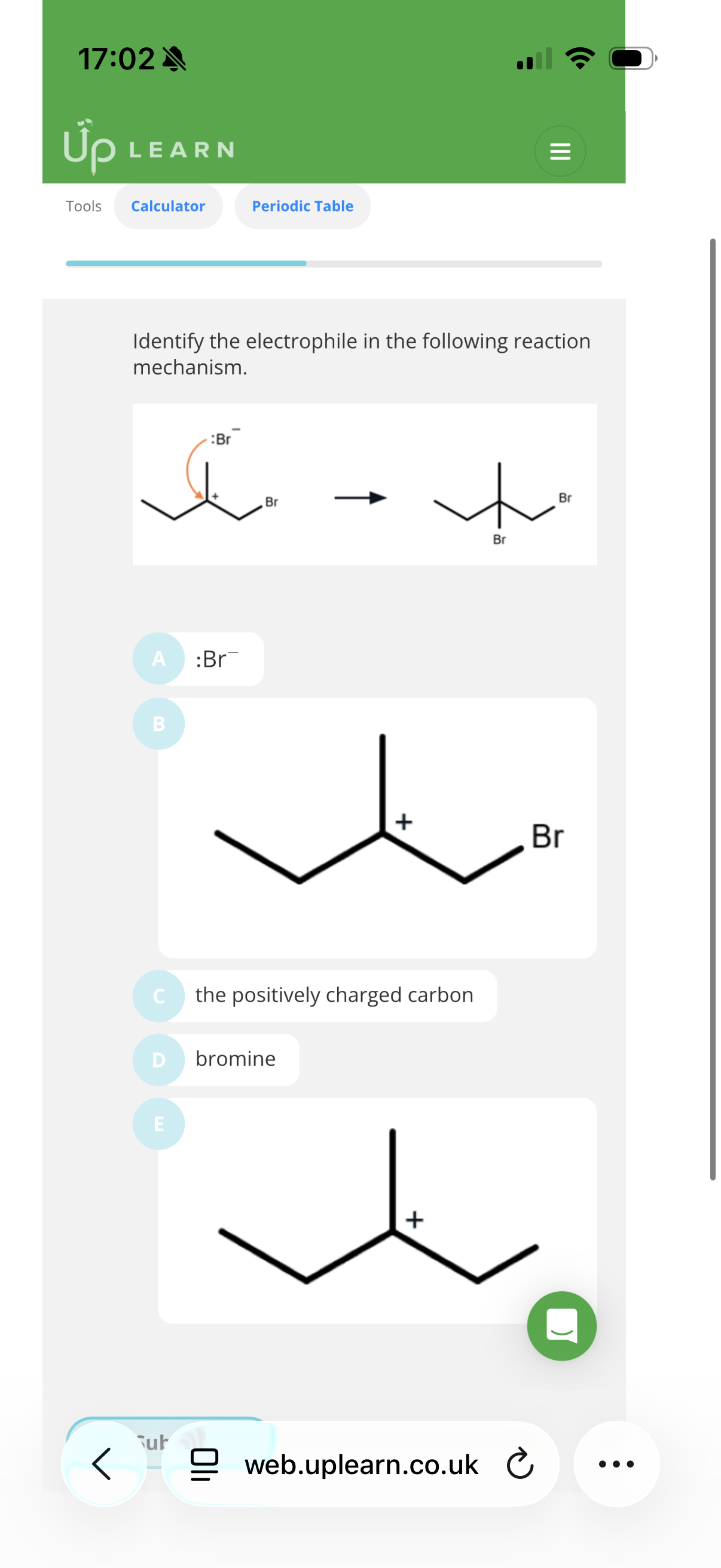
Nucleophiles and electrophiles:
What is a nucleophile?
What is an electrophile?
A nucleophile is a substance that donates an electron pair to form a new covalent bond.
An electrophile is a substance that accepts an electron pair to form a new covalent bond.
C and B
Answer image question?
Which of these species could act as a nucleophile?
Select all that apply
Correct answersYour answers
A: BH3
B: NH4+
C: PH3
D: SiH4
E: F−
B
C and E (Boron only has 3 valence electrons, so BH3 has no lone pair to donate to form a covalent bond)
(Nitrogen has formed 4 bonds, so has no lone pair to donate to form a covalent bond)
(Silicon only has 4 valence electrons, so SiH4 has no lone pair to donate to form a covalent bond)
(P is group 5 and only forms 3 bonds with H. 5+3=8 8/2=4 Since P has 3 bonds with H but wants to make 4, it has 1 lone pair.
Note:The arrow of the nucleophile goes to the most electron deficient area.

Electrophillic Addition
Note- An addition reaction is where a molecule is added to another molecule
What are the 3 questions that will help you solve any mechanism?
Why can alkenes do addition reaction?
What are the 2 conditions for stereoisomerism?
Using the image, explain how an induced dipole is produced Br2 when it gets close to an alkene? (Full details)
Now explain how an induced dipole is formed when an alkene reacts with a halogen?
Where do the arrows go in the first step of electrophillic addition when an alkene reacts with a halogen.
(Image on the other side shows what the answer looks like)
1) Are there any charges or dipoles?
2) Where are the most available electrons?
3) Where do the electrons want” to go?
Alkenes can do addition reactions because the double bond can break and form more bonds
• Restricted Rotation: There must be a double bond (which consists of a \sigma bond and a \pi bond) or a ring structure that prevents the atoms from rotating freely.
• Two Different Groups on Each Carbon: Each carbon atom in the double bond must be attached to two different groups.
•Alkenes have a double bond which is an electron dense area(4 electron shared).
•When Br2 gets close the shared electrons in the closest bromine will be repelled by the electrons in the double bond(same charges repel). So the electrons will move to the further bromine atom.
•This causes the Bromine closest to the double bond to be slightly electron deficient whilst the bromine further away from the double bond will be slightly electron dense/negative.
•Therefore a Br-Br will have an induced dipole from repulsion by C=C electrons.
The electron-rich C=C bond polarises an approaching electrophile by repelling its shared electrons, resulting in an induced dipole
1) The first arrow goes from the double bond to the electron deficient bromine(slightly positive) Q:Where do the electrons want to go
2)Since 2 electrons are pushed into Br2, 2 electron will be pushed out so the arrow wil be from the single bond in the Br-Br to the slightly electron dense (delta negative Br).


Electrophillic addition
What is a carbocation?
What is the intermediate?
Note: The image shows the electrophillic addition mechanism for ethene with bromine.
3 Draw the electrophillic addition mechanism for 1-chlorobut-2-ene with iodine?
note: remember for electrophillic addition,the nucleophile in the intermediate step with always have a lone pair and be a negatively charged ion.
What are the 2 conditions to turn an alkene into a halogenoalkane?(pink line)
A positively charged carbon
Middle step
HX or X2 (HX works because of the different electronegativities creating a permanent dipole)


Electrophillic addition
Outline the reaction mechanism with cyclohexene and bromine? (Answer is in this image)
Explain how an induced dipole is produced when alkene reacts with Br2?
State and Outline(draw) the mechanism when But-1,4-ene reacts with excess Bromine. Draw it in skeletal formula. (Answer is in the image on the back)
The electron dense C=C double bond causes the shared electrons in Br2 to move closer to the Br atom that is further away from the C=C bond. This causes Br-Br to become polarized.The negative electrons in the C=C double bond would therefore be attracted to the slightly positive bromine atom.


Electrophillic addition but using H-X (instead of X2)
An electronegativity difference above …1? Is considered a “polar” bond.
Why is H-X always polar?
Outline the electrophillic addition reaction mechanism when But-2-ene reacts with HCl?
Note- H-X has a dipole where the slightly positive region is H because halogens are more electronegative than hydrogens and so electrons are pulled closer towards the halogen.
0.4
Because the electronegativity difference is above 0.4


Electrophillic Addition
What are the 2 conditions to react an alkene to form an alcohol?(pink line)
Note: image has an example electrophillic addition mechanism of ethene reacting with H2SO4 -(also note it’s similar to alkenes reacting with HX but you need an extra arrow showing water is added)
Note:You draw phosphoric acid in the same way as sulphuric acid but you replace one of the oxygens double bond with a single bond + you add an extra H somewhere.
Outline the mechanism where propene reacts with H2SO4 then water? (In the other side image) Hint-use this image here to help
1)Cold sulfuric acid(H2SO4) then water or 2)Concentrated phosphoric acid(H3PO4) and steam(high temp and pressure)


Electrophillic addition: Markovnikov’s rule
What is a primary carbocation?
(secondary would then be attached to 2) (tertiary would be attached to 3)
Note- Alkyl groups donate electron density to the carbocation which makes it more stable.
Explain why secondary carbocations are more stable than primary carbocations?
note- Alkyl groups can push electrons,which we call a “positive inductive effect”. This helps stabilize a positive charge on a carbocation that forms in a reaction.
• A more stable …3? intermediate is most likely to form.
• Primary carbocations are always the …4? Product and less likely to form than secondary or tertiary carbocations which are most likely to be the …5? product.
Outline the mechanism for the reaction between 2-methyl-but-2-ene and sulphuric acid. (That forms the major product)
A primary carbocation is one where the carbon with the positive charge is attached to only one other alkyl group (carbon chain).
Secondary carbocations are bonded to more alkyl groups and these alkyl donate electron density to the carbocation which makes it more stable than primary carbocations.
Carbocation. 4.minor 5.major
6. (The major product will have the tertiary carbocation intermediate)(therefore the OH will be bonded to the Carbon that is bonded to 3 alkyl groups)

(Continued from q6 in last flashcard).
Draw the minor product of the reaction mechanism
Explain why more of the major product is likely to form compared to the minor product?
(The OH is bonded to a carbon that has 2 alkyl groups bonded it.
1)The major product is formed by a tertiary carbocation intermediate and the minor product is formed via a secondary carbocation intermediate.
2)The tertiary carbocation is more stable than the secondary carbocation because more alkyl groups donate electron density which stabilize the carbocation.


Polymers:
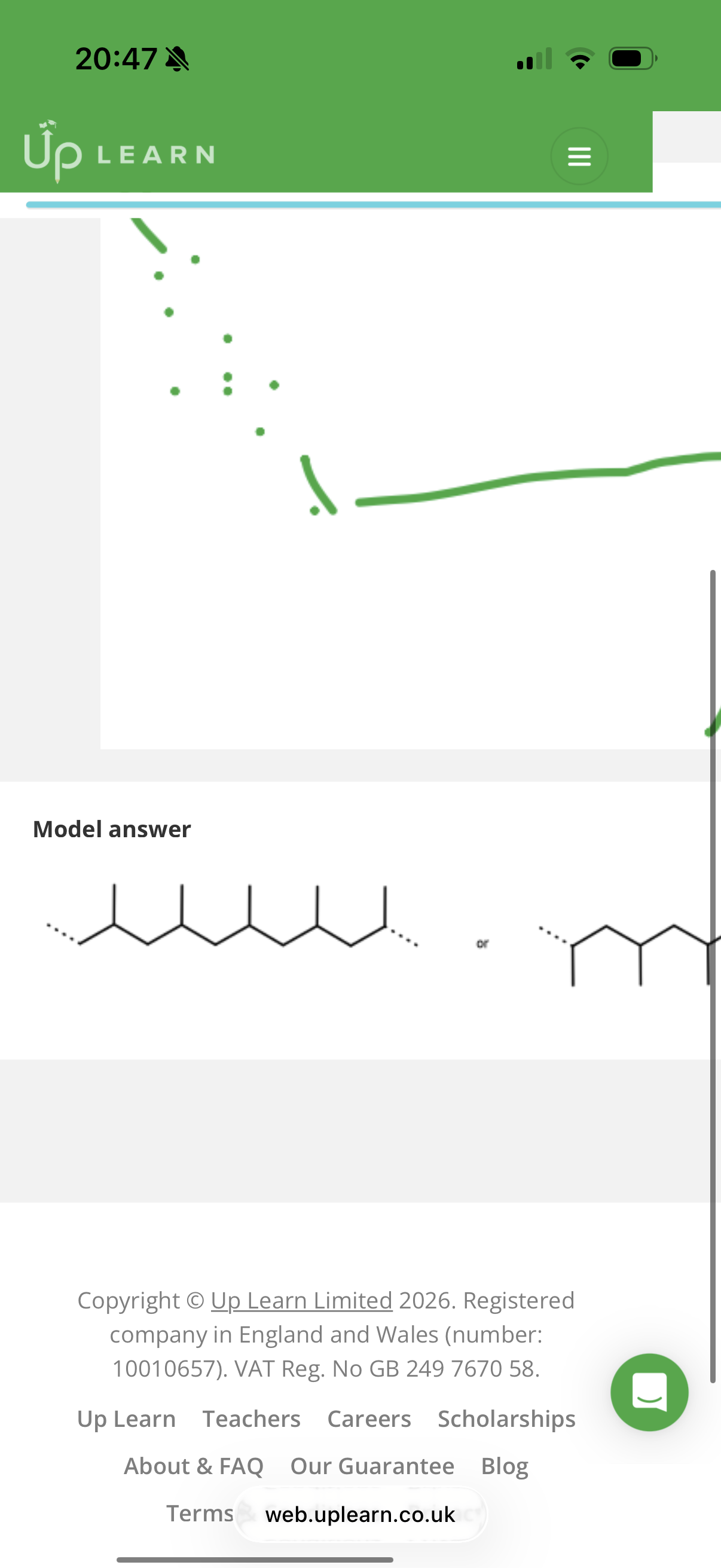
What is the only way to make polymers?
When naming polymers what should you add at the beginning of the nomenclature?
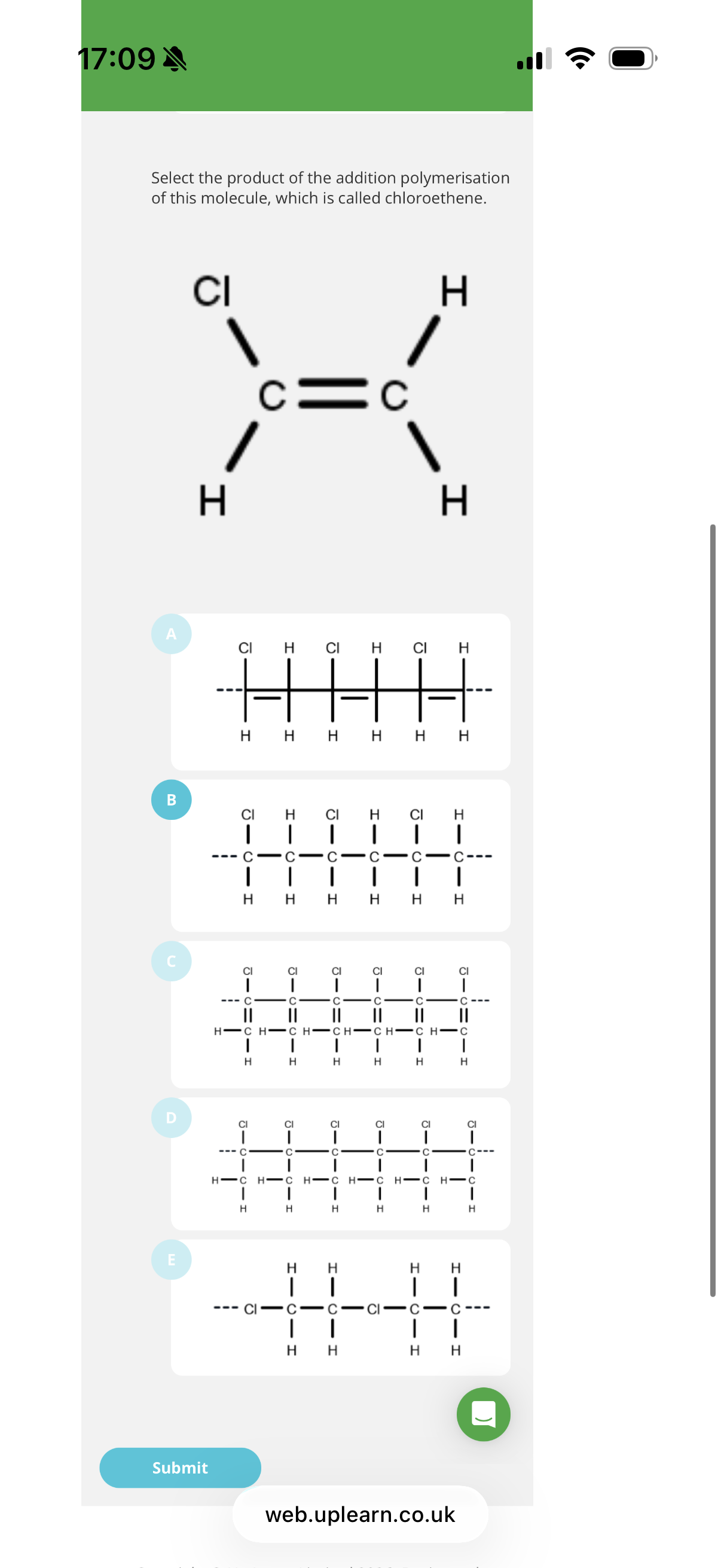
info: the image shows the polymerisation of 1-Chloro-1-bromo-2-Fluoro-Iodoethene into a polymer. (You always remove the pi bond and draw brackets over the extended lines.
what does inert mean?
Why are polymers non-polar?
Why do polymers have high melting points?
Select all the true statements about addition polymers.
A: Addition polymerisation produces macromolecules.
B: Addition polymerisation occurs whenever multiple molecules with double bonds are together.
C: The products of addition polymerisation must contain double bonds.
D: There’s no limit to the number of molecules which can participate in an addition polymerisation reaction.
E: There can be multiple products from an addition polymerisation reaction.
F: When molecules with one double bond undergo addition polymerisation, the product has no double bonds.
Addition polymerisation
Poly e.g. poly(ethene)
Non-reactive
The C-C and C-H bonds have no large difference in electronegativity.
They are made up of units repeating monomers and so have many electrons→ so have strong van der waals forces which require energy to break down.
A,D,F(B correction- Addition polymerisation occurs only under certain conditions.)
(C correction- The reactants in addition polymerisation reactions always contain double bonds)
(E correction- Only one type of product is formed in an addition reaction)
Answer image question?
The molecules which are added together in a polymerisation reaction are called…
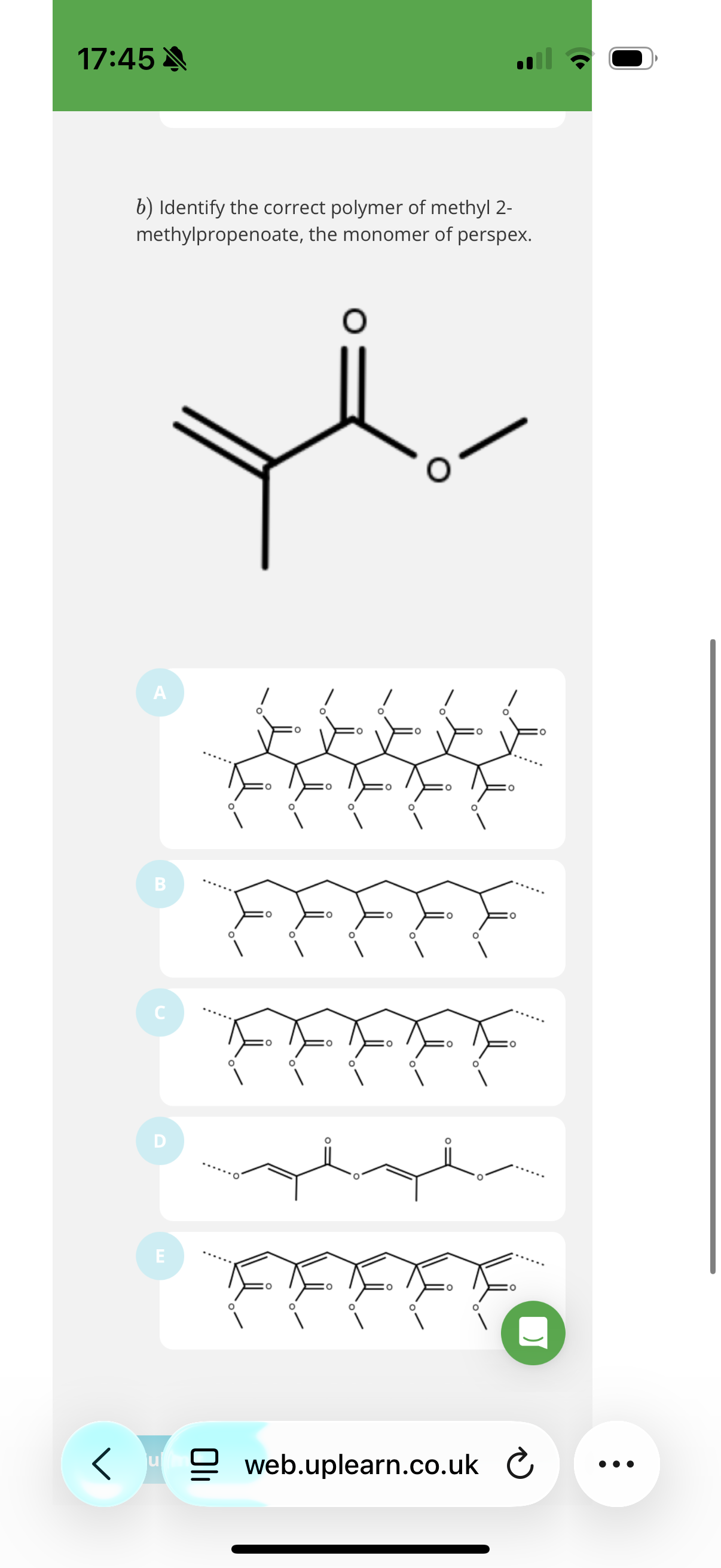
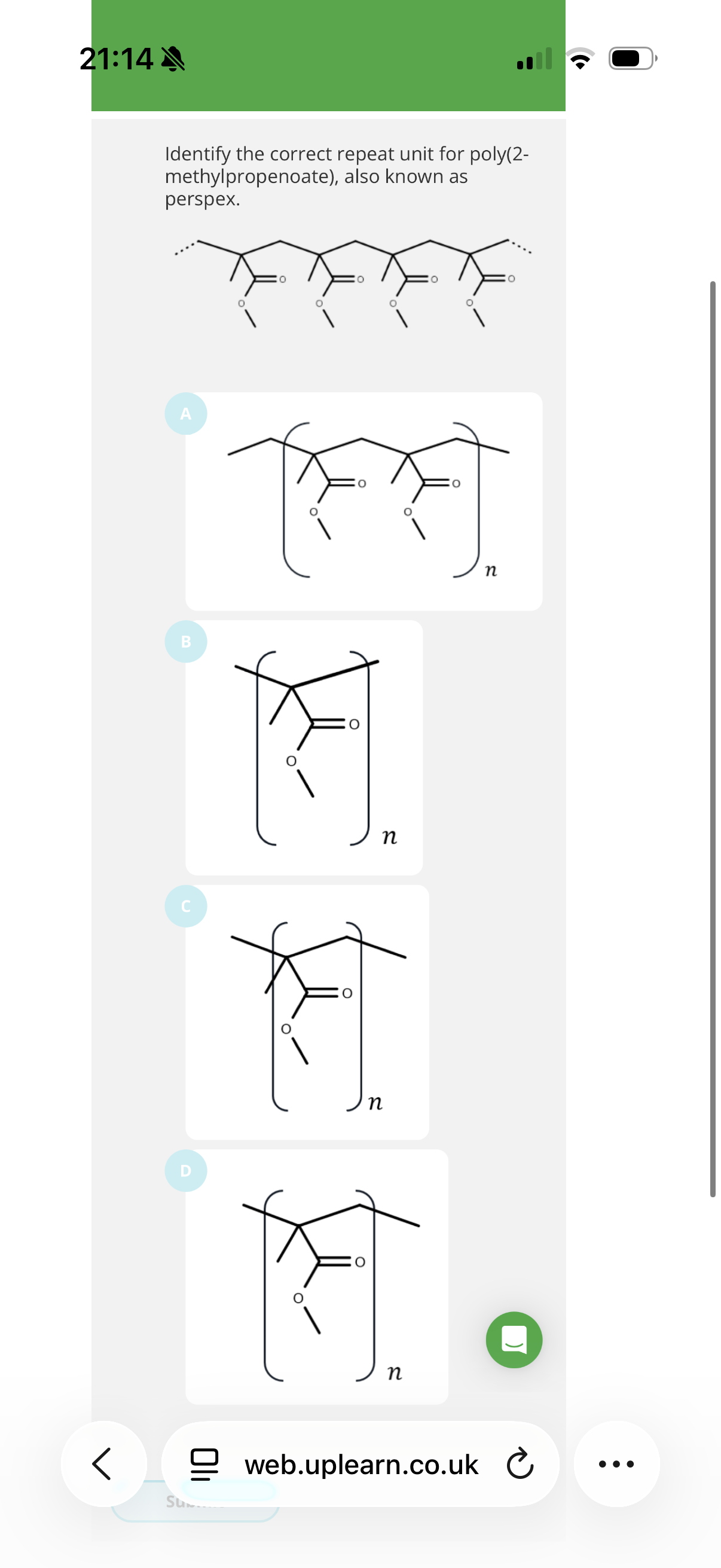
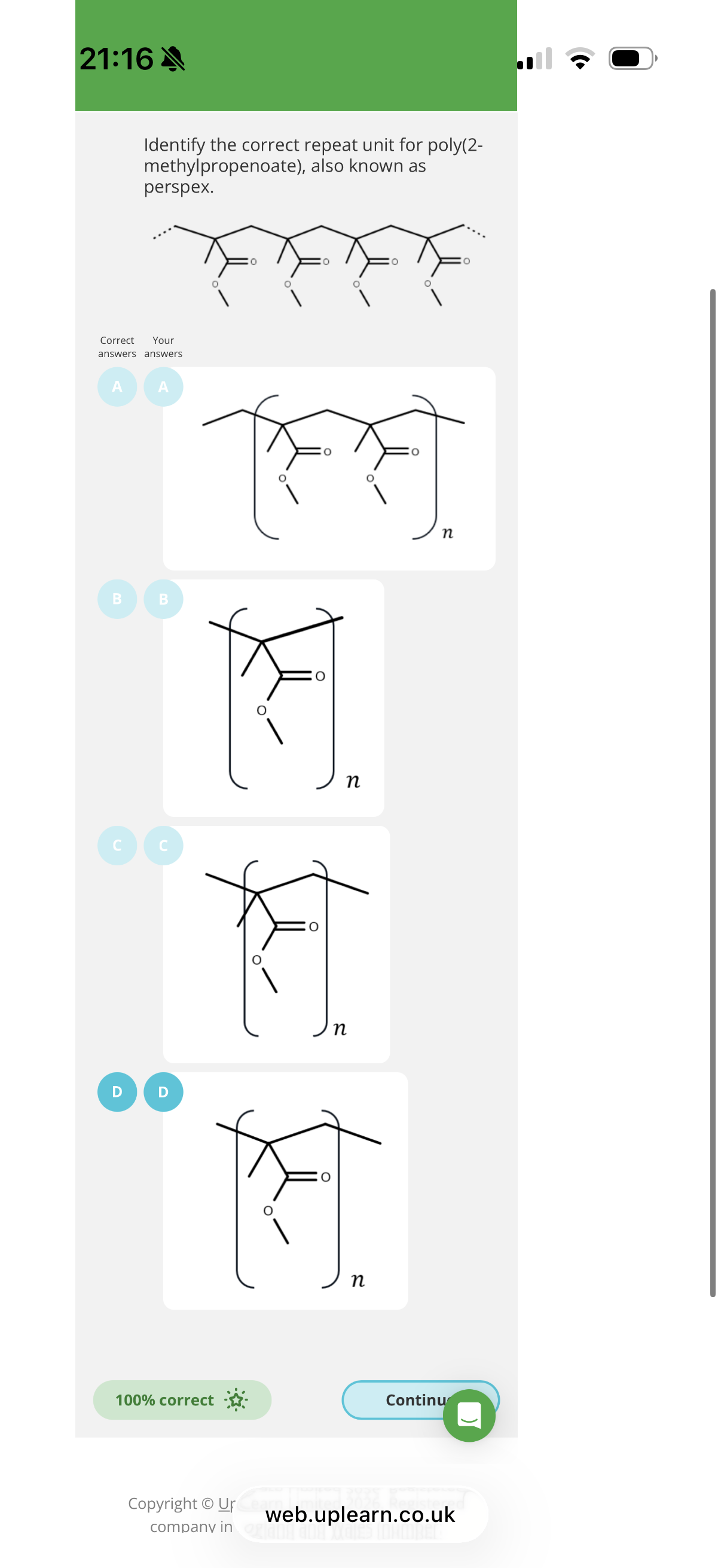
3.The molecule methyl 2-methylpropenoate can undergo addition polymerisation.
The product of this reaction is widely known as perspex.
Give the IUPAC name for this product.
4.A lump of ethene melts at −169°C.
All types of poly(ethene) melt above 100°C.
Why does poly(ethene) melt at a higher temperature than ethene?
Select all that apply
A: poly(ethene) chains have more electrons than ethene.
B: poly(ethene) has stronger van der Waals forces than ethene.
C: poly(ethene) has stronger hydrogen bonds than ethene.
D: poly(ethene) has more C-C bonds than ethene.
E: poly(ethene) has stronger C-C bonds than ethene.
A lump of poly(ethenol), made up of the polymer chains pictured above, is held together by...
A: Van der Waals forces only.
B: Permanent dipole-dipole forces only.
C: Hydrogen bonds only.
D: Van der Waals and permanent dipole-dipole forces.
E: Permanent dipole-dipole forces and hydrogen bonds.
F: van der Waals and hydrogen bonds
The IUPAC name for the polymer(in the image) commonly called PVC is…
Explain why addition polymers are usually solid at room temperature?
Explanation in image. 2.monomers
Poly(methyl 2-methylpropenoate). Anything after poly must be in brackets
4. A and B. 5.F
Poly(chloroethene)
Addition polymers are solid at room temperature because their long chains create strong van der waals forces between the polymer molecules, that require more energy to break, thus keeping them solid at room temperature.

we’ve seen that PVC has many uses, including…
Select all that apply
A: window-frames.
B: wall plug housings.
C:plastic water bottles.
D:aircraft windshields.
E: credit cards.
F: vinyl records.
G: glue.
H: inflatable boats.
I: phone charger cables.
2.PVC is normally quite rigid because…
Select all that apply
A: the carbon-chlorine bond is polar.
B: the carbon-chlorine bond is non-polar.
C: the carbon-chlorine bond is inert.
D: dipole-dipole forces act between PVC molecules.
E: hydrogen bonds are formed between PVC molecules.
F: van der Waals forces act between PVC molecules.
Which of these PVC products contain plasticiser? (Hint-flexible)
Select all that apply
A: A food container.
B: Cling film.
C: The coating of a USB cable.
D: Glass frames.
E: A shower curtain.
Info: Adding a plasticiser to PVC makes the plastic less brittle and more flexible. Plasticised PVC is used for electrical cable insulation and fake leather.
Why is Poly(ethene) more stable than ethene?
Addition polymerisation takes place…
Select all that apply
Correct answersYour answers
A: because saturated molecules are more stable than unsaturated molecules.
B: because saturated hydrocarbons are more stable than unsaturated hydrocarbons.
C: given the right conditions, like the presence of a catalyst.
D: given the right conditions, like a critical mass of monomers.
When chemists don’t want PVC to be rigid(not easily bent) they add a …?
A,B,C,E,F,H,I. 2. A,D,F. 3.B,C,
Because saturated hydrocarbons are more stable than unsaturated hydrocarbons
B,C
Plasticiser
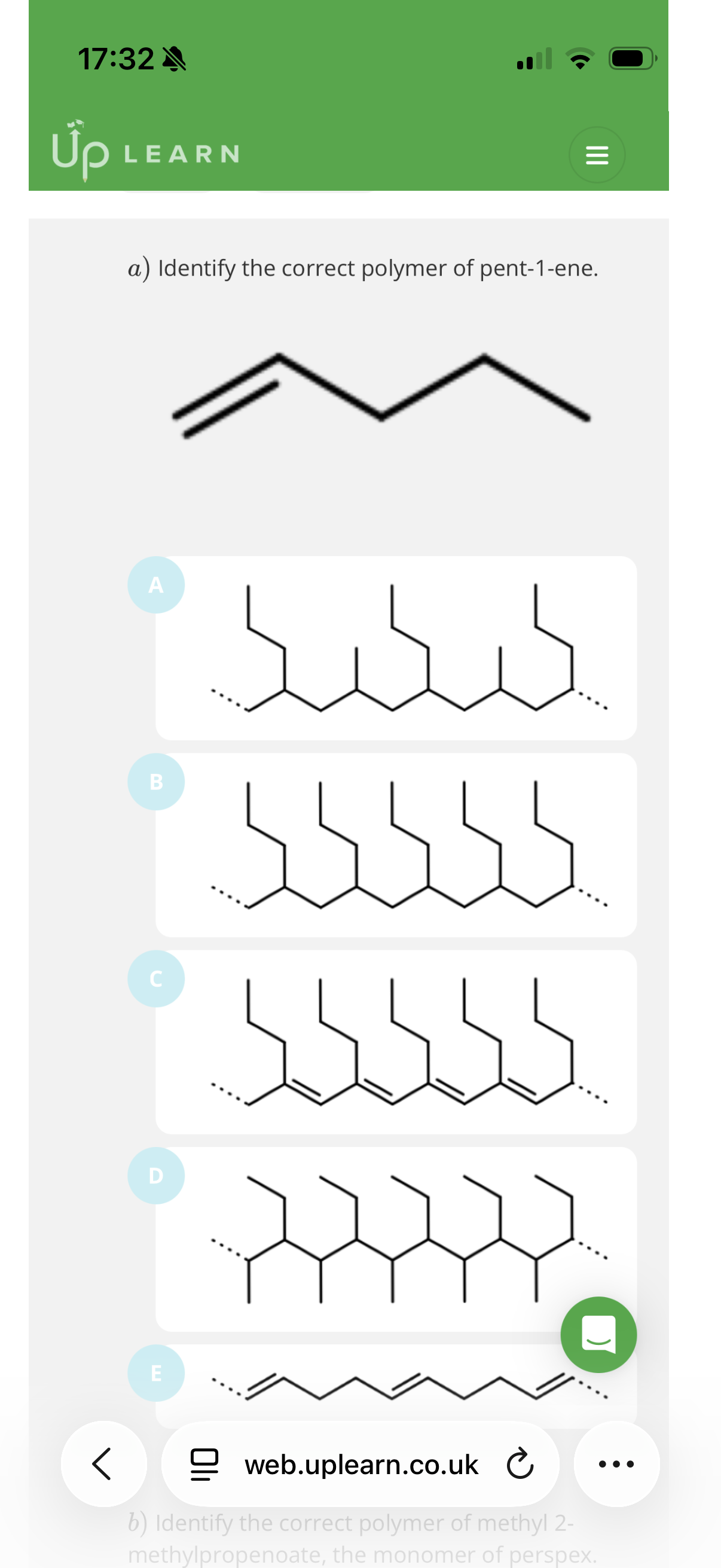
Image q?
The correct answer is B.
1. Identify the Monomer
The monomer is pent-1-ene. In skeletal formula, it looks like this:
• Carbons 1 and 2 have the double bond (C=C).
• Attached to Carbon 2 is a propyl group (a 3-carbon chain: -CH_2-CH_2-CH_3).
2. The Polymerization Rule
When pent-1-ene polymerizes:
1. The double bond between C_1 and C_2 breaks.
2. These two carbons form new single bonds to the next monomers, creating a continuous carbon-carbon backbone.
3. Everything else attached to those two carbons becomes a side chain (pendant group) hanging off that backbone.
3. Why B is Correct
In structure B, you can see a repeating unit where:
• The backbone consists of 2 carbons per monomer unit.
• Every second carbon in the backbone has a 3-carbon chain (propyl group) sticking out.
• This matches the structure of pent-1-ene perfectly.
-In this image, note that the blue highlighted bit shows the extended double bond line.The first section of the yellow bit contains the other extended dotted line of the double bond(which is coloured fully black). The side chain that is shown between the blue and yellow circle is there because everything other than the carbons in the double bond becomes a side chain. Now the second part of the yellow circle shows where pent-1-ene repeats again and note that the extended dotted line of that pent-1-ene is coloured fully black since it is joined with the first repeating units extended line(which like I mentioned way earlier is coloured fully black). If u look at the end of the whole polymer, you will see that it ends off with a dotted line, so this is reassurance if u didn’t understand what u was saying earlier
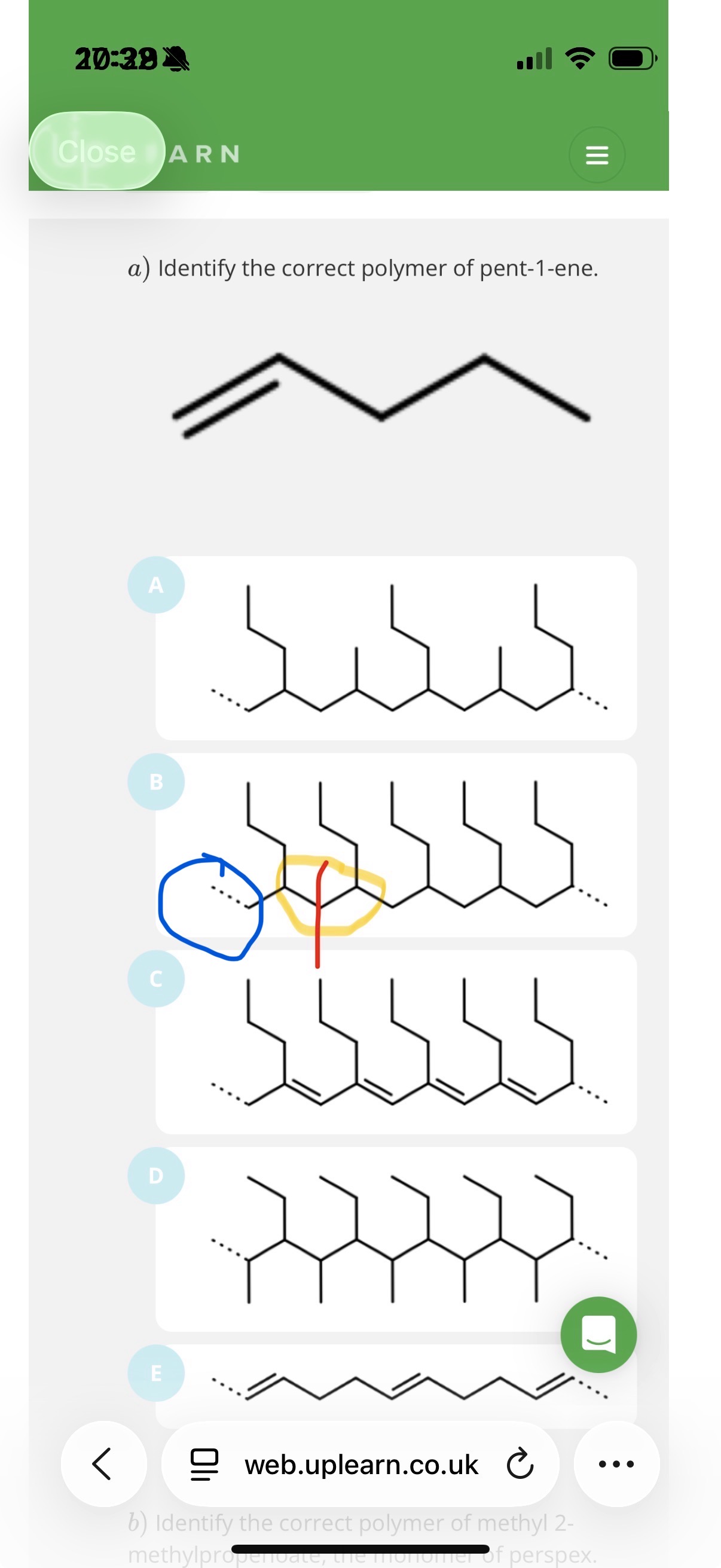
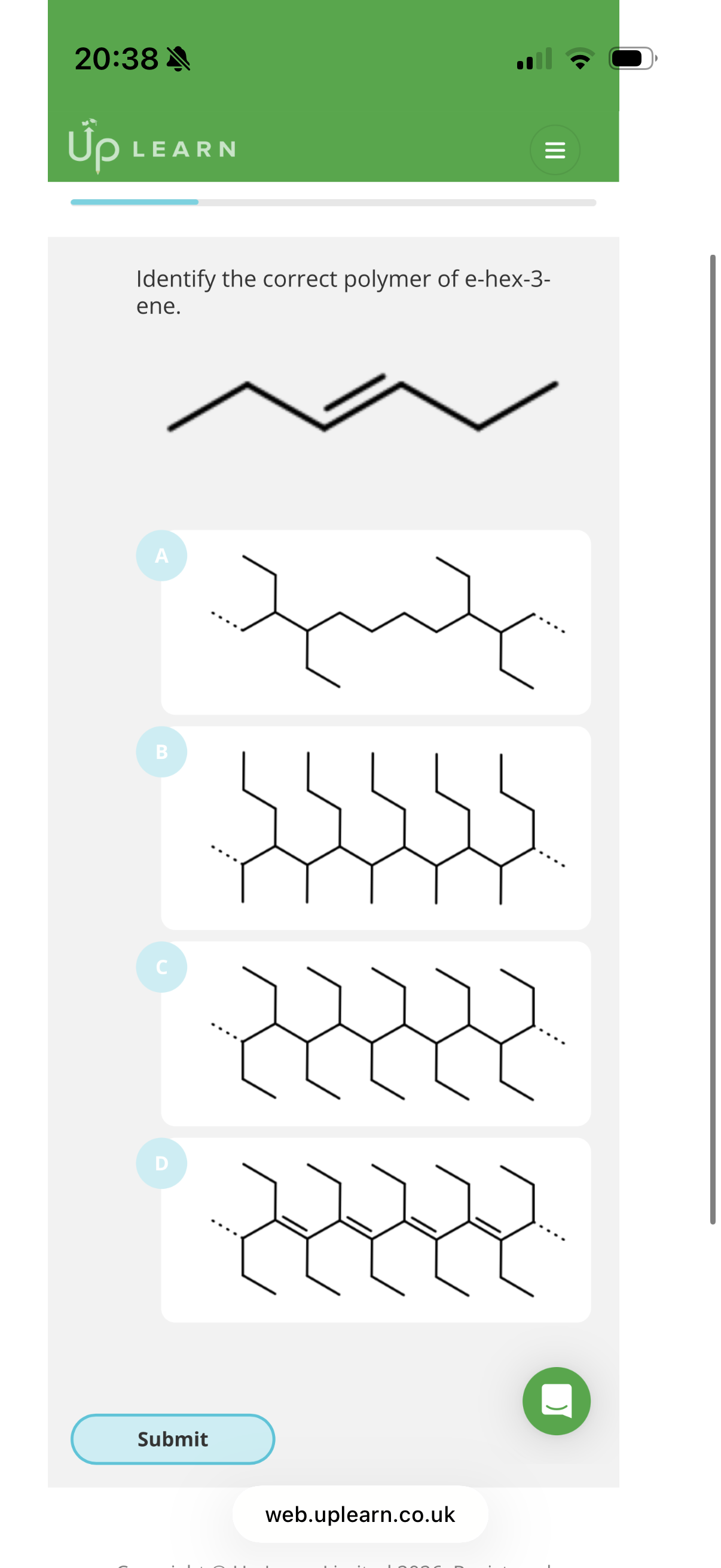
Image a?
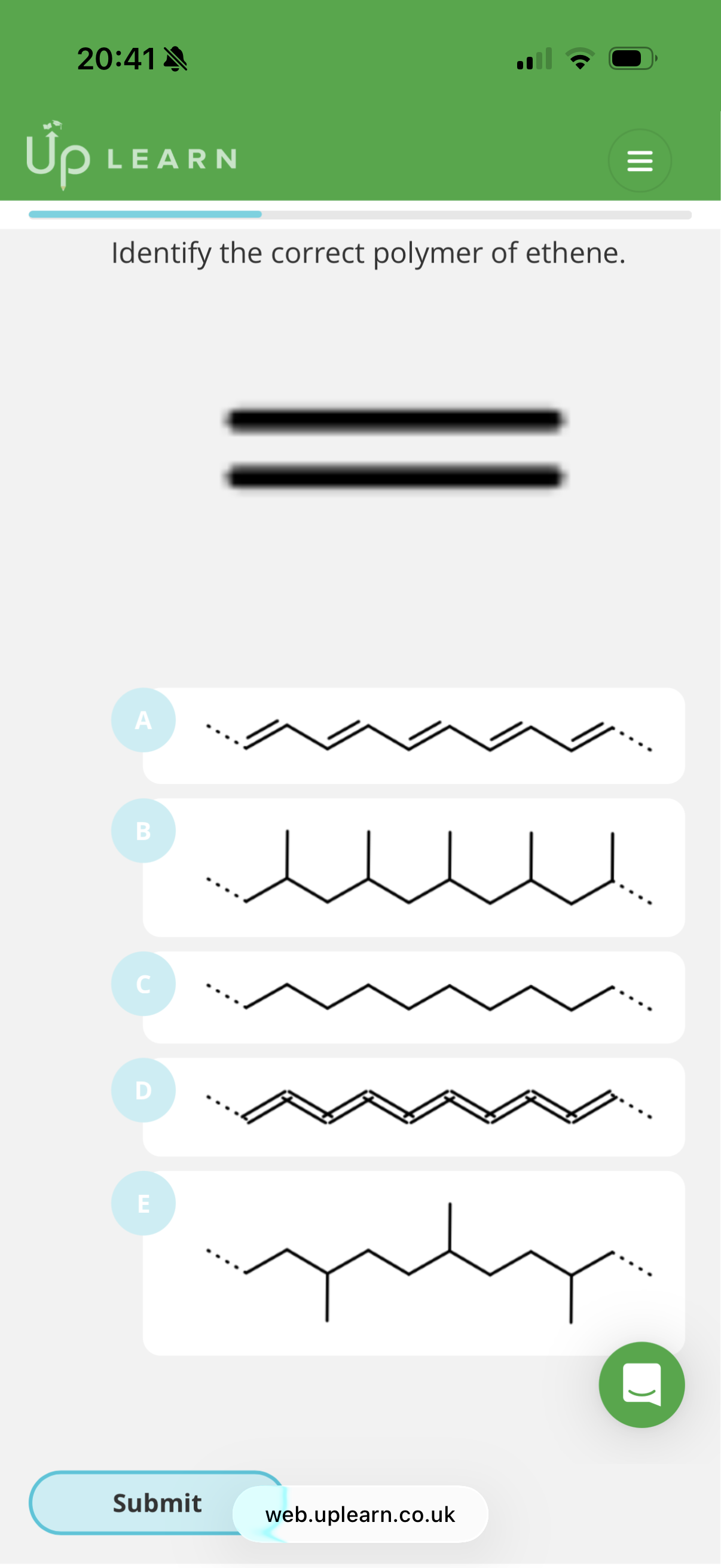
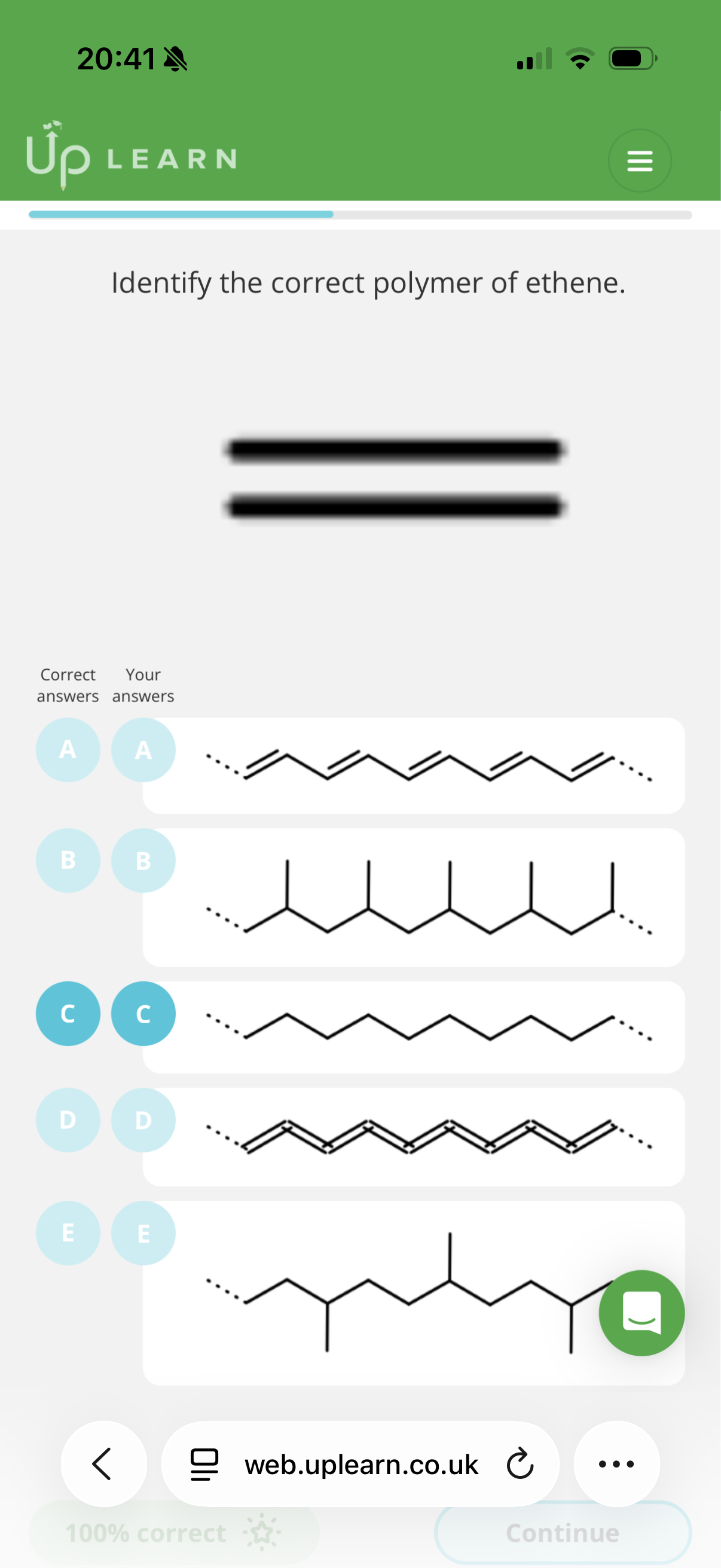
Answer image q?
Answer image q?
C
Identify the Monomer Parts
Look at the carbons involved in the double bond (C=C):
• The Left Carbon (CH_2): This carbon has two hydrogens (not shown).
• The Right Carbon: This is the "busy" carbon. It has two specific groups attached to it:
1. A methyl group (-CH_3, the single line pointing down).
2. An ester group (-COOCH_3, the group with the C=O and -O-).
2. Form the Polymer
When this molecule polymerizes, the double bond opens up to form the backbone. The "busy" carbon must have both the methyl group and the ester group hanging off it at the same point.
• The Backbone: A repeating pattern of two carbons (C—C—C—C).
• The Side Chains: Every second carbon in that backbone should have one methyl group AND one ester group.
3. Why C is the Winner
In structure C, if you look at the "peak" of every zig-zag in the backbone:
• Pointing up/out is the methyl group.
• Pointing down is the ester group.
• The carbons in between the peaks (the "valleys") have nothing but hydrogens (implied), which matches the CH_2 side of our monomer.
Why the others are wrong:
• A: This structure shows two ester groups on the same carbon, which doesn't match our monomer.
• B: This is missing the methyl group side chain entirely. It only shows the ester group.
Answer image q?
A
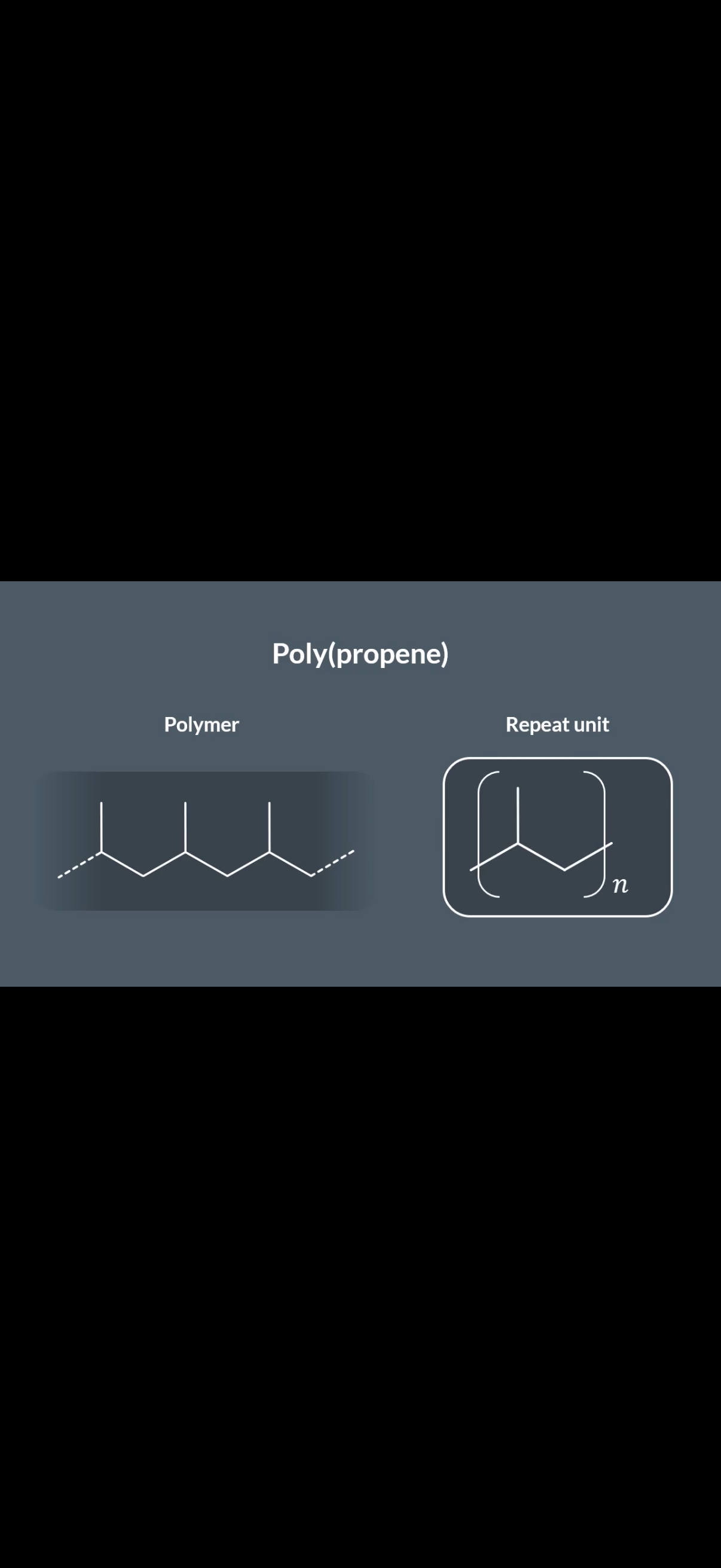
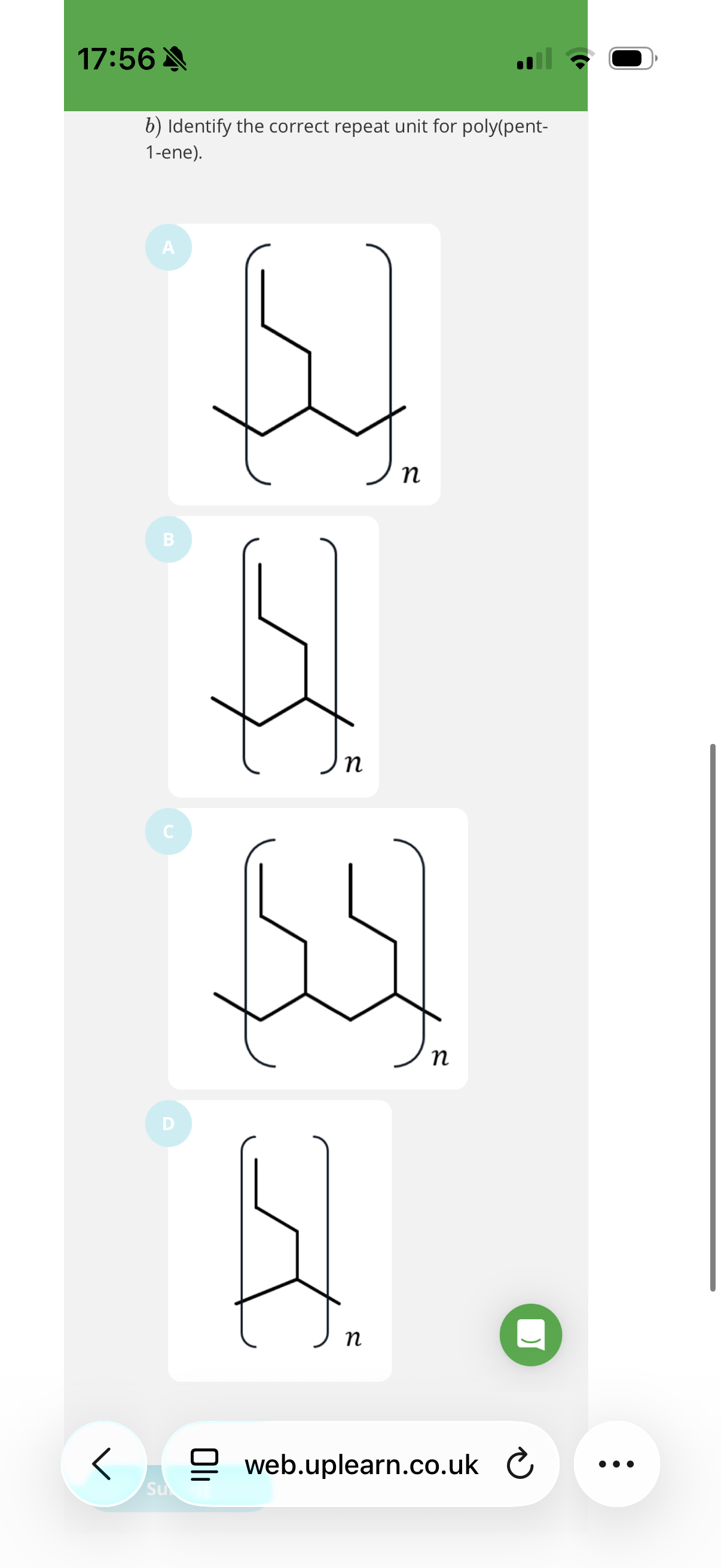
Image q?
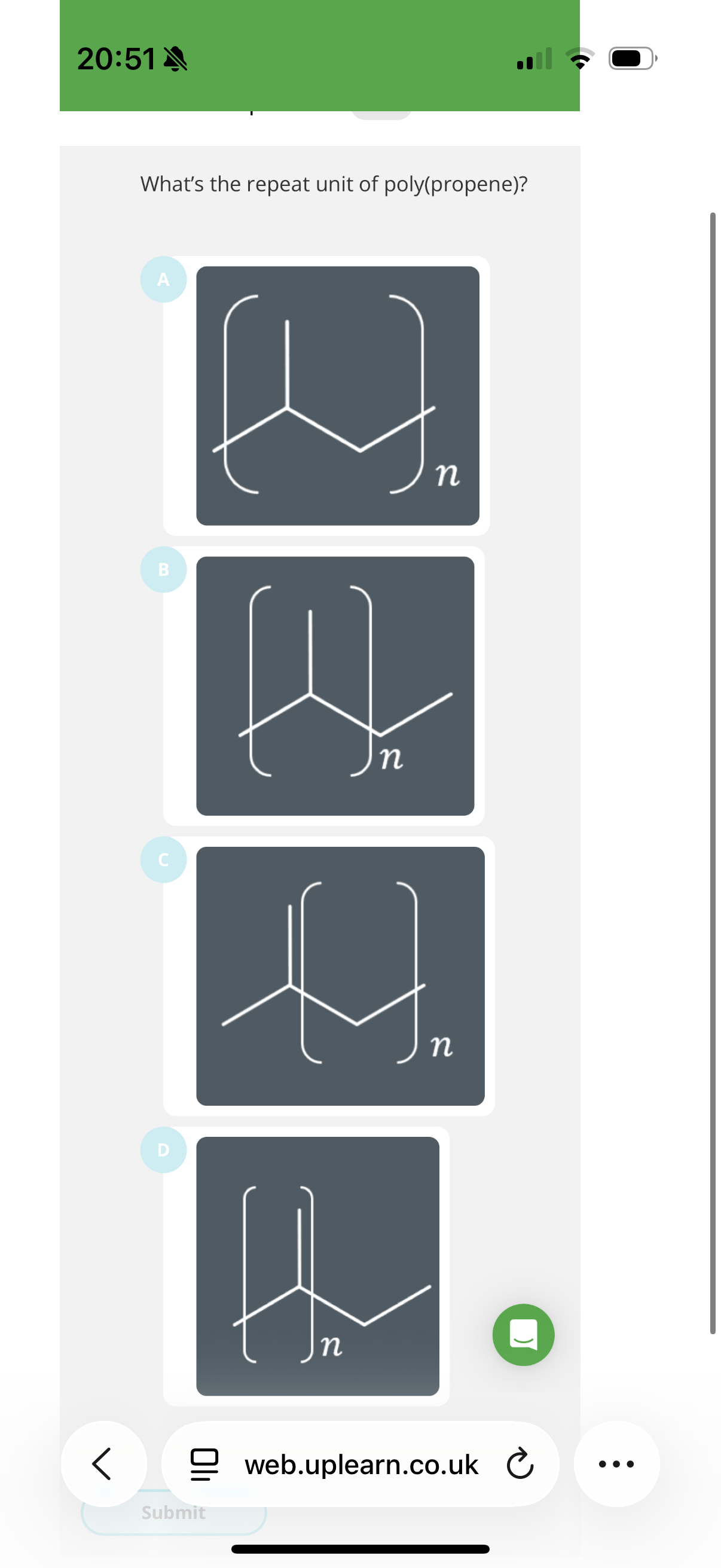
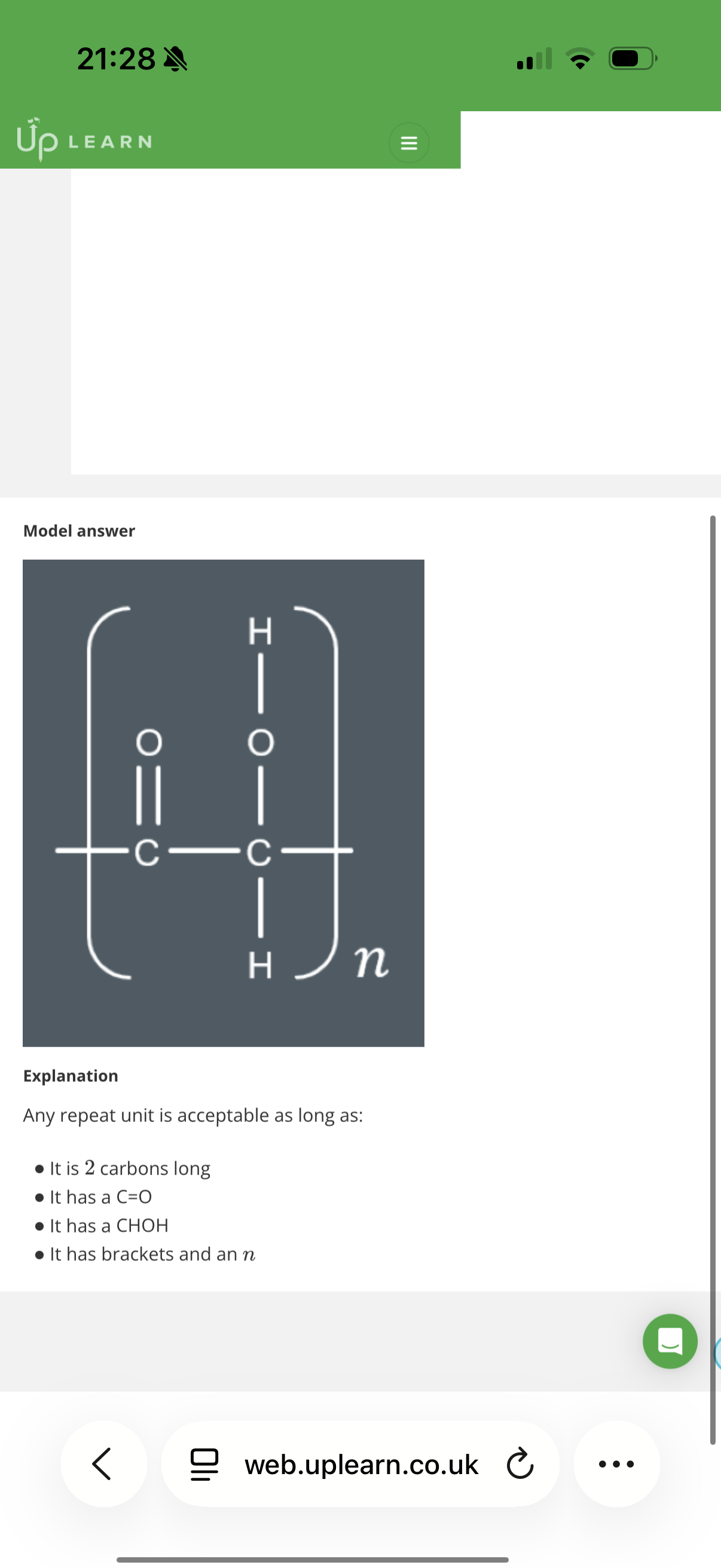
The repeat unit is the shortest section of the polymer that is repeated.
But by convention, we never show a repeat unit shorter than …2? carbons.
Answer is B
Two
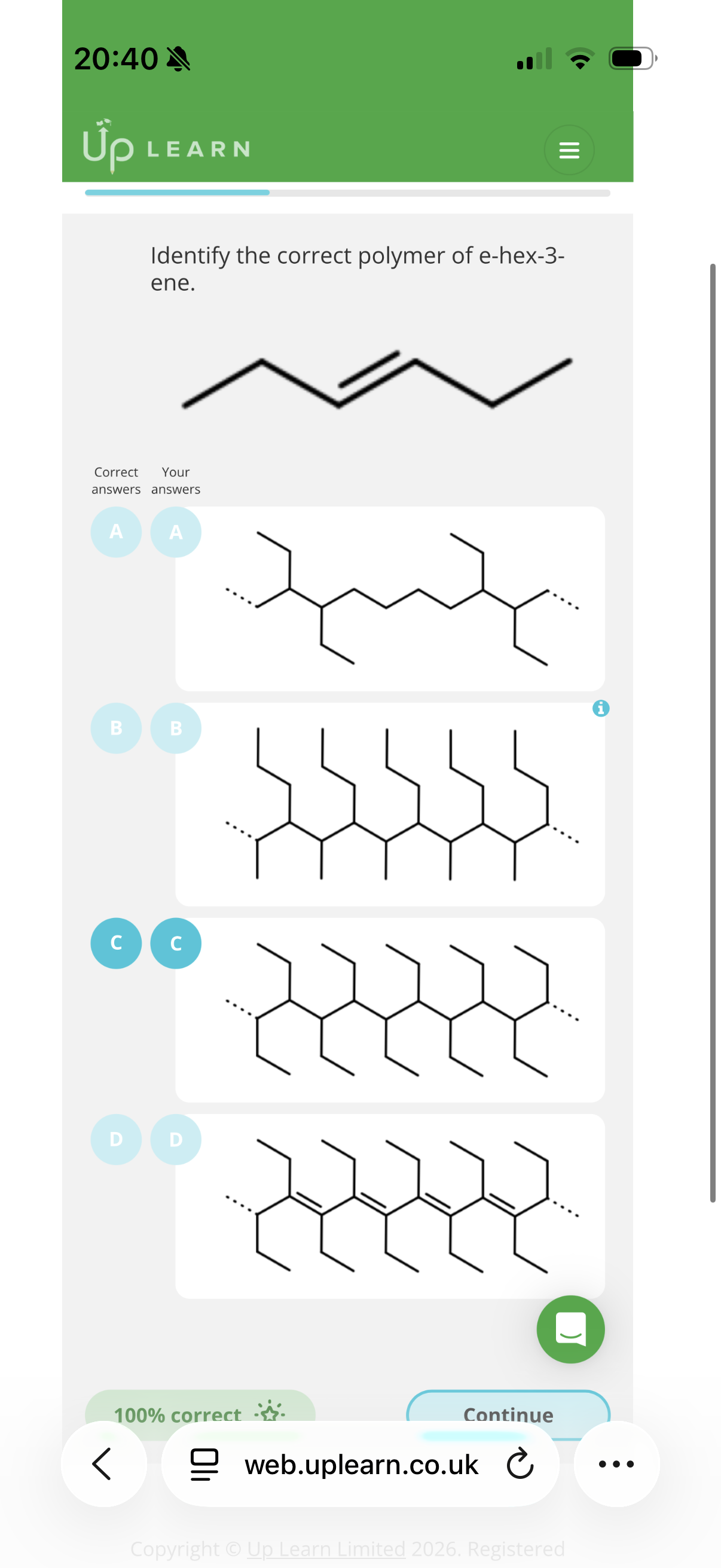
Image q?
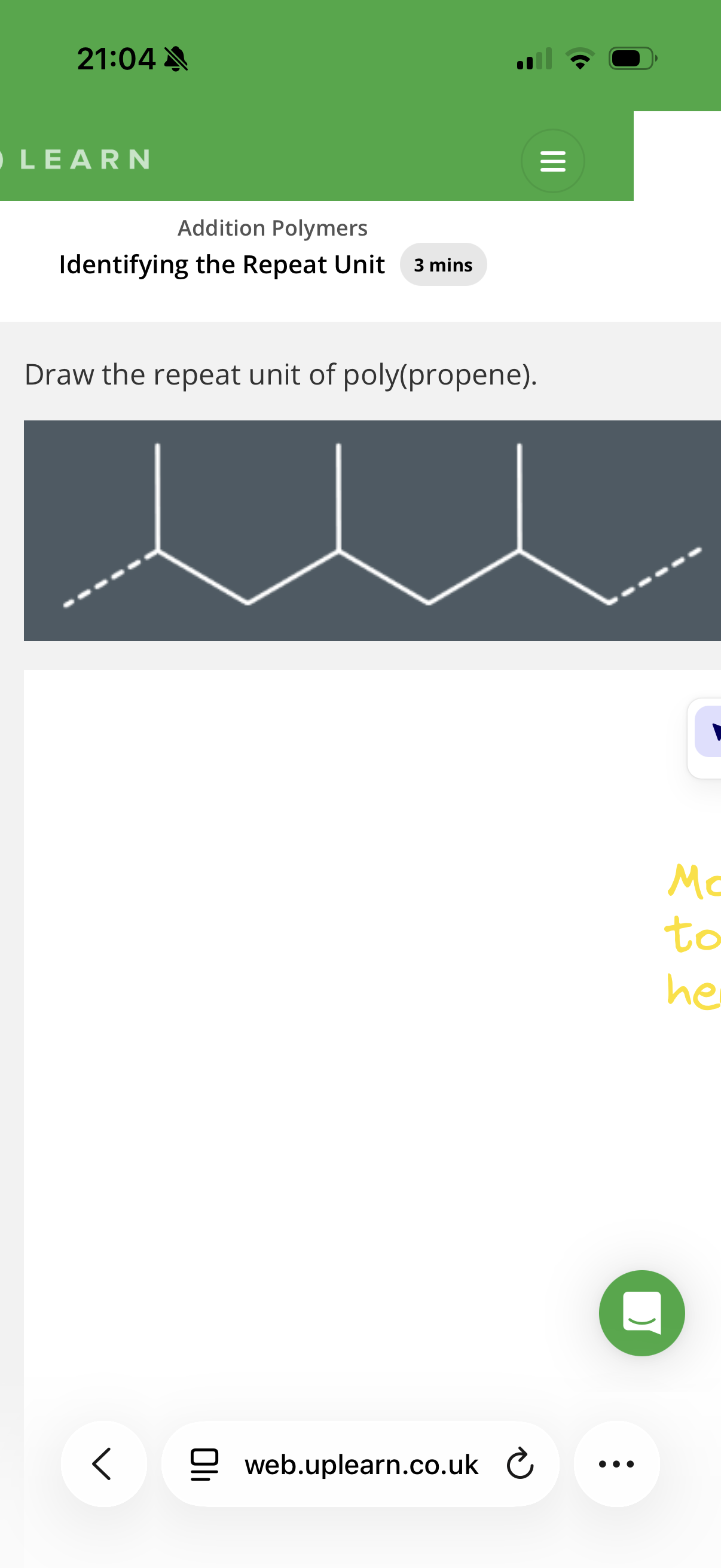
Answer image q?
Hint: To identify repeat units, look at the carbons in pairs and see if they have the same substituents as the other pair of carbons.
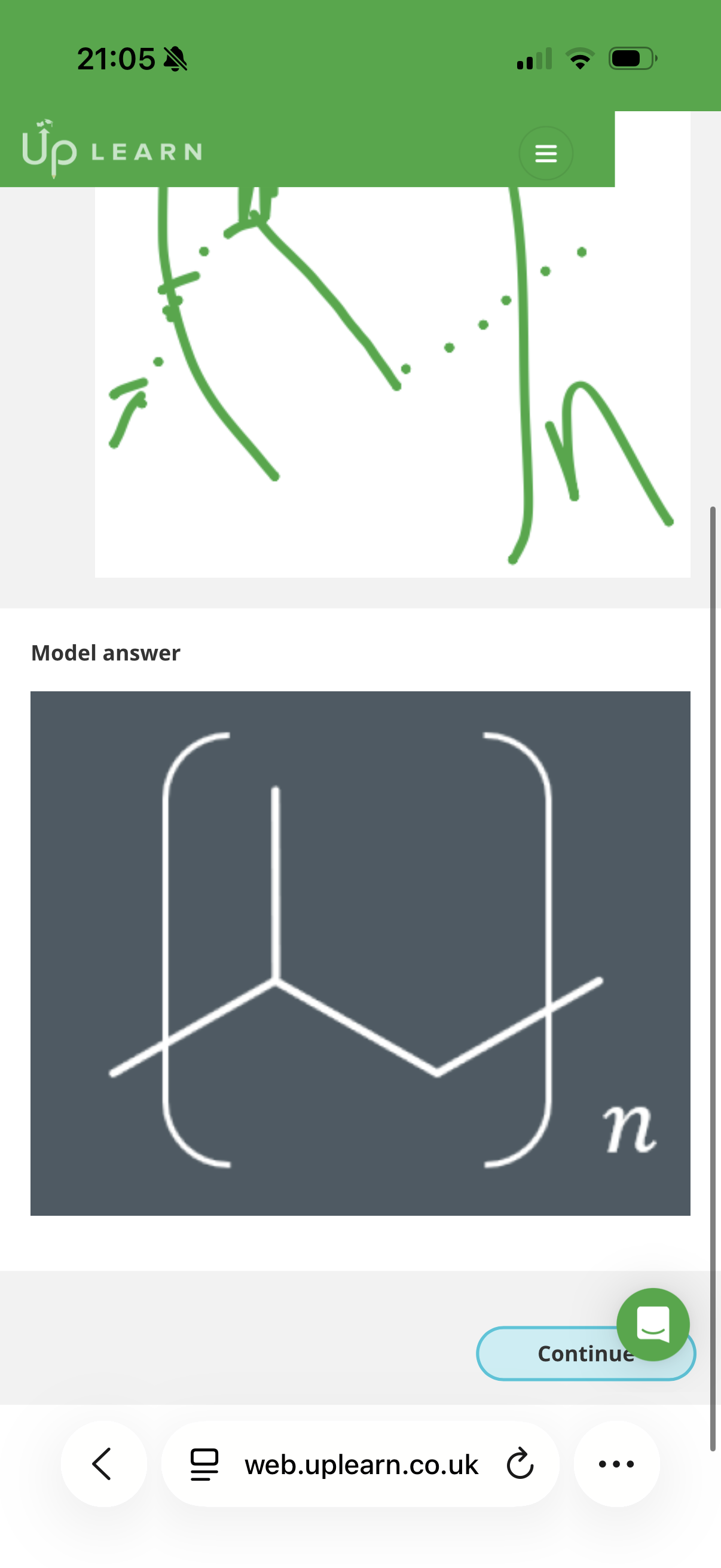
Answer image q?
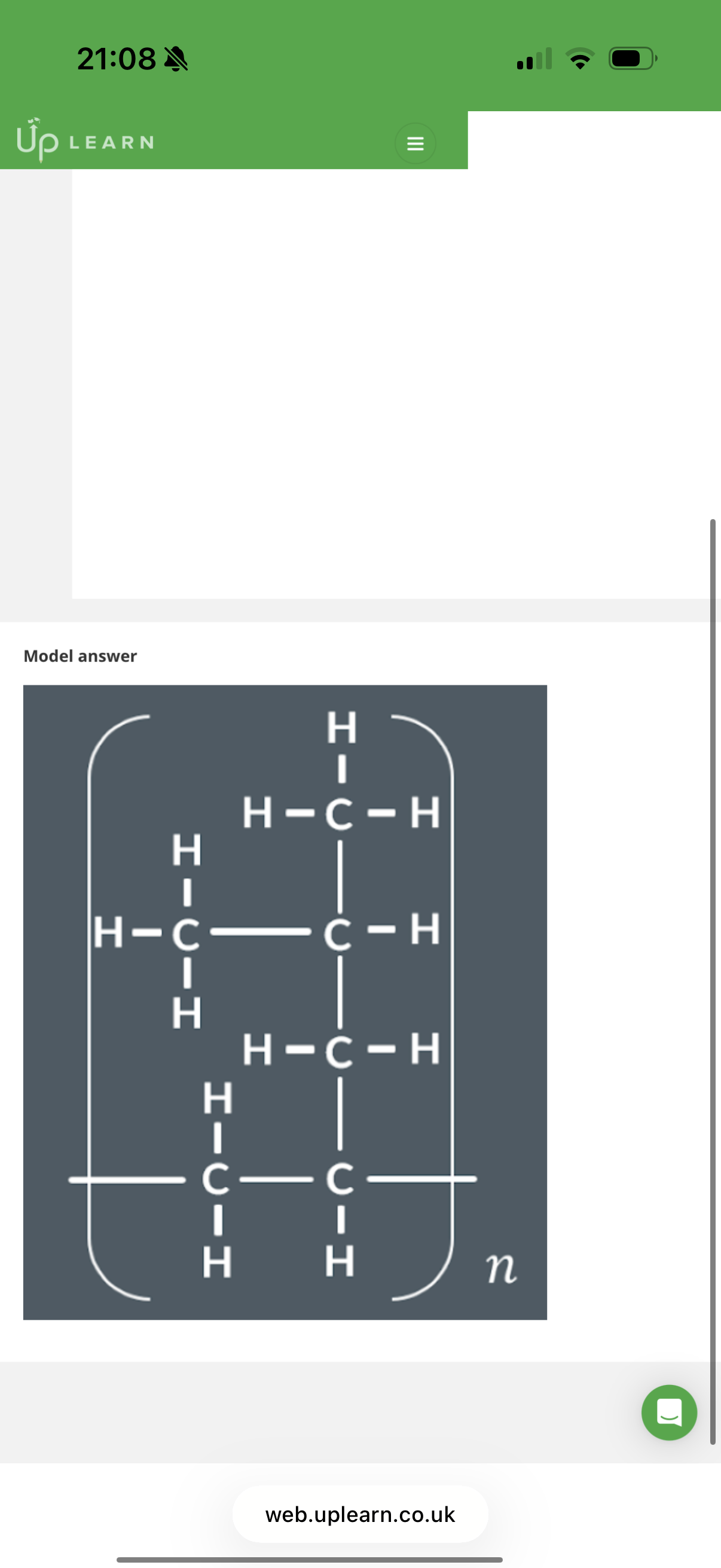
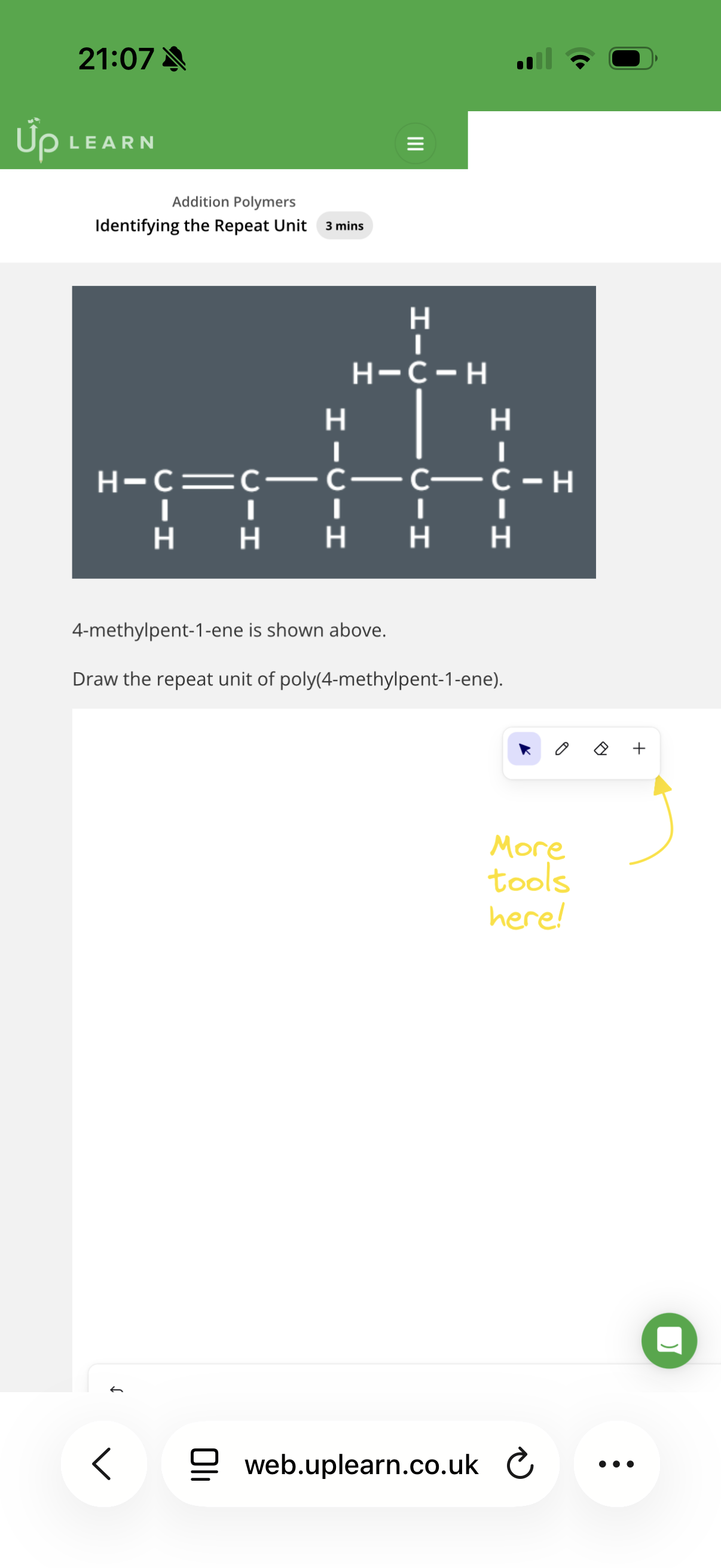
Answer image w?
Answer image q
note: Look at the carbons in pairs and check if they have the same substituents. Then remove the ends that attach to the other repeat units and form a double bond.
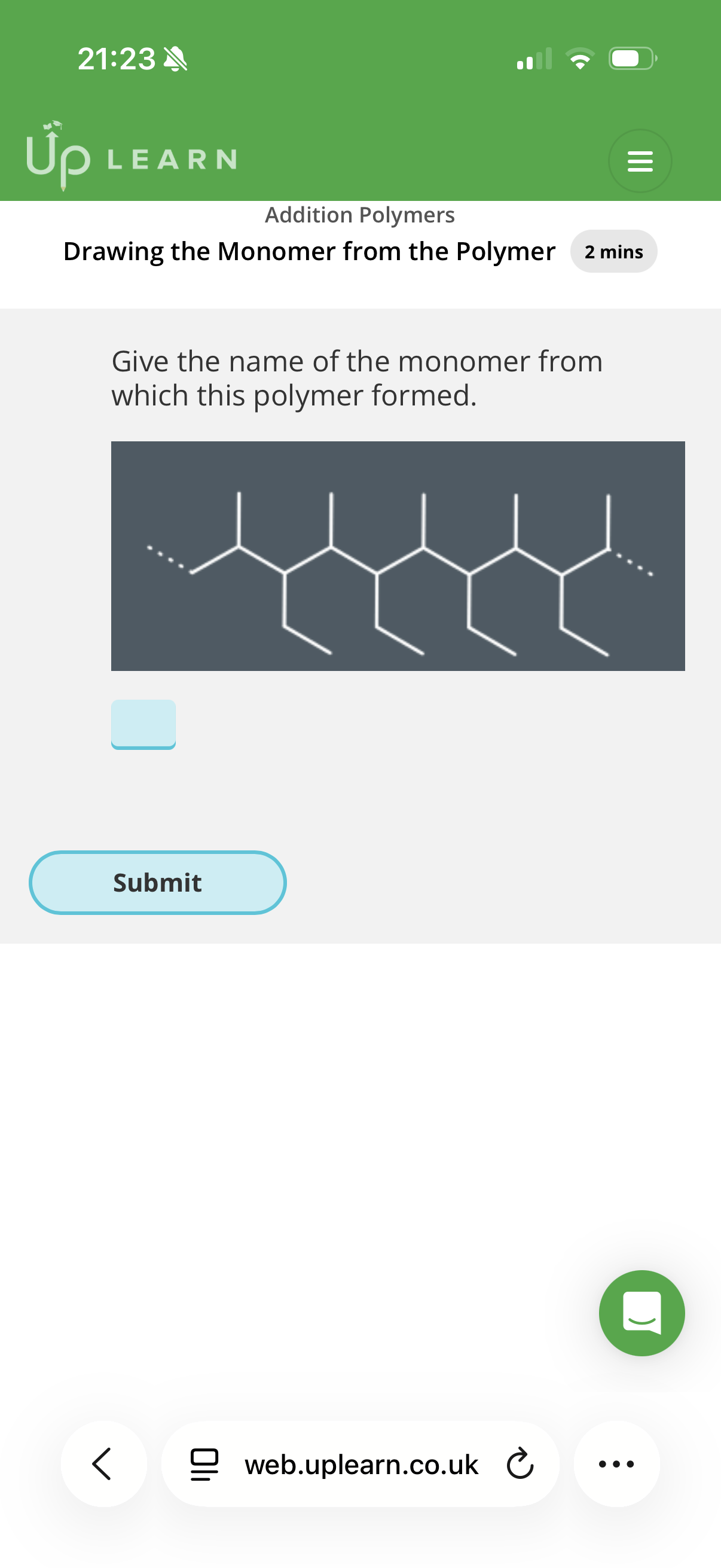
Answer image q?
E-pent-2-ene
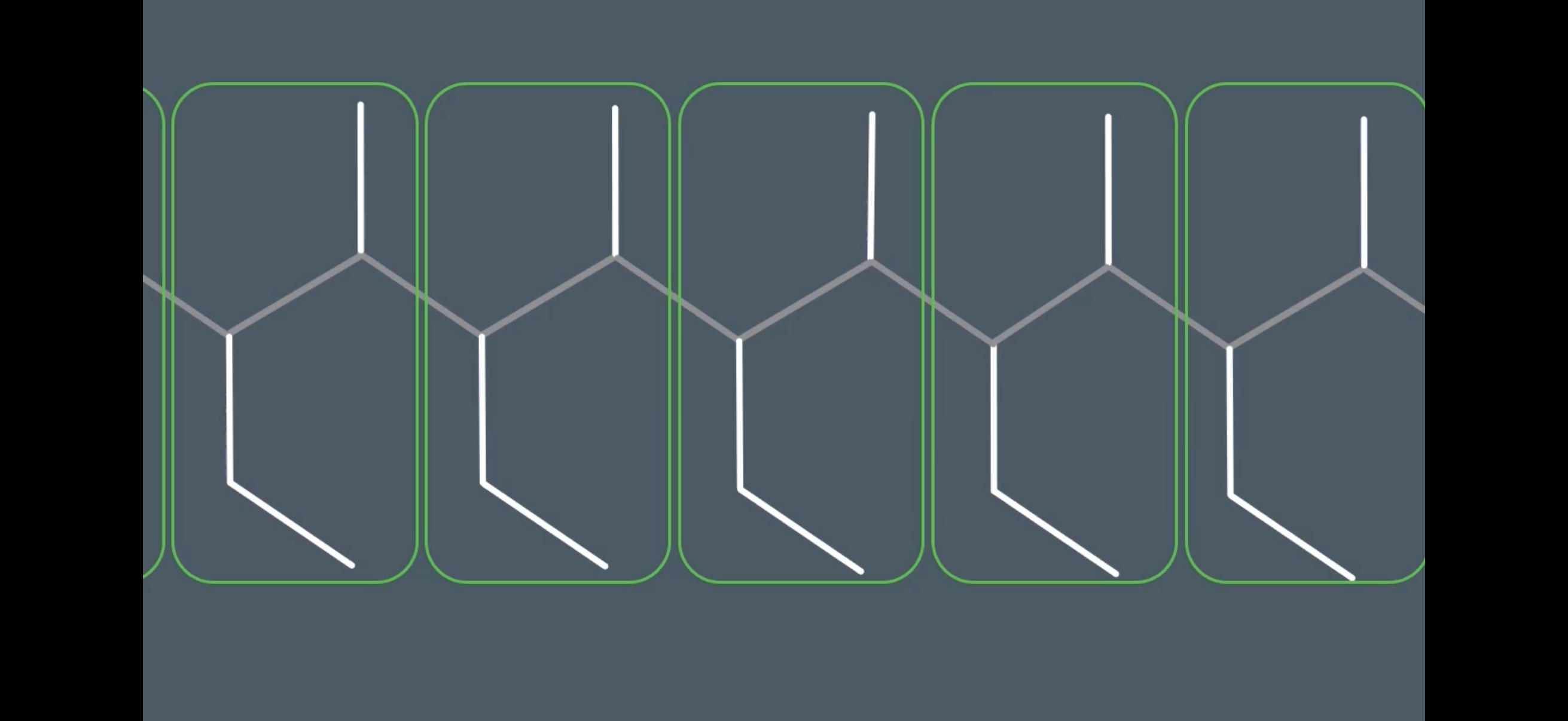
Answer image q?
Note: Some repeat units may have their substituents in different places(e.g. the methyl group in 1-methyl-eth-1-ene can be placed either up or down. This is why u look at the molecule in carbon pairs to identify if they have the same substituents as the other carbon pairs. This will help you identify the repeating unit.
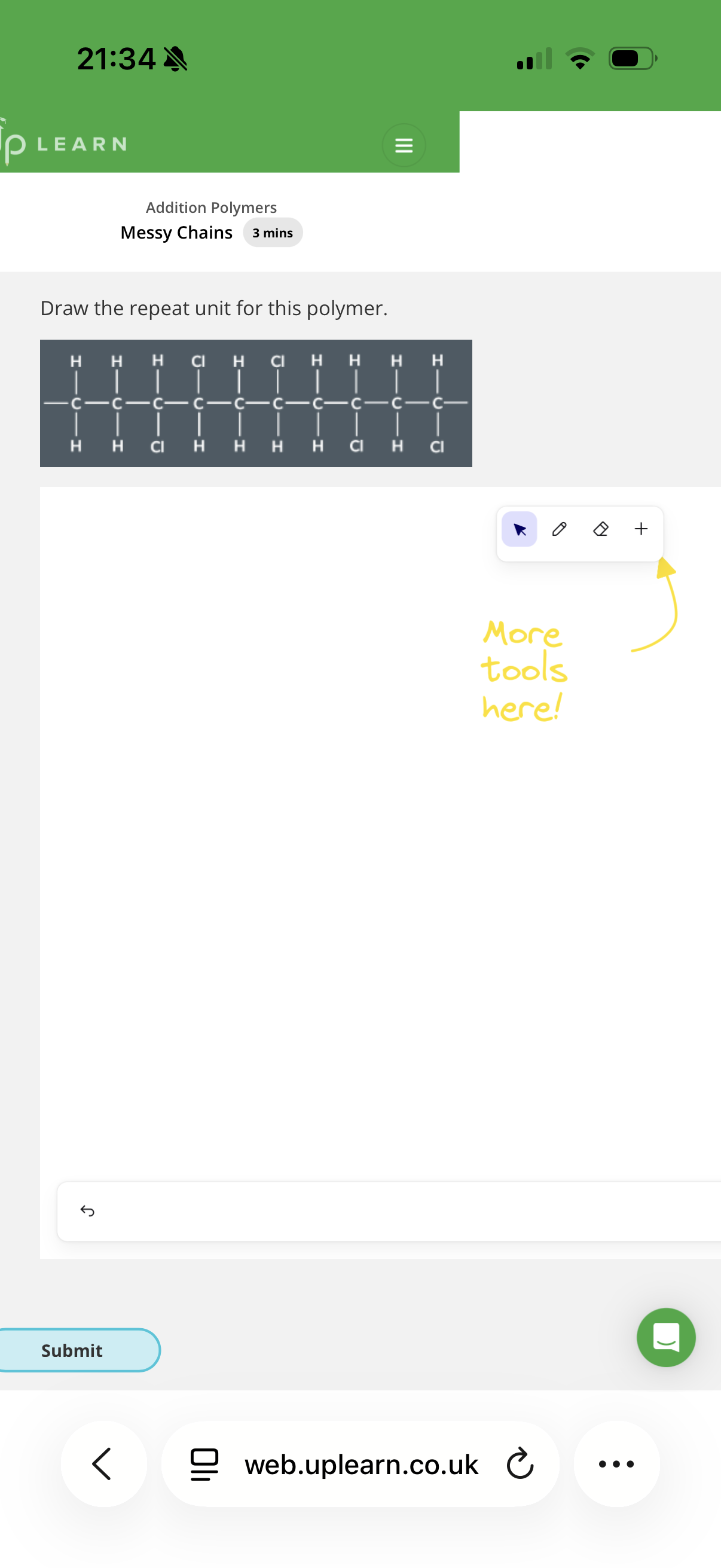
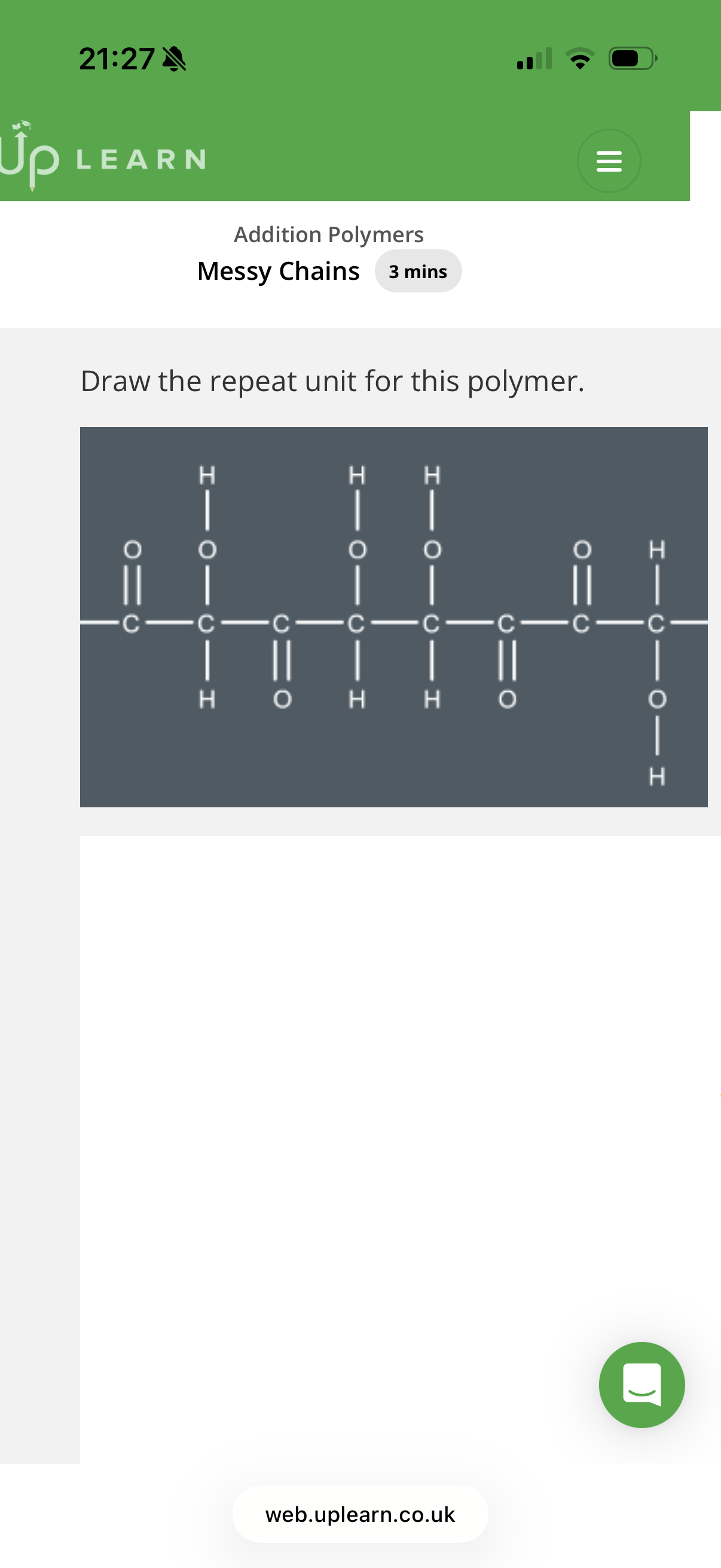
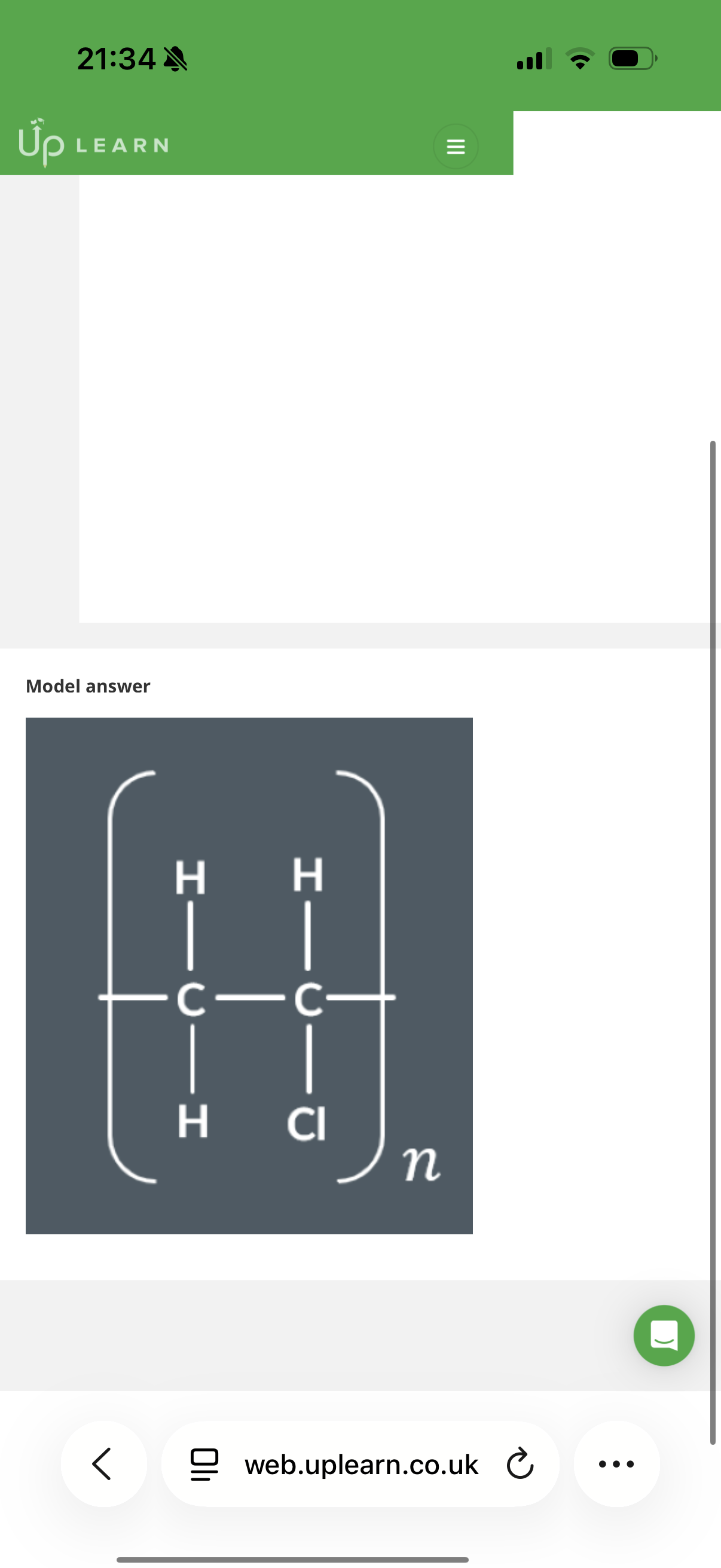
Answer image q?
Answer image q?
Note: monomers may be upside down or turned around when joining with other monomers to form polymers, hence why we get messy chains.
Note: Also bare in mind that it may be the second or third carbon that forms the repeat unit and sometimes we should ignore the first carbon.
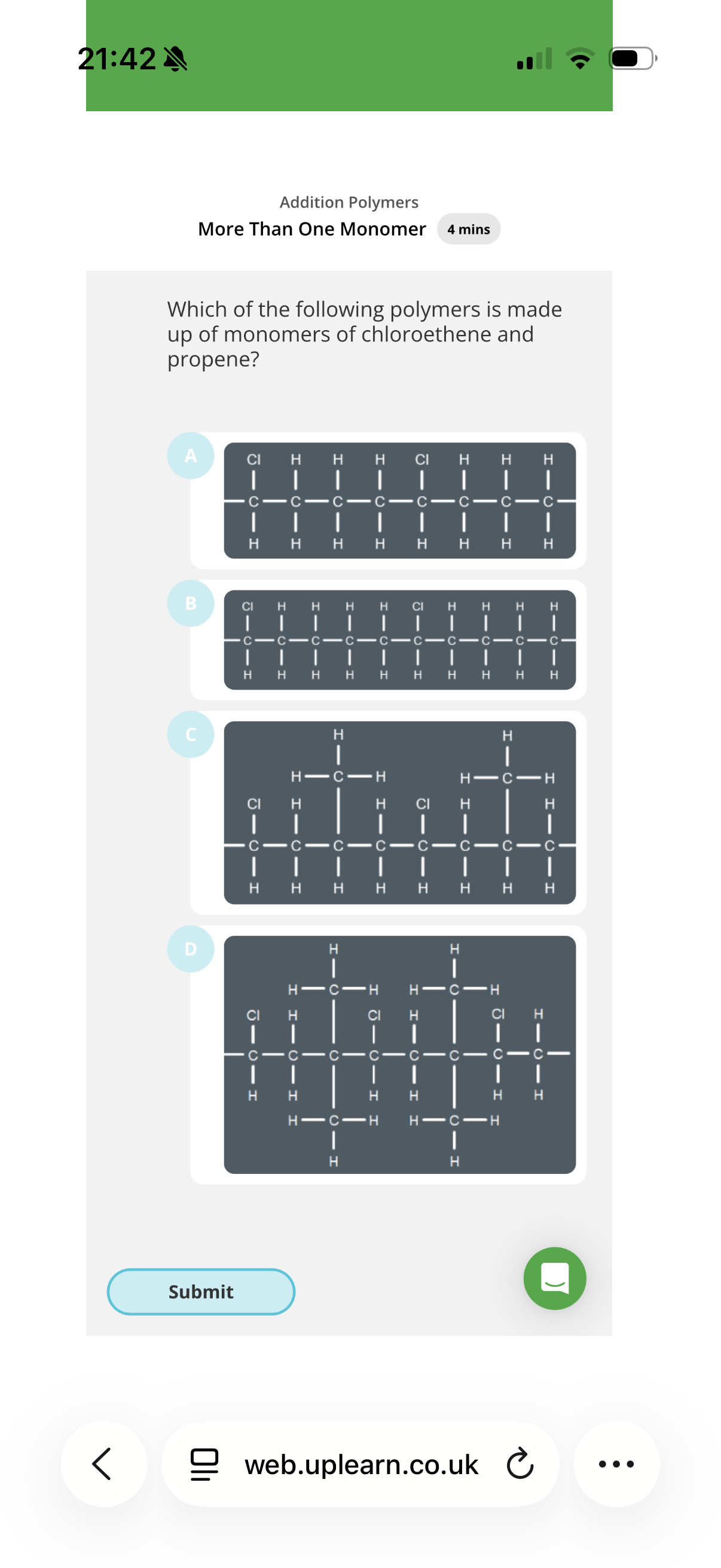
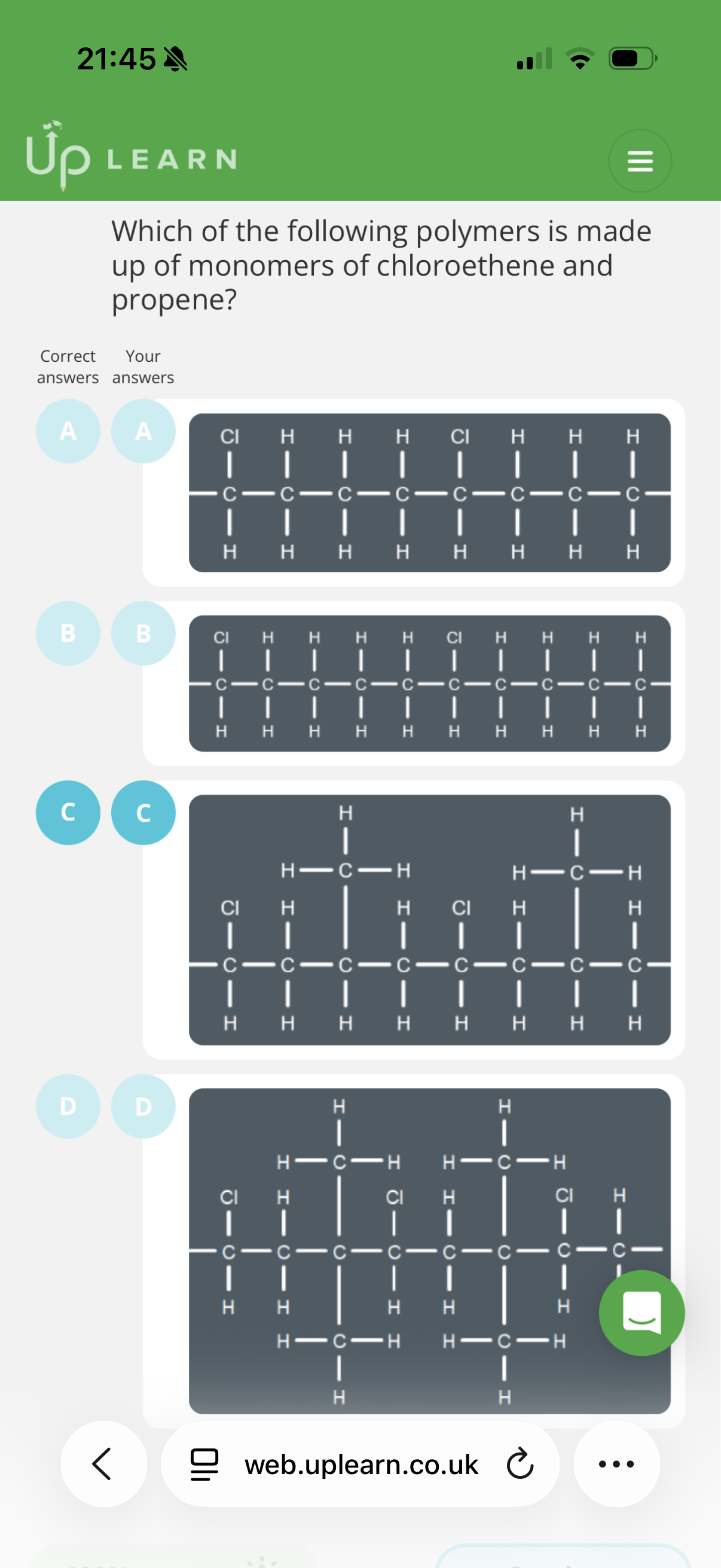
Answer image q?
note: the monomers that make up this polymer don’t always align in this simple manner e.g. the chlorethenes could be aligned next to eachother etc.
Hint: Split up the main polymer chain into pairs of carbons and check the substituents. Then see if each pair has the same substituents. If they don’t, see if the chain is made up of two monomers.
Explanation:
Chloroethene: When this forms an addition polymer, the double bond opens, leaving a two-carbon chain segment with one chlorine atom.
• Propene : When this forms an addition polymer, the double bond opens, leaving a two-carbon chain segment with a methyl group attached to one of the carbons:
We are looking for a polymer chain that contains both of these specific repeating units so the answer is C.
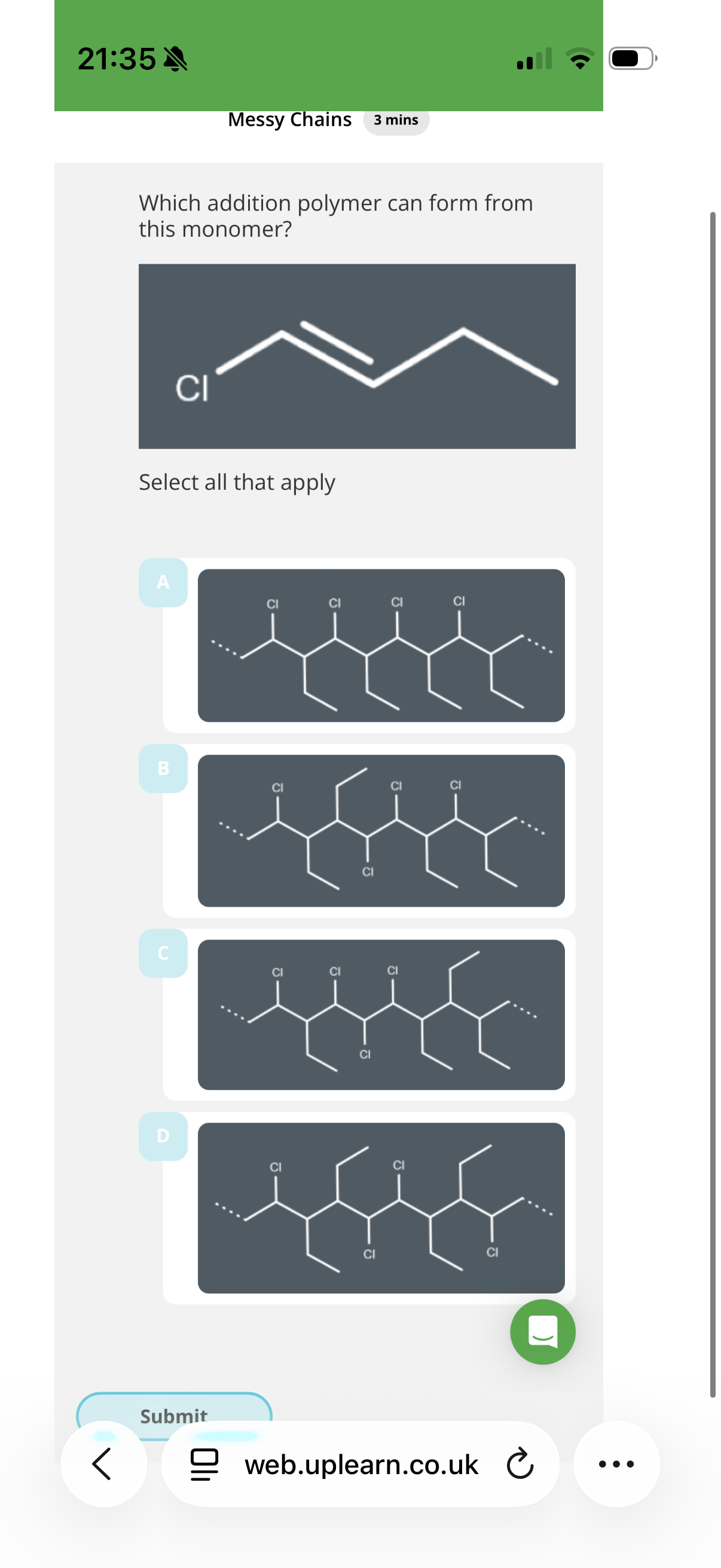
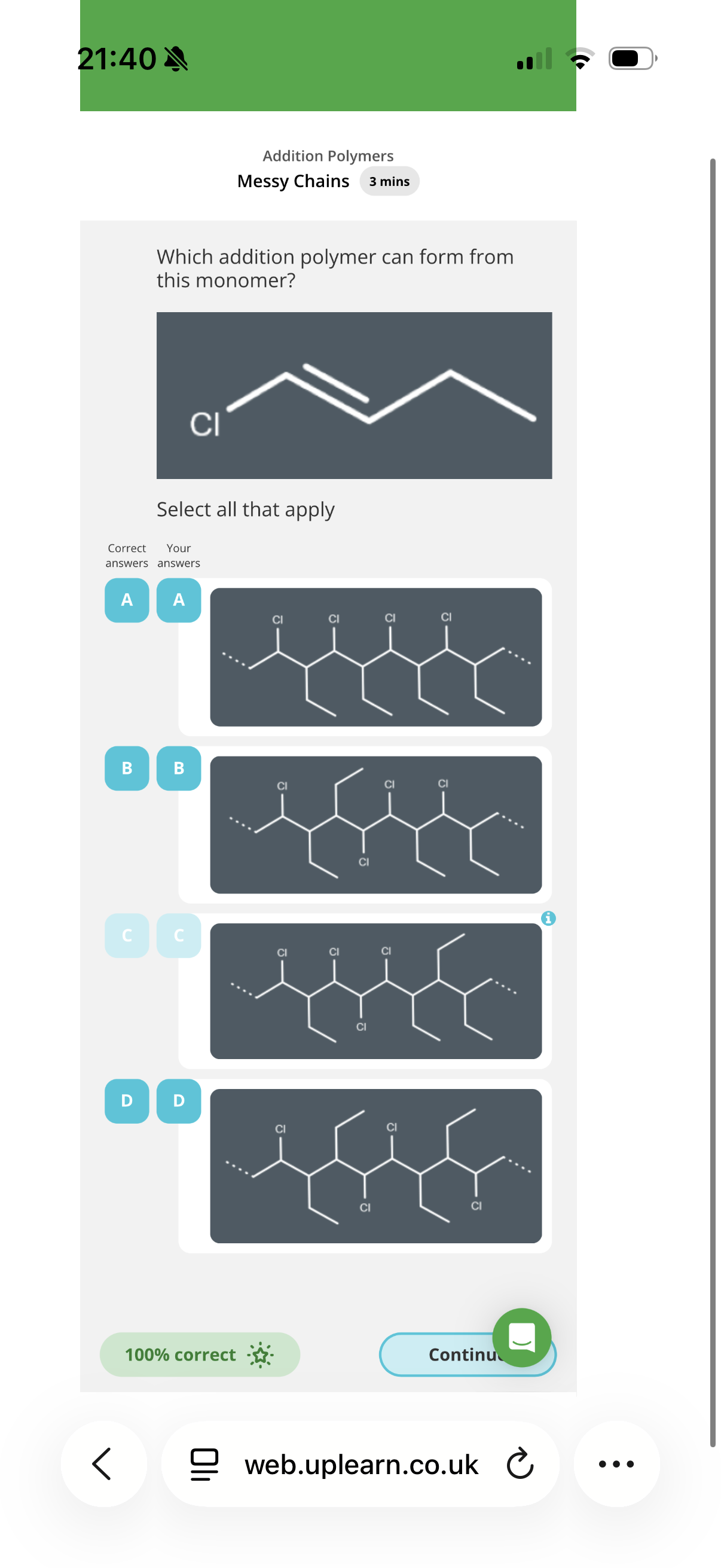
Answer image q?
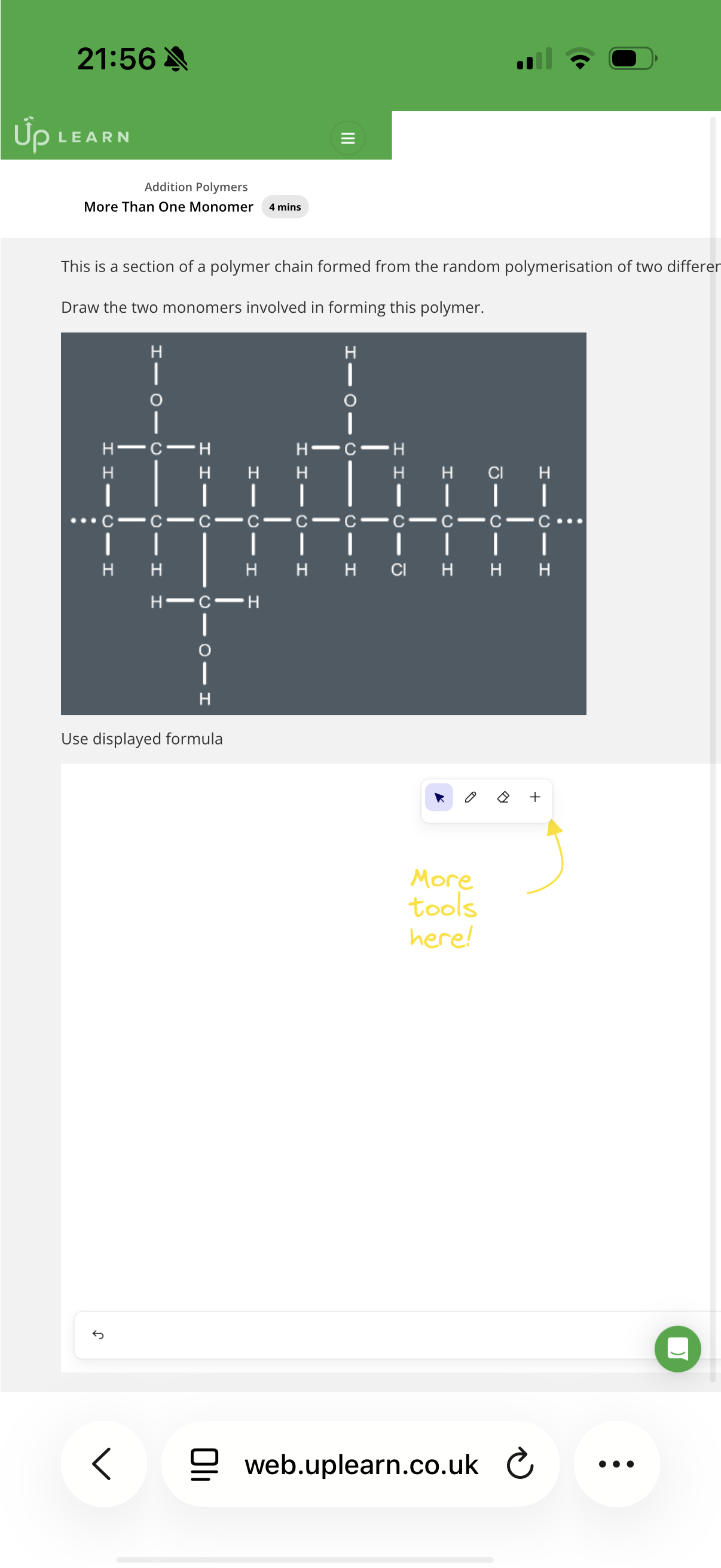
Answer image q?
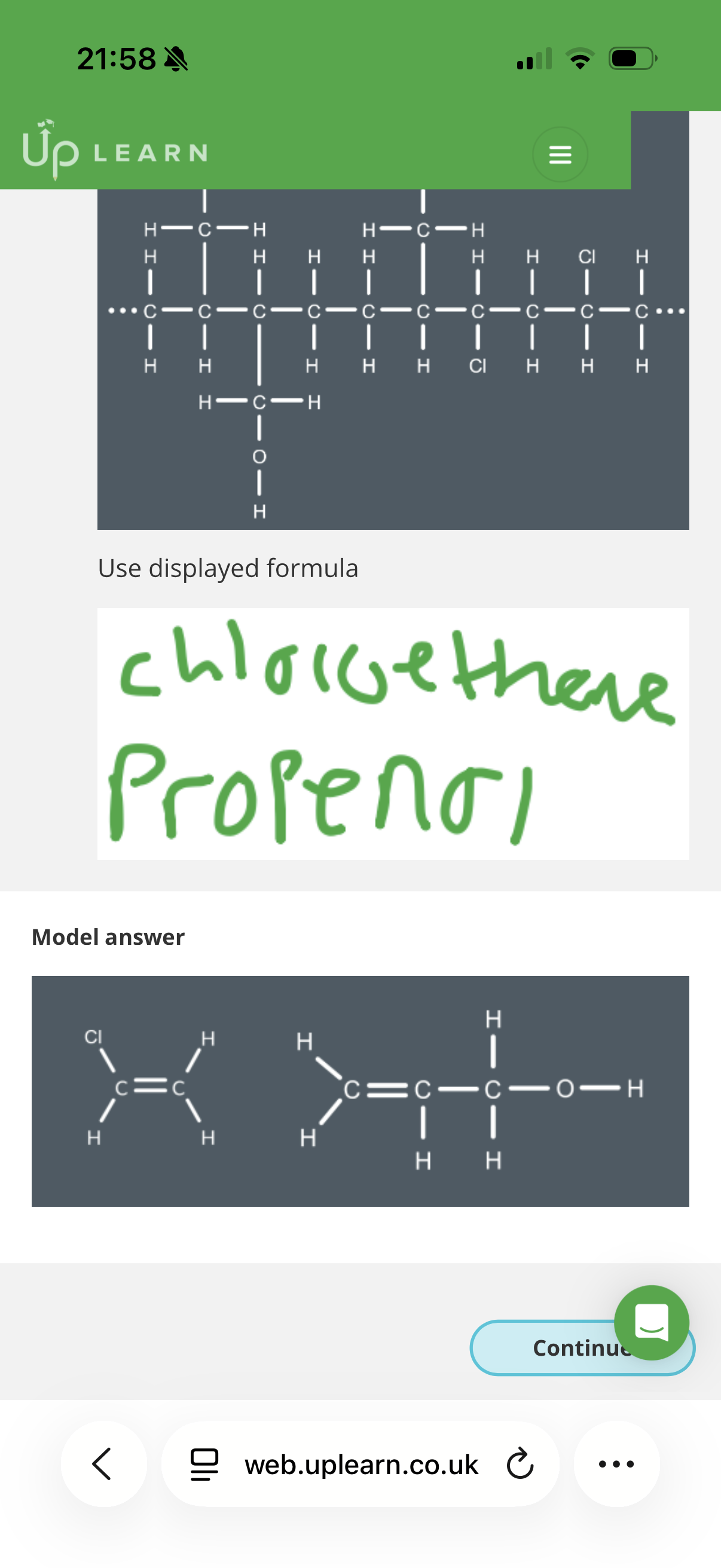
If a polymer was formed by one monomer with two double bonds, then the exam is likely to ask you to draw…
Select all that apply
A:the monomer
B:the repeat unit
If a polymer was formed by two monomers, then the exam is likely to ask you to draw…
Select all that apply
C:the monomers
D:the repeat unit
Answer image q?
B then C
A and D
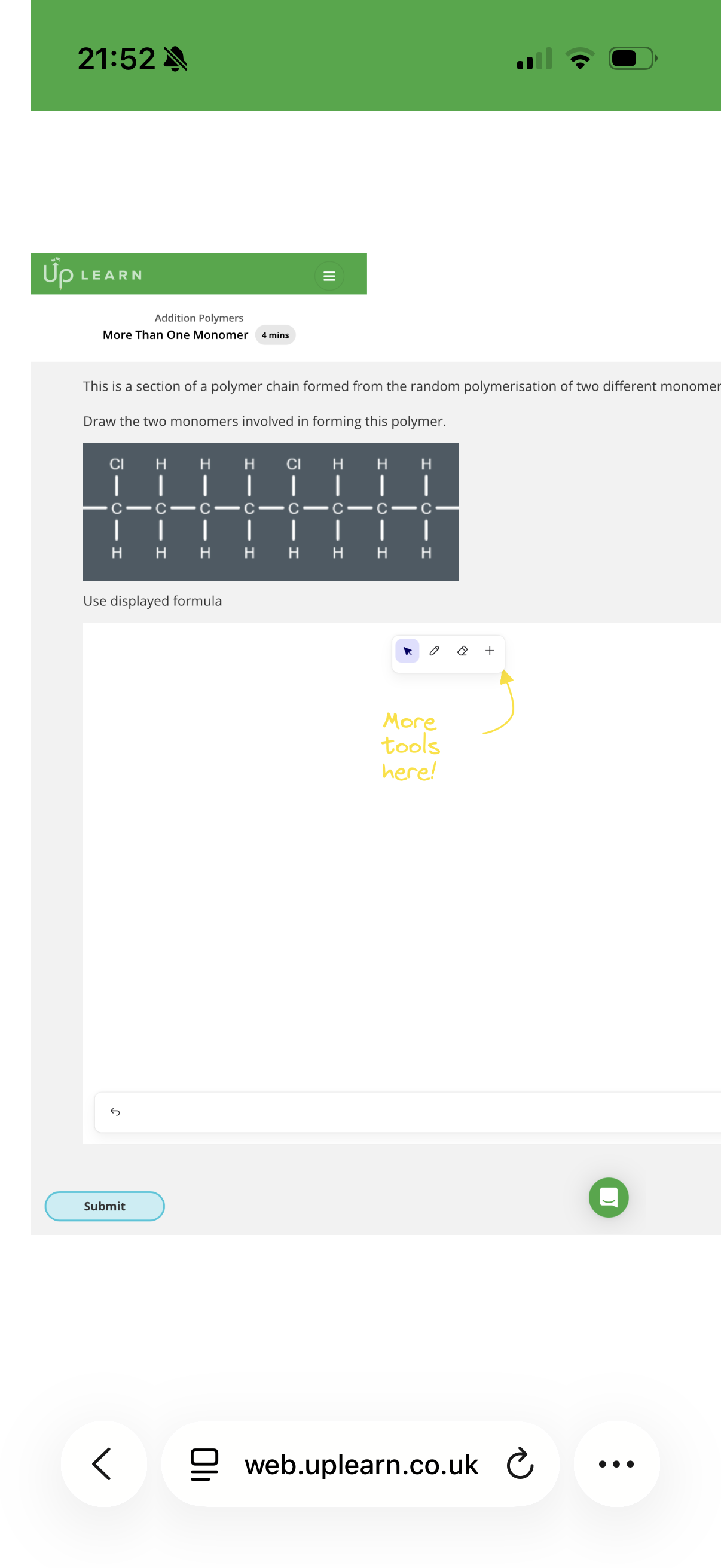
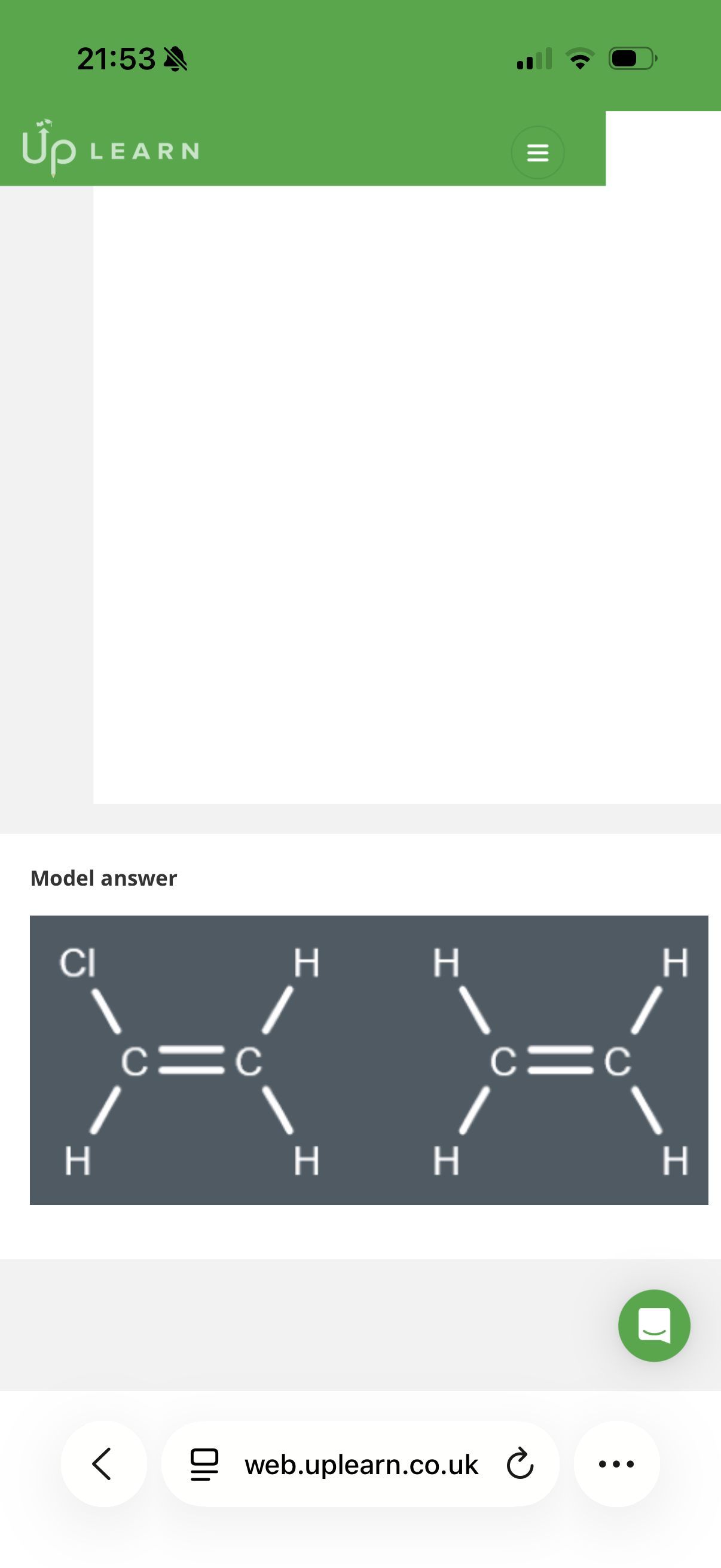
How to solve this:
To find the monomers, you need to break the polymer backbone into 2-carbon segments. Each 2-carbon segment represents where a double bond used to be.
1. Analyze the first segment (Left side)
Look at the first two carbons on the left of the polymer chain:
• Both carbons only have Hydrogen atoms attached.
• If you "put the double bond back," you get Ethene (CH_2=CH_2).
• This matches Option D.
2. Analyze the second segment (Middle)
Look at the next two carbons in the chain:
• The carbon on the left has two Hydrogens.
• The carbon on the right has one Hydrogen and one CH_2F group (a fluoromethyl group).
• If you "put the double bond back," you get a 3-carbon chain with a fluorine on the end.
• This matches Option A.

Polymers:
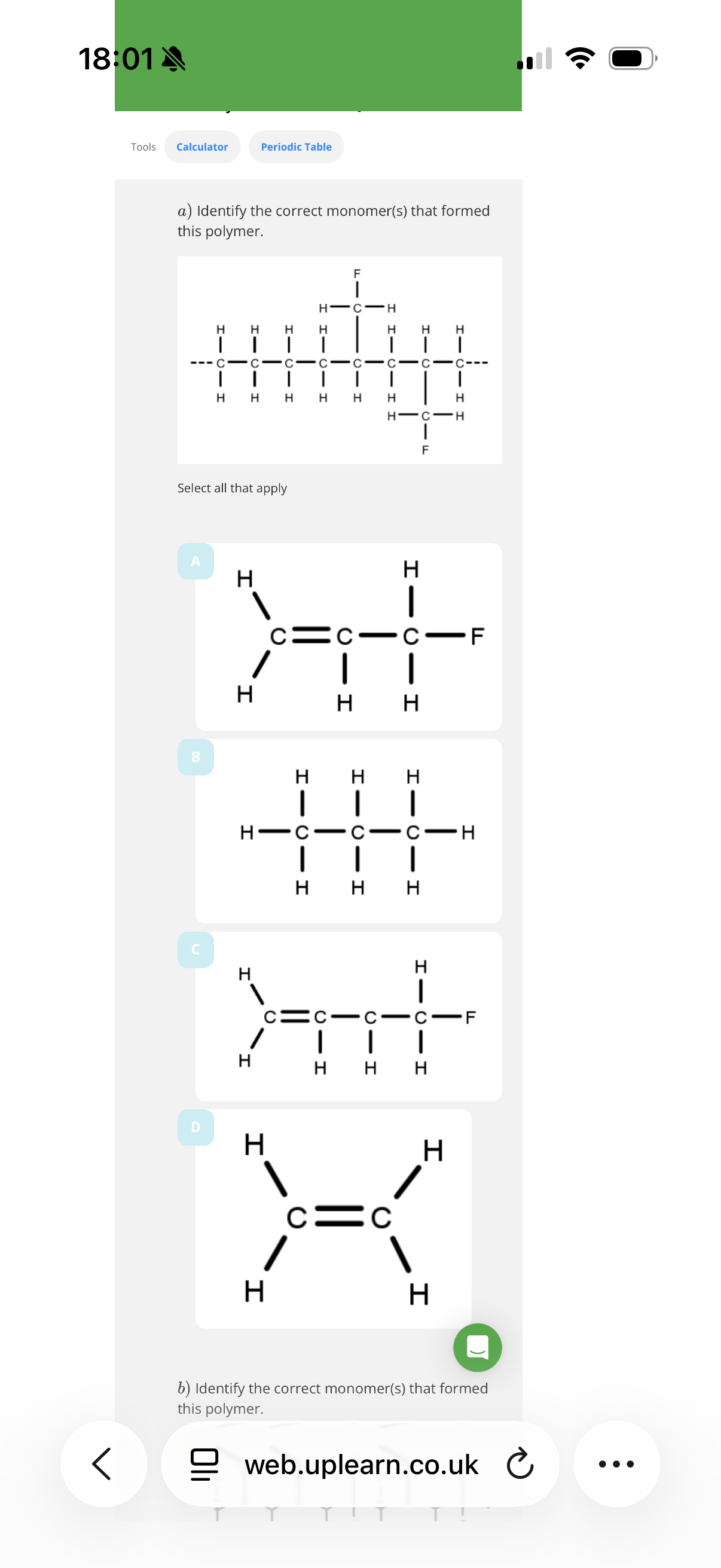
outline the addition polymerisation mechanisms for these alkenes in the image.


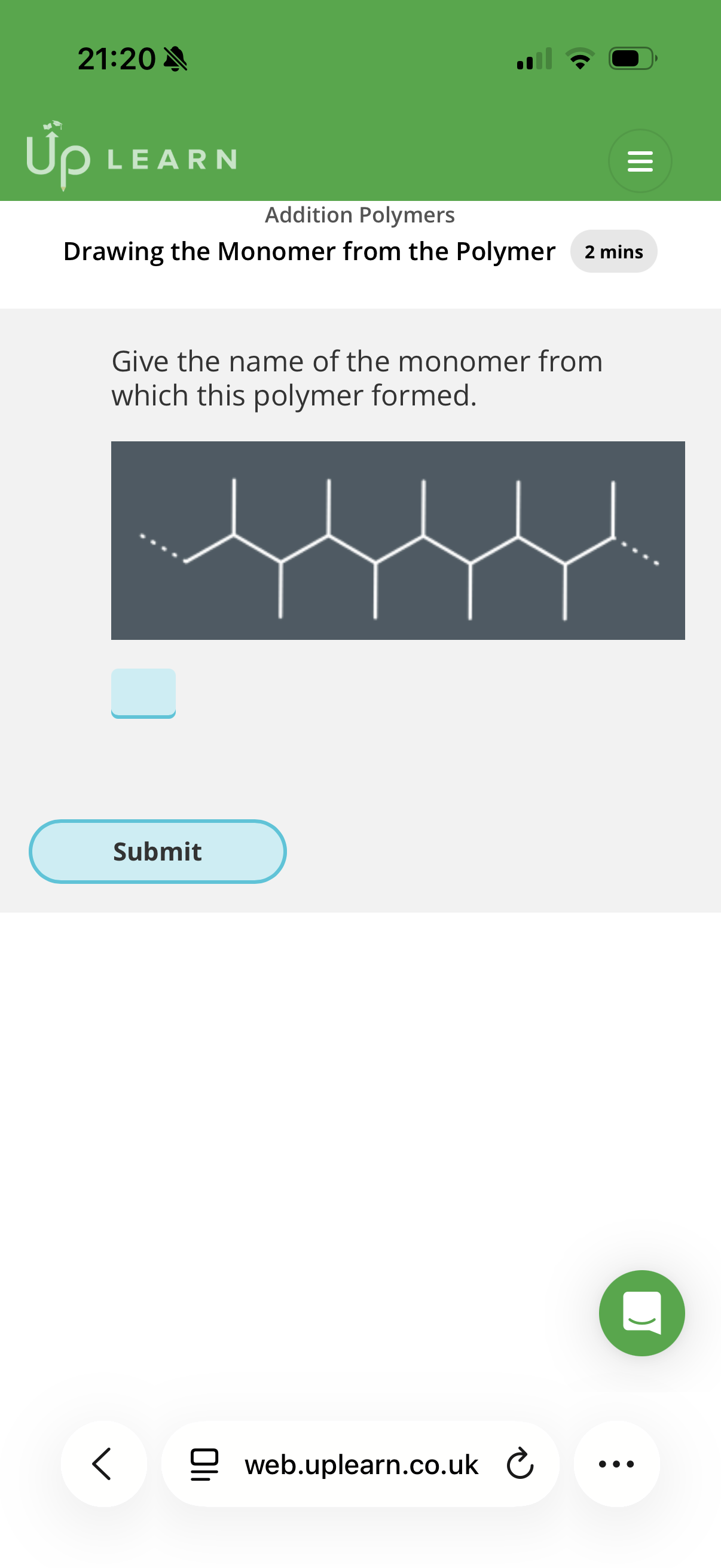
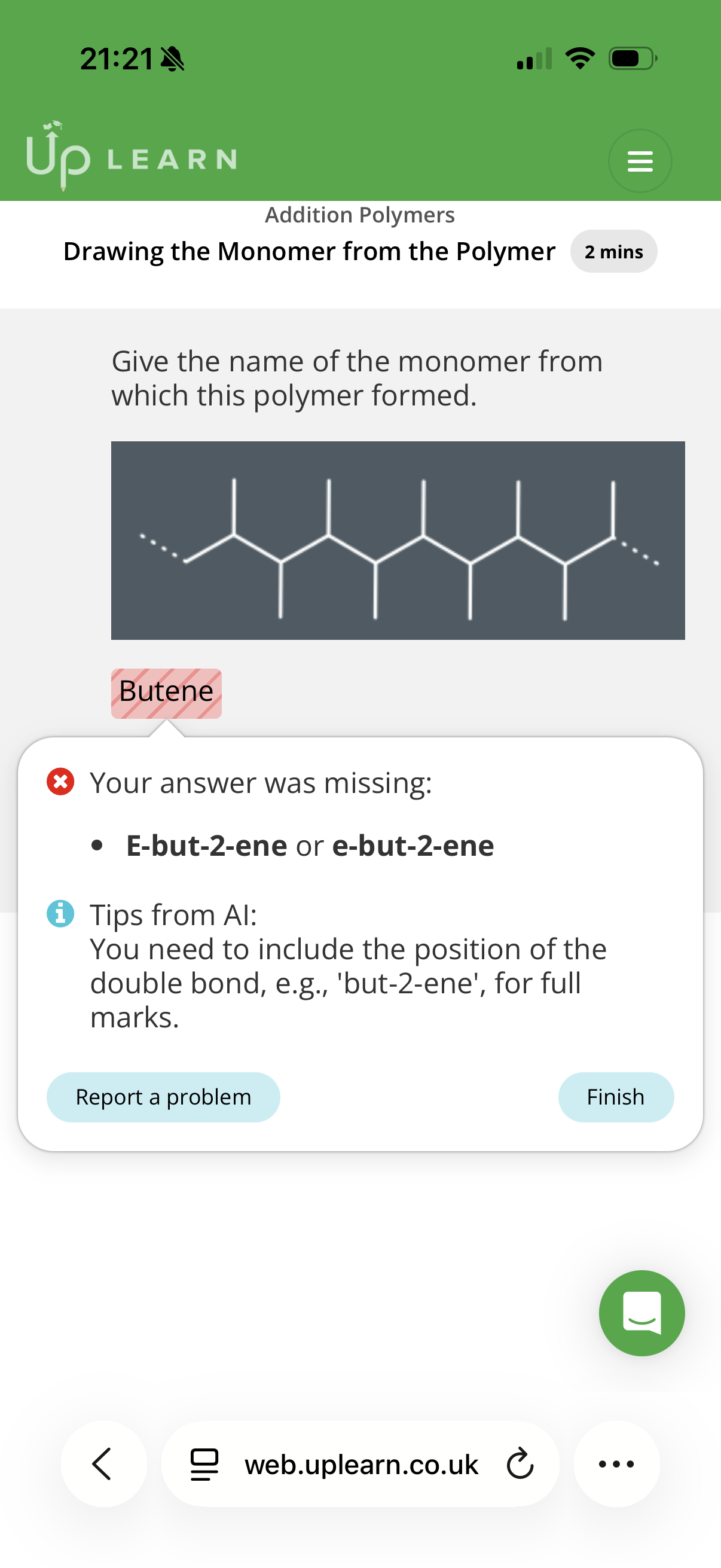
Identifying the monomer(s) used to make an addition polymer
Info: An addition polymers must have a backbone(chain of atoms) of …1? atoms and the monomer must contain at least…2? Carbons, so that there can be a C C …3? bond.
note: Where some of the carbon atoms have substituents, the monomer must have the …4? As well as a double bond.
Modifying the plastics
How can the properties of polymer materials be modified?
What are plasticisers?
Biodegradability
•Polyalkenes, in spite of their name, have a backbone which is a long chain saturated 7? molecule.
Why are polymers very Unreactive?(this is a useful property in many ways)
•Because polymers are Unreactive, they aren’t attacked by biological agents like …9? And so they aren’t …10? 11.Why is this a problem?
High and low density polythene
How is Low density poly(ethene) (polythene) made?
What is the product of the process used to make low density polythene?
• The branched chains(in low density polythene) don’t pack …14? particularly well and the product is quite …15?,stretches well and has a fairly low …16?
Give 3 examples of what these properties are used for ?
How is high density polythene made?
What is the result of the process for making high density polythene.
• (Answer to q19 extended) This makes the density of plastic …20? and its melting point …21?.
Name 3 uses for high density polythene?
The solutions to pollution by plastics
•To reduce the amount of plastic it can be reused or recycled
What is the steps to Mechanical recycling?(Simplest recycling form
What is the process to Feedstock recycling (Where they break down the chemical)
What is a thermoplastic polymer?
What is the problem with recycling?
The image on the other side contains info you need to know.
Carbon. 2. Two. 3.double. 4.substituent
By using additives such as plasticisers.
Small molecules that get between the polymer chains forcing them apart and allowing them to slide across each other.(making polymers flexible)
e.g. PVC is rigid enough for use as drainpipes,but with the addition of a plasticiser it becomes flexible enough for making aprons.
Alkane
8.They have strong non-polar C-C and C-H bonds.
Enzymes. 10. Biodegradable
Waste disposal is becoming more difficult and more landfill sites are getting filled
Its made by polymerising ethene at high pressure and temperature via a free radical mechanism.
A polymer with a certain amount of chain branching(a consequence of the random nature of free radical reactions-uncontrollable chain reactions that can occur at any point)
Together
Flexible . 16.density.
packaging(plastic bags), sheeting and insulation for electric cables
At temperatures and pressures a little greater than room conditions and uses a Ziegler-Natta catalyst.
A polymer with much less chain branching and the chains can pack together well.
Greater. 21. Higher
Milk crates, buckets and bottles (low density polythene is insufficiently rigid(cannot bend)
1) Seperate the different types of plastic.
2)The plastics are then washed and once they are sorted they may be grounded up into small pellets.
3) These can then be melted and remoulded (e.g. recycled soft drink bottles made from PET are used to make fleece clothes)
The plastics are heated to a temperature that will break the polymer bonds and produce monomers. (These can then be used to make new plastics)
It will soften when heated so it can be melted and re-used.
Polymers can only be recycled a limited number of times because each time it’s heated, some of the chains break and become shorter which degrades the plastic’s properties.


Addition polymers:
Answer image questions?


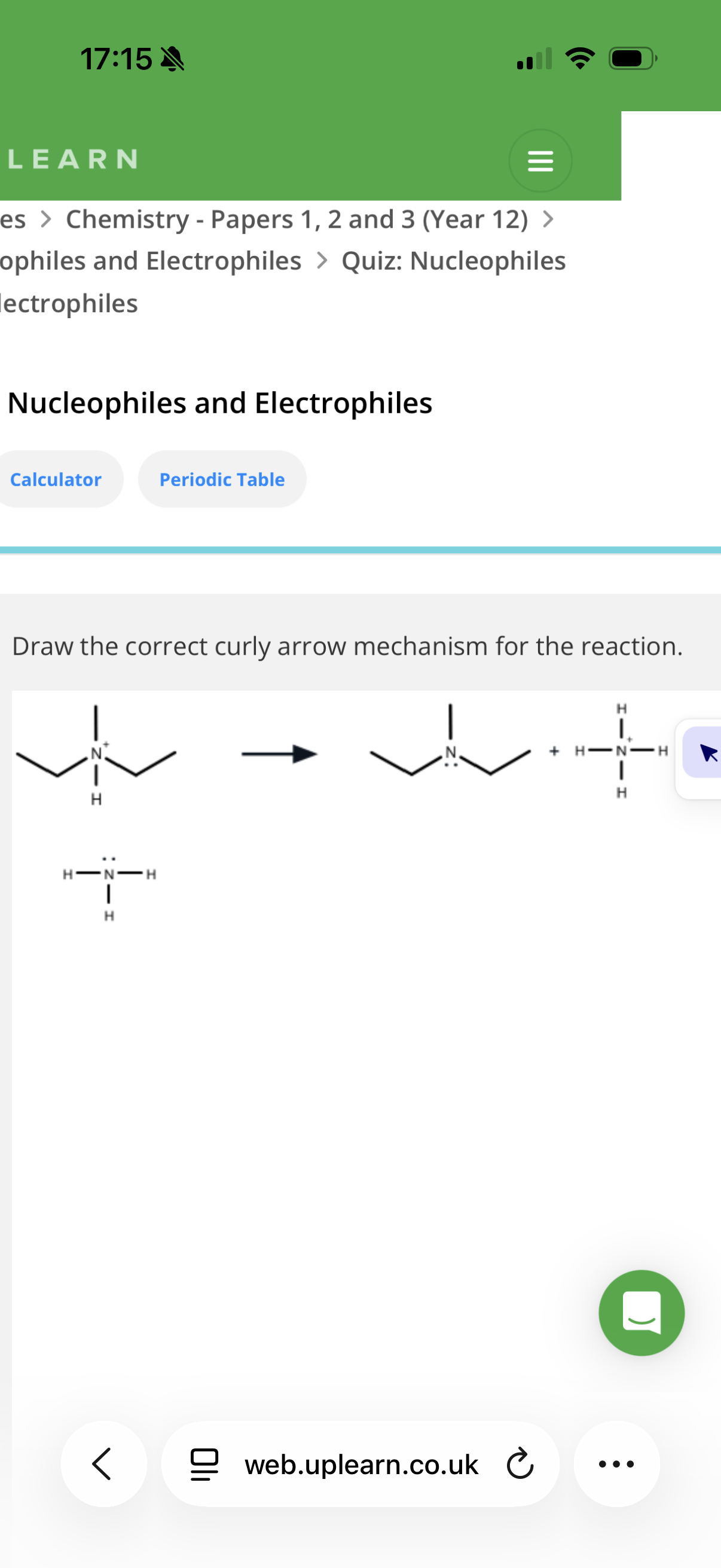
Nucleophillic substitution:
What are to conditions to react a halogenoalkane to form an alcohol?(red line)
What are the conditions to react a halogenoalkane to form a Nitrile?
What are the conditions to react a halogenoalkane to form an Amine?
The reaction to make a nitrile must be in the solvent of ethanol. This is called…
What is a substitution reaction?
Explain why water can’t be used instead of ethanolic when you warm the halogenoalkane with KCN to form a Nitrile?
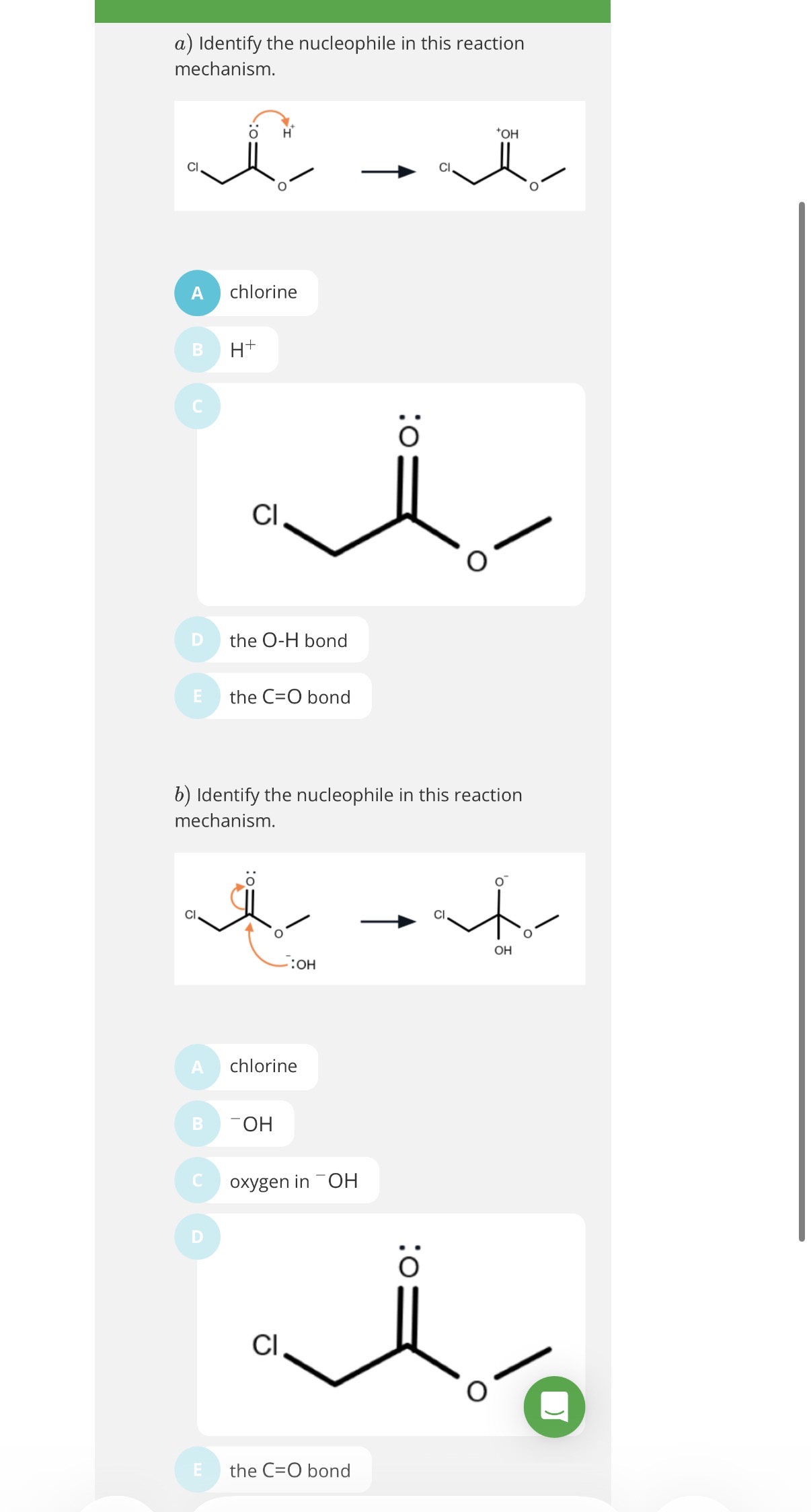
info- image shows an example of a Nucleophillic substitution reaction of Chloromethane with KOH(aq) to form an alcohol.
Note: image shows an example of a substitution reaction(note halogens always form polar bonds with C)
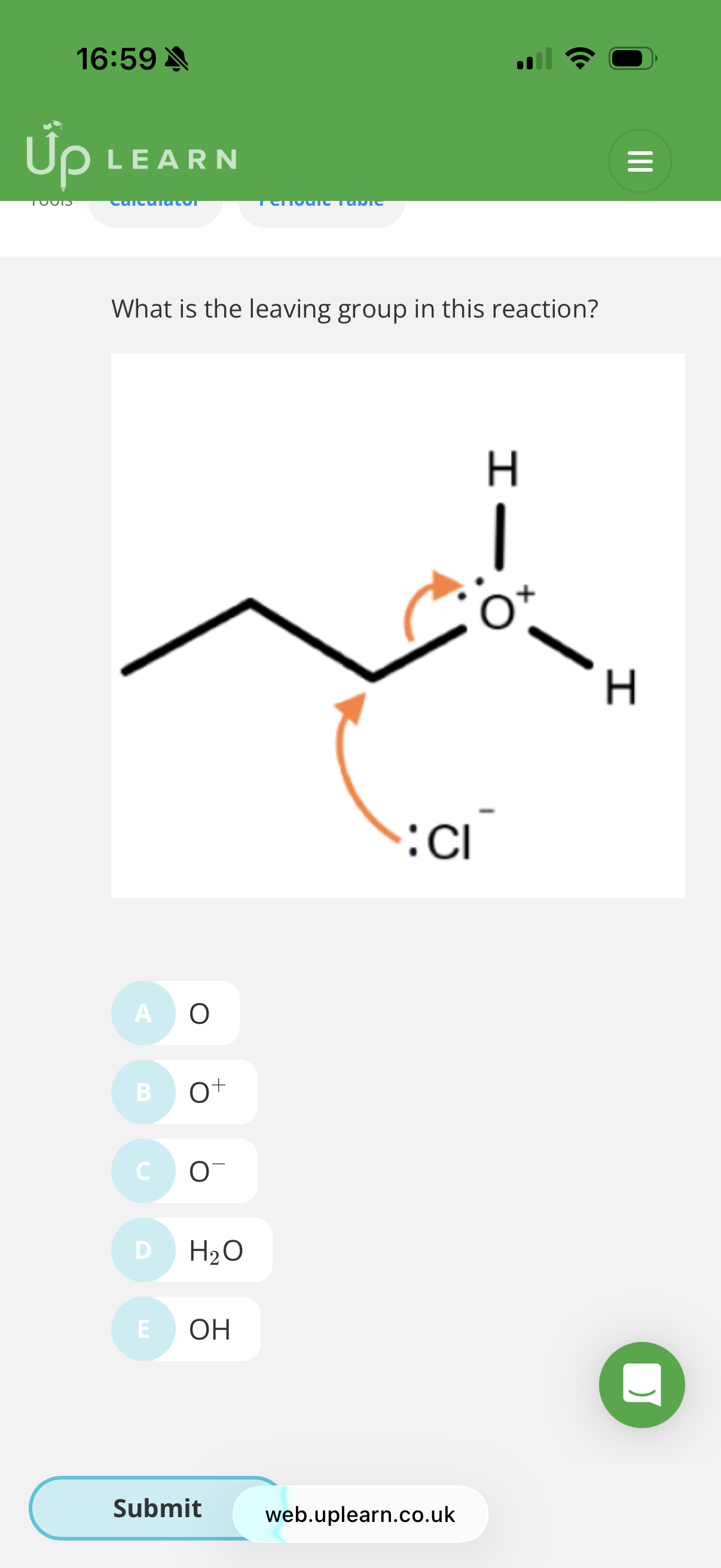
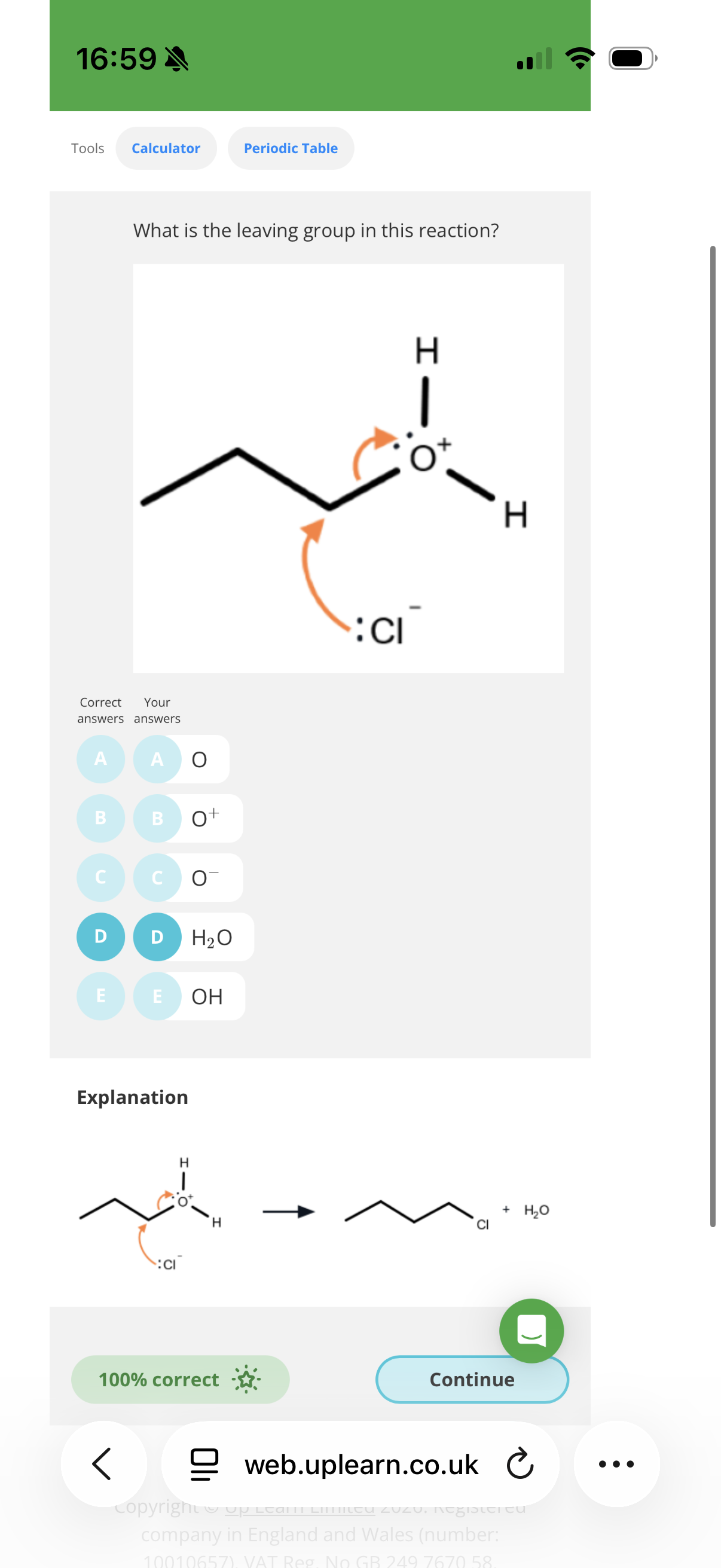
image explanation- The nucleophile (OH-) transfer its lone pair of electrons to the slightly positive carbon.Therefore two electron will have to be pushed out and so the slightly negative Cl gets the shared pair of electrons and breaks off from the molecule
KOH(aq) (or any aqueous hydroxide)
Warm with KCN in aqueous ethanol(ethanolic)
Warm with excess ethanolic ammonia
Ethanolic
A reaction where 1 molecule is replaced another molecule.
Water can dissociate into H+ and OH- ions and since there would be some OH-, some alcohol product would also be made.


Name and Outline the mechanism when Chloromethane reacts with warm KCN in aq ethanol?
Name and outline the mechanism when 3-Chloro-prop-1-ene reacts with KOH (aq)? → Answer is in this image
Name: Nucleophilic substitution(mechanism is in the image)
(You just formed Ethanenitrile)


•Nitriles have a …1? Bond between C and N.
Why are Nitriles important?
The image shows you an example of
note- amine classification:
Primary (RNH2): One hydrocarbon group attached to nitrogen.
Secondary (R2): Two hydrocarbon groups attached to nitrogen.
Tertiary (R3N): Three hydrocarbon groups attached.
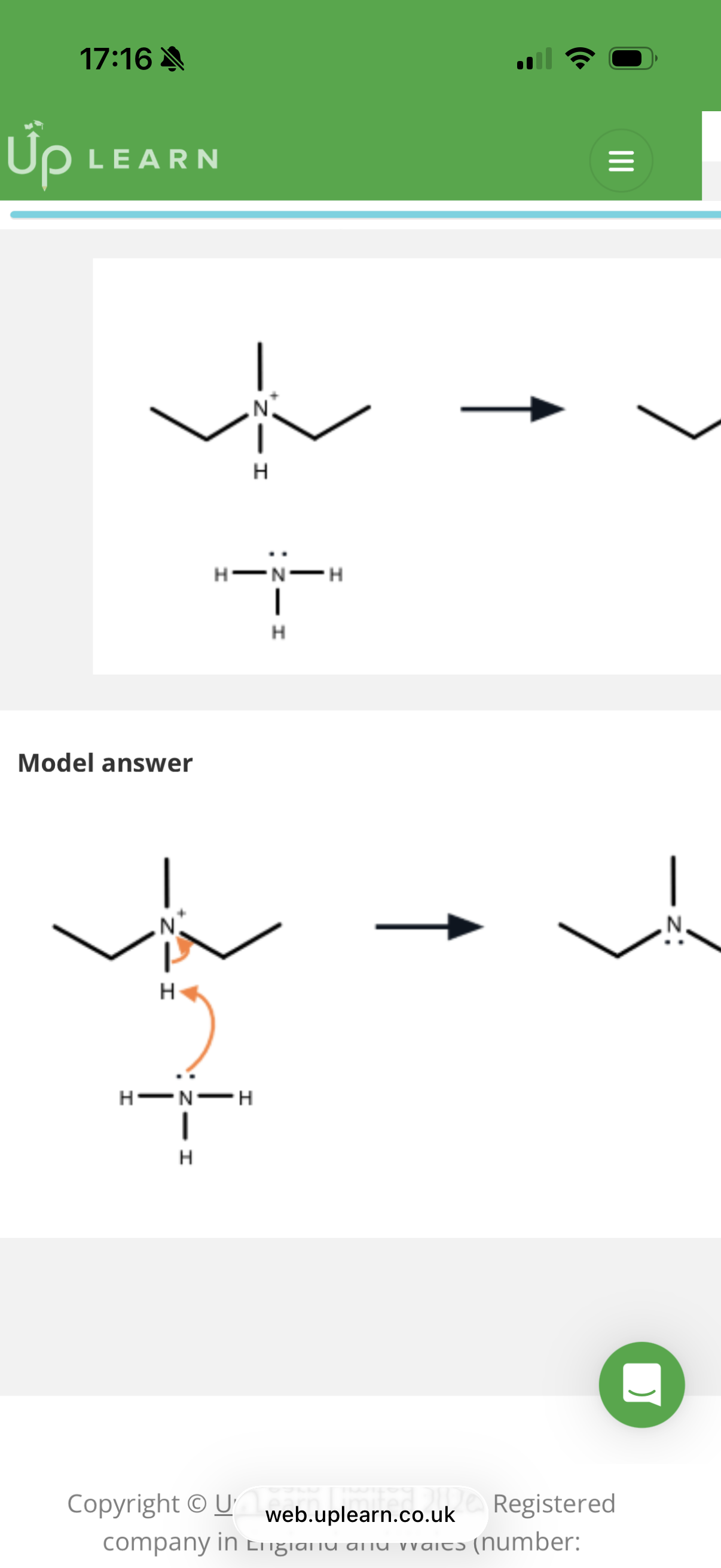
The image on the other side shows the reaction when a halogenoalkane reacts with excess ammonia(NH3) to form an amine.
Triple
They make the carbon chain longer
Note- the excess ammonia will always be incorporated in the intermediate step.
note- in the intermediate step, Nitrogen has 1 too many bonds and so will gain the shared pair of electrons which will break an N-H bond.
Note- All products broken off from the molecule will join together to form one molecule which u need to show by adding + then the molecule.

Draw the reaction between a Iodoethane and excess ammonia and name the mechanism
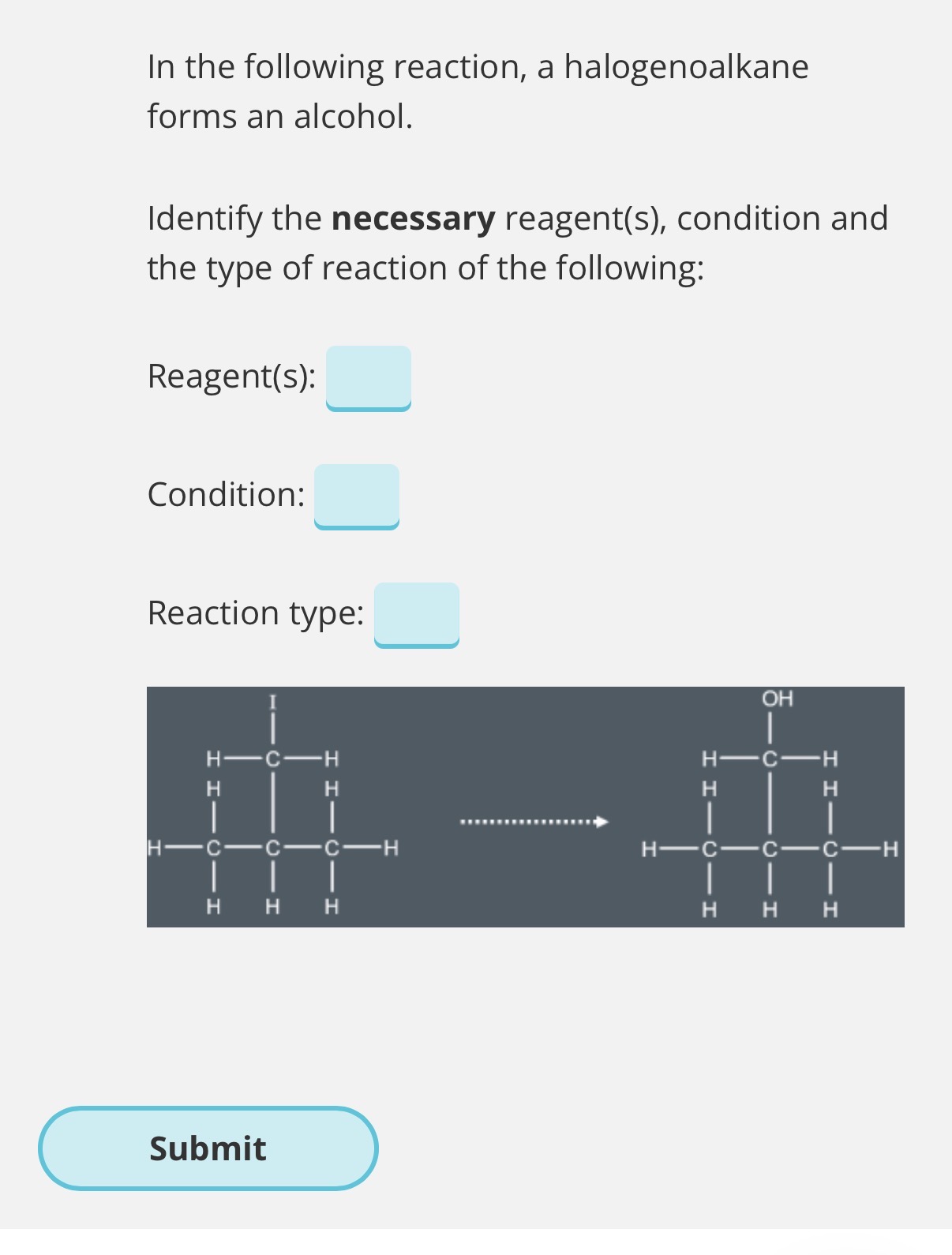
Answer this image question?
Name: Nucleophillic substitution (the Nucleophile is always substituted in)
Reagent: KOH. Condition: aqueous. Reaction type: Nucleophilic substitution
(Explanation
In this reaction, an iodine atom is substituted by an OHgroup. Therefore, this is a nucleophilic substitution reaction.
For a halogenoalkane to undergo nucleophilic substitution, they must react with an aqueoushydroxide.

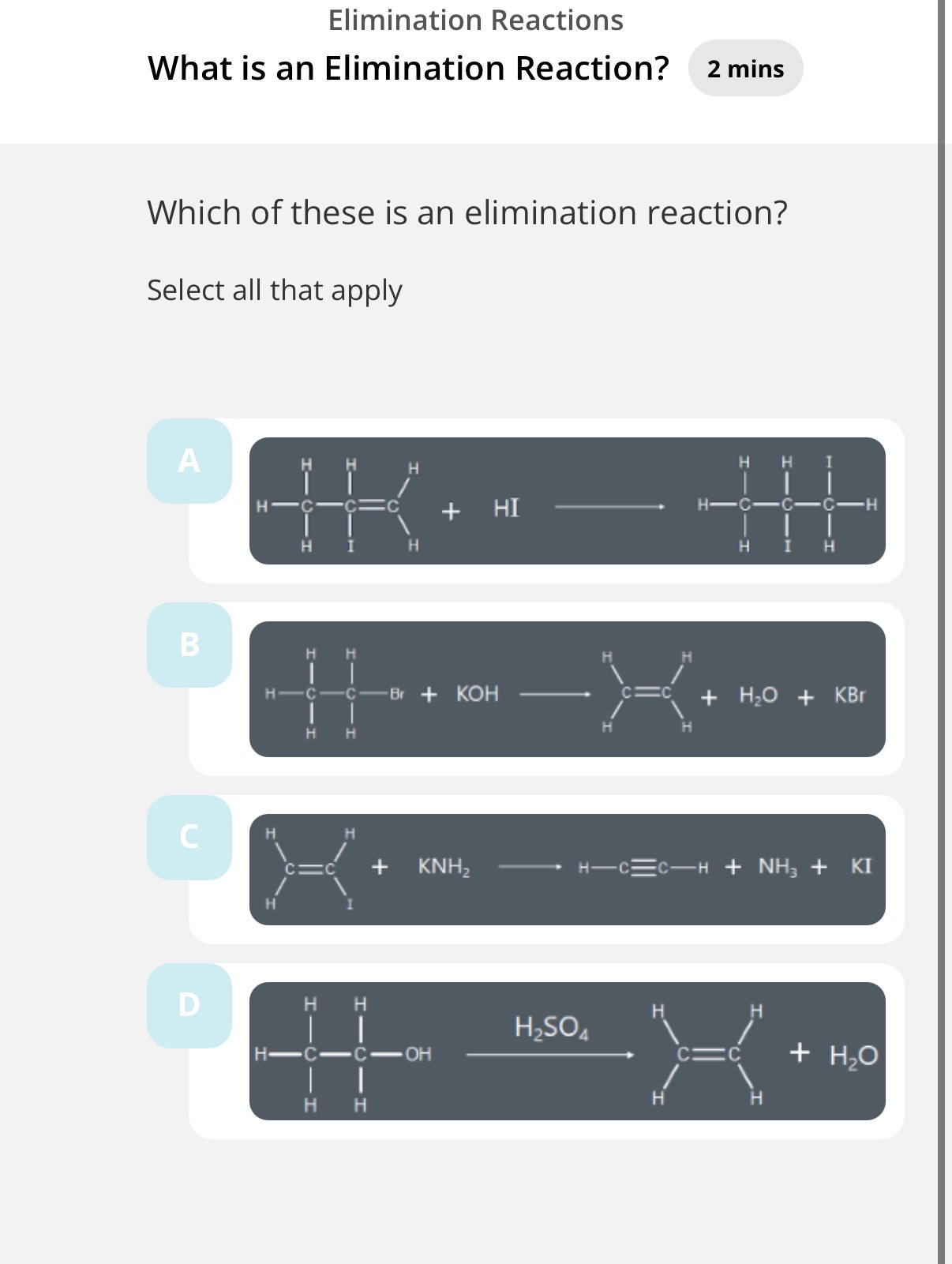
Elimination reactions:
What are the conditions to react a halogenoalkane to form an alkene?(green line)
What are the 2 conditions to react an alcohol to form an alkene? (Green line)
What is an elimination reaction?
4 answer image question?
Ethanolic KOH under reflux
Concentrated H3PO4 or
H2SO4 under reflux
1)where 2 sigma bonds break
2) a pi bond forms
B,C,D

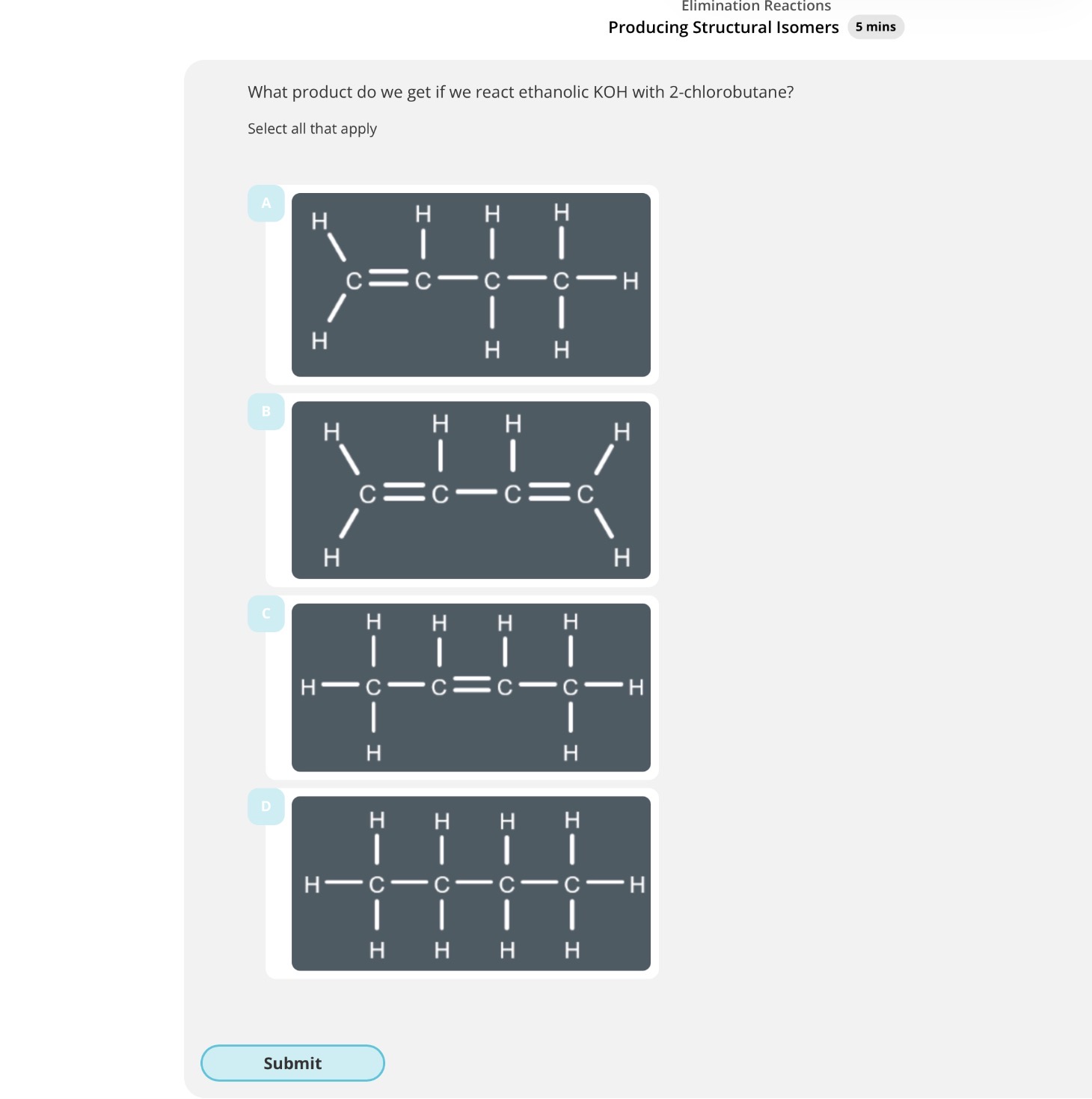
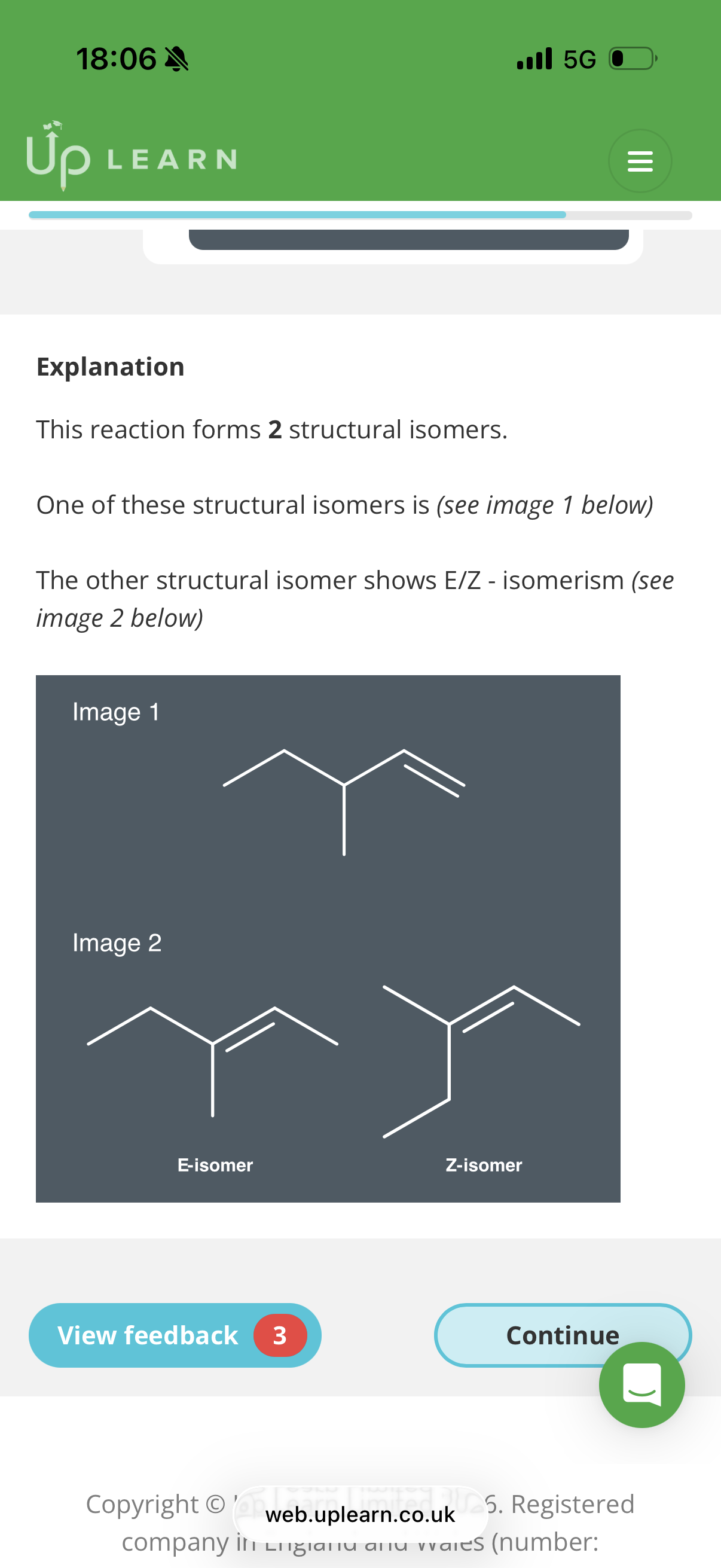
Elimination structural isomers and E-Z isomerism
What product do we get if we react ethanolic KOH with 2-chloropropane?
Answer image q?
But-1-ene and but-2-ene are examples of...?
In an elimination reaction, up to …4? structural isomers can be produced.(depends on which hydrogen is removed during the reaction)
Alkenes have restricted rotation around their double bond. This means that if an alkene has two substituents, it will have
…5? isomers.
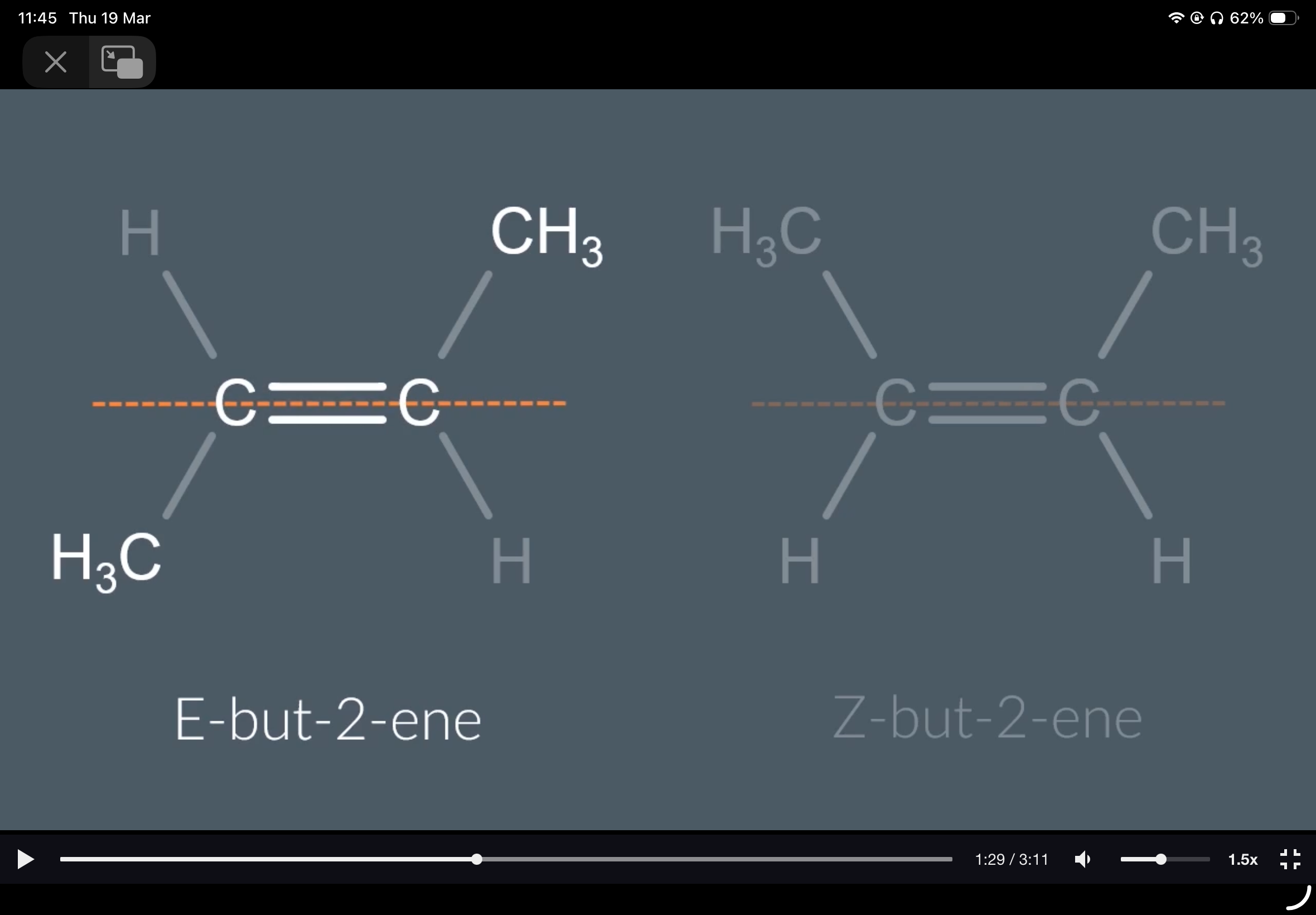
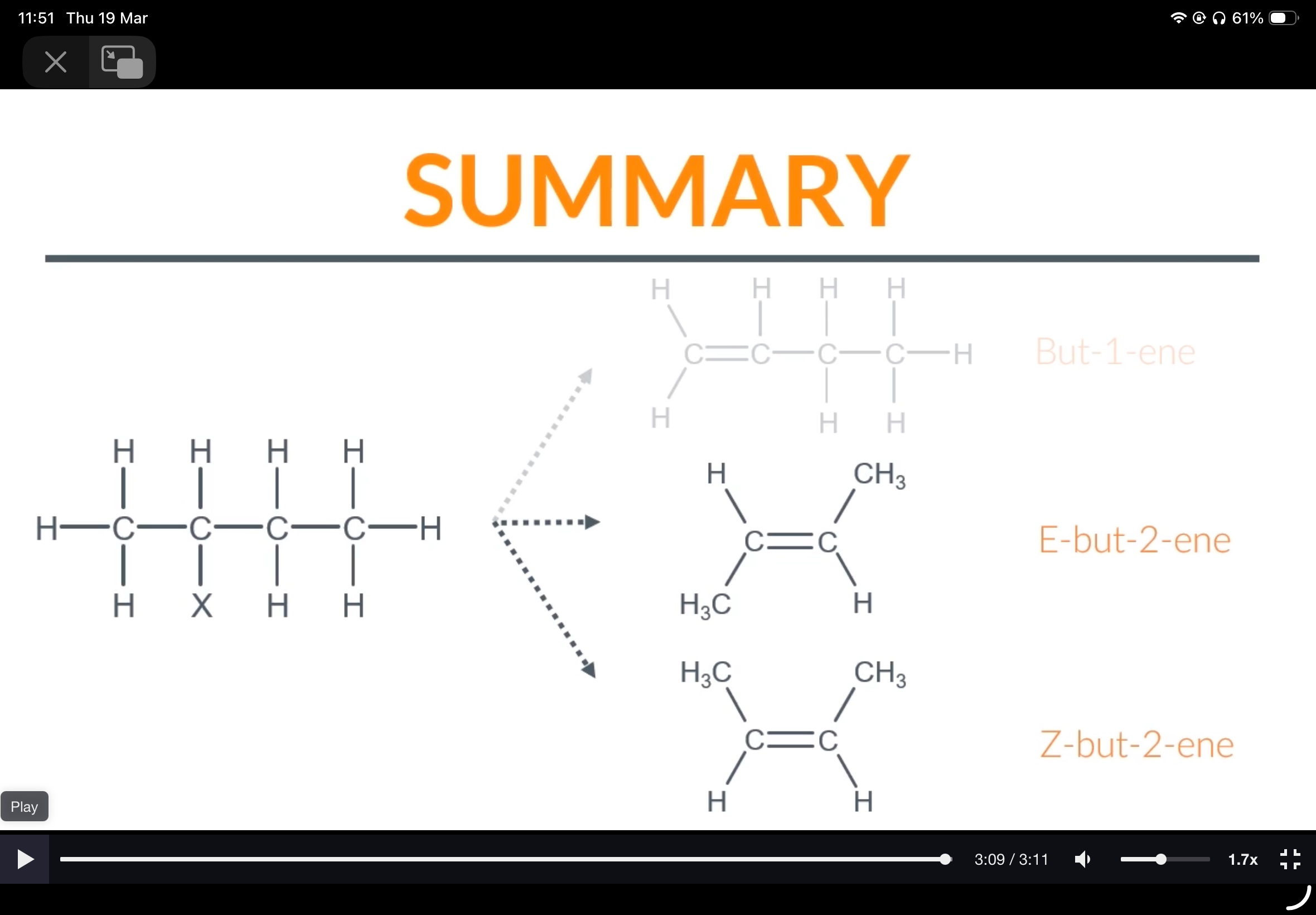
Draw the structures of E-but-2-ene and Z-but-2-ene
Propene. 2.A and C. 3.B. 4. two 5. E-Z
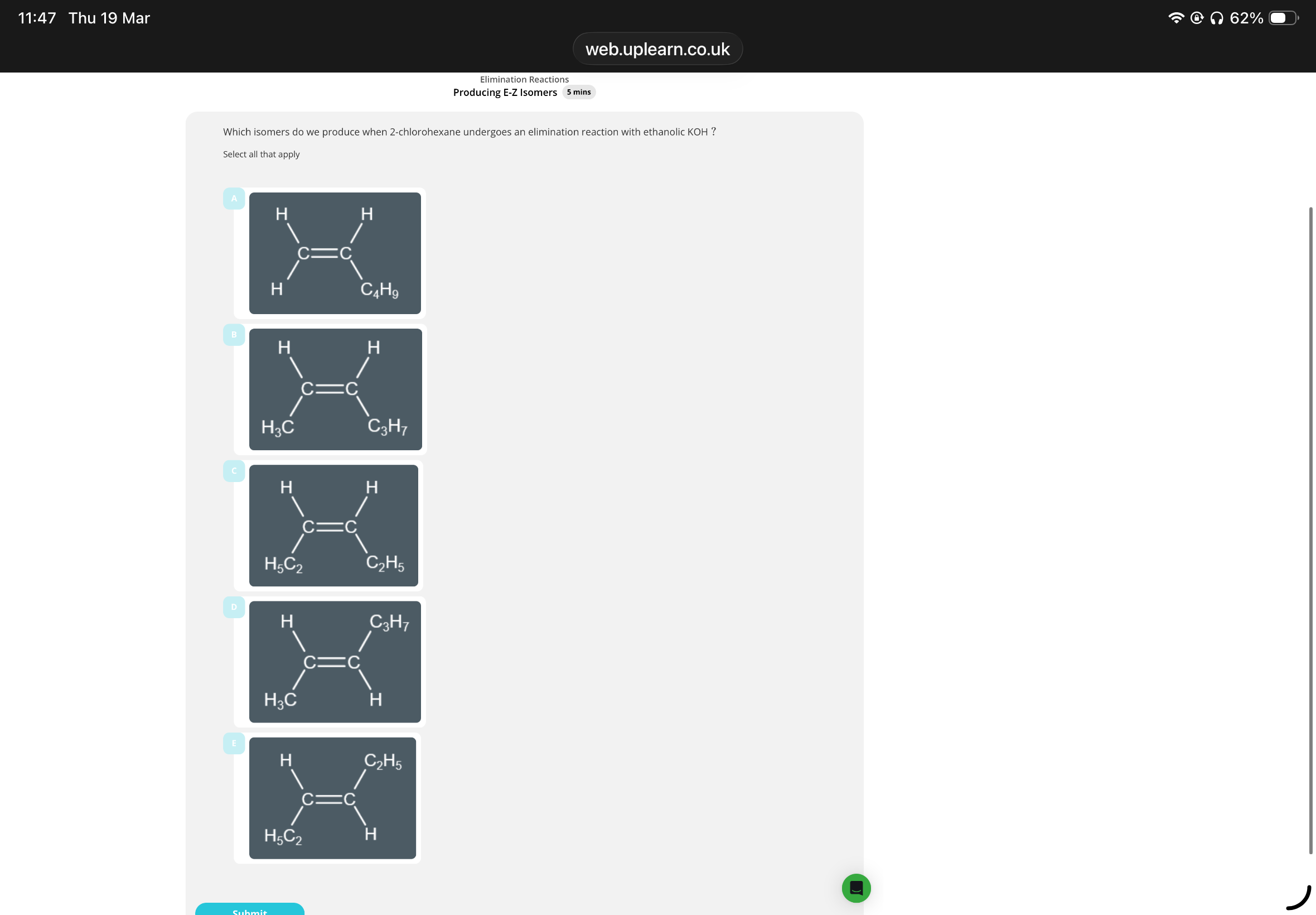
Answer image question?
During an elimination reaction, we can form up to two
…3? isomers and at least one of these isomers can show
…4? isomerism
A,B,D. 3.structural 4.E-Z

Elimination:
Elimination is forms an …1? Compound. A hydrogen is lost from the carbon …2?(next to) to the OH or X functional group. The elimination reaction is going to form a mixture of stereoisomers in addition to structural isomers. (Image on the other side)
Unsaturated. 2. Adjacent


Note this image shows an example elimination reaction meachanism:
( The OH acts as a base forming a bond with a H adjacent to the Carbon with an X. The H that has recieved the electrons transfers the shared pair to the C-C bond where the X is bonded to one of the carbons(forming a pi bond). Since C and X have different electronegativities, the shared electron pair in the C-X bond will move to the X causing X to break off. The products formed will be an alkene + H2O + halogen )
Draw the reaction mechanism where 1-bromopropane reacts with Ethanolic KOH under reflux?

in the image it shows Butan-2-ol reacting with aluminium oxide (conc phosphoric acid and sulfuric acid would also be represented by H+ since they are all acids in this elimination reaction)
Example of the elimination reaction mechanism with propan-2-ol and aluminium oxide(note- These acid catalysts can be represented as H+)
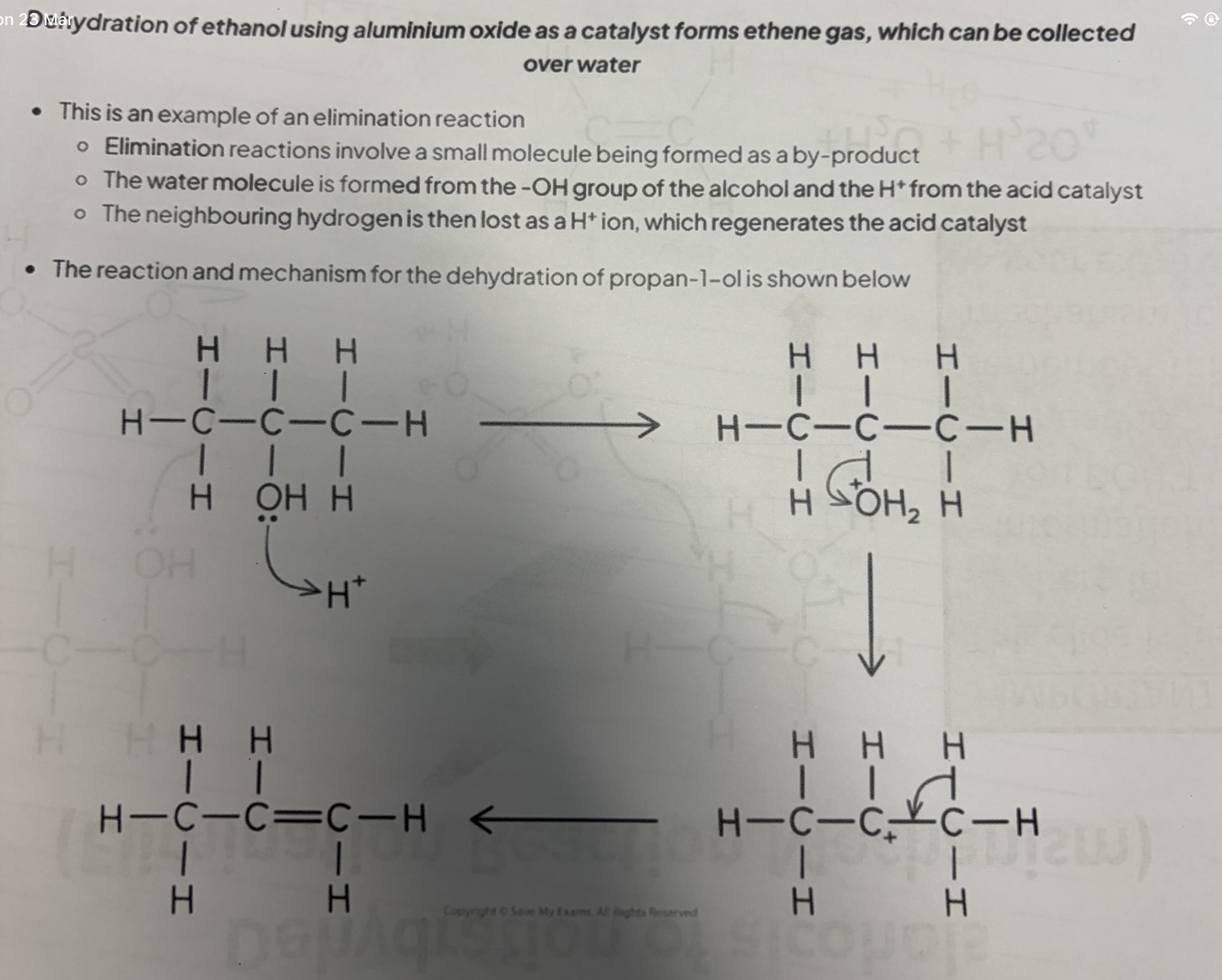
Explanation:
The lone pair on the OH transfers its lone pair to the H+ from the acid and this forms a bond and creates water and the O becomes + whilst H+ becomes neutral.
The bond attaching the carbon and H2O breaks and the shared pair of electrons go the the O which eliminates water and the C becomes positive.
A bond between H and C is broken and the shared electrons move to the carbon carbon bond which has the C+. This forms a double bond between the carbon and causes the C+ to become neutral. The H is also removed.
This forms propene.
Draw the mechanism for the dehydration of propan-1-ol when it reacts with an acid.
Draw the reaction mechanism when butan-1-ol with H2SO4 under reflux?
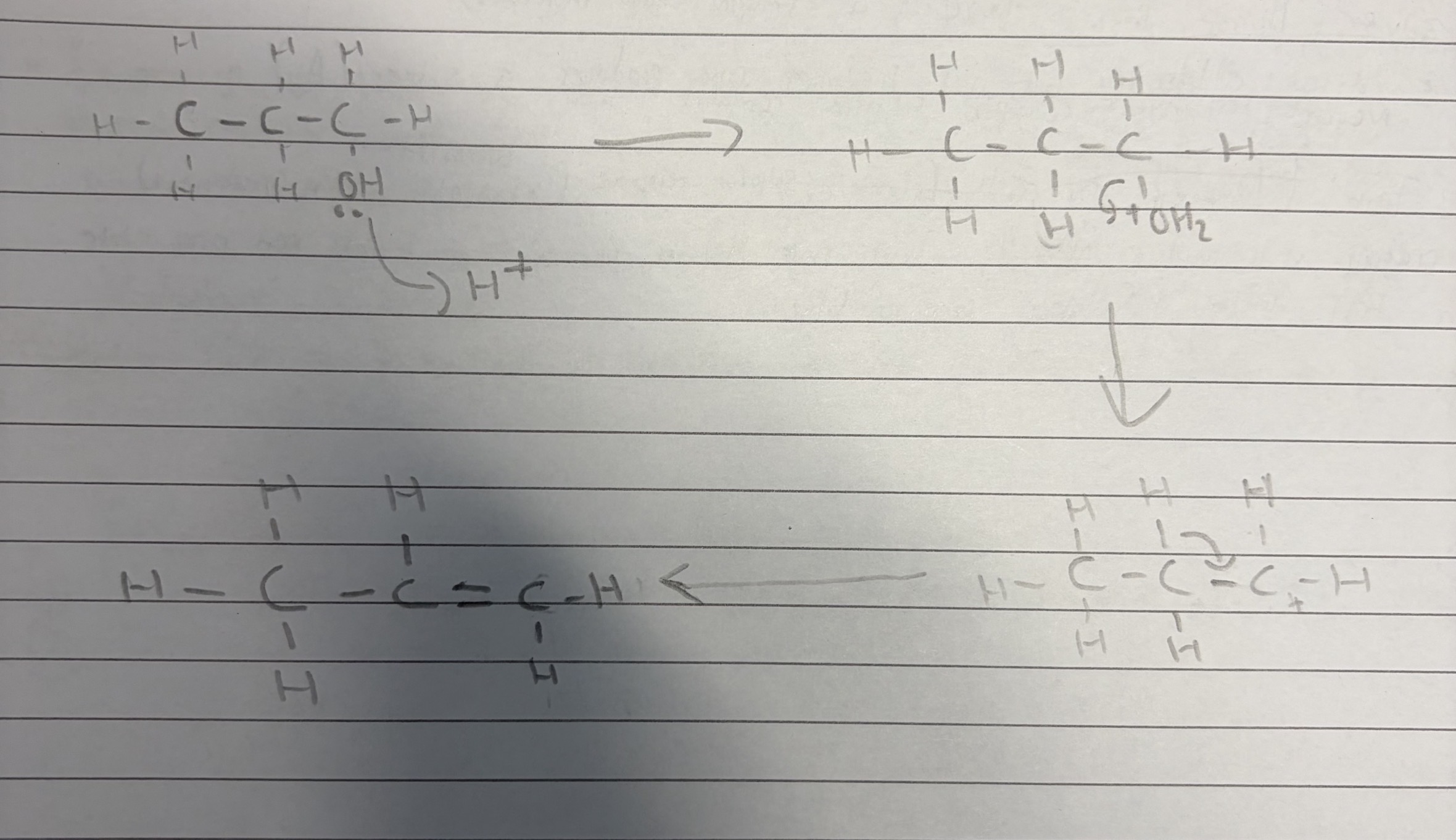
Elimination reactions
Substitution or Elimination?
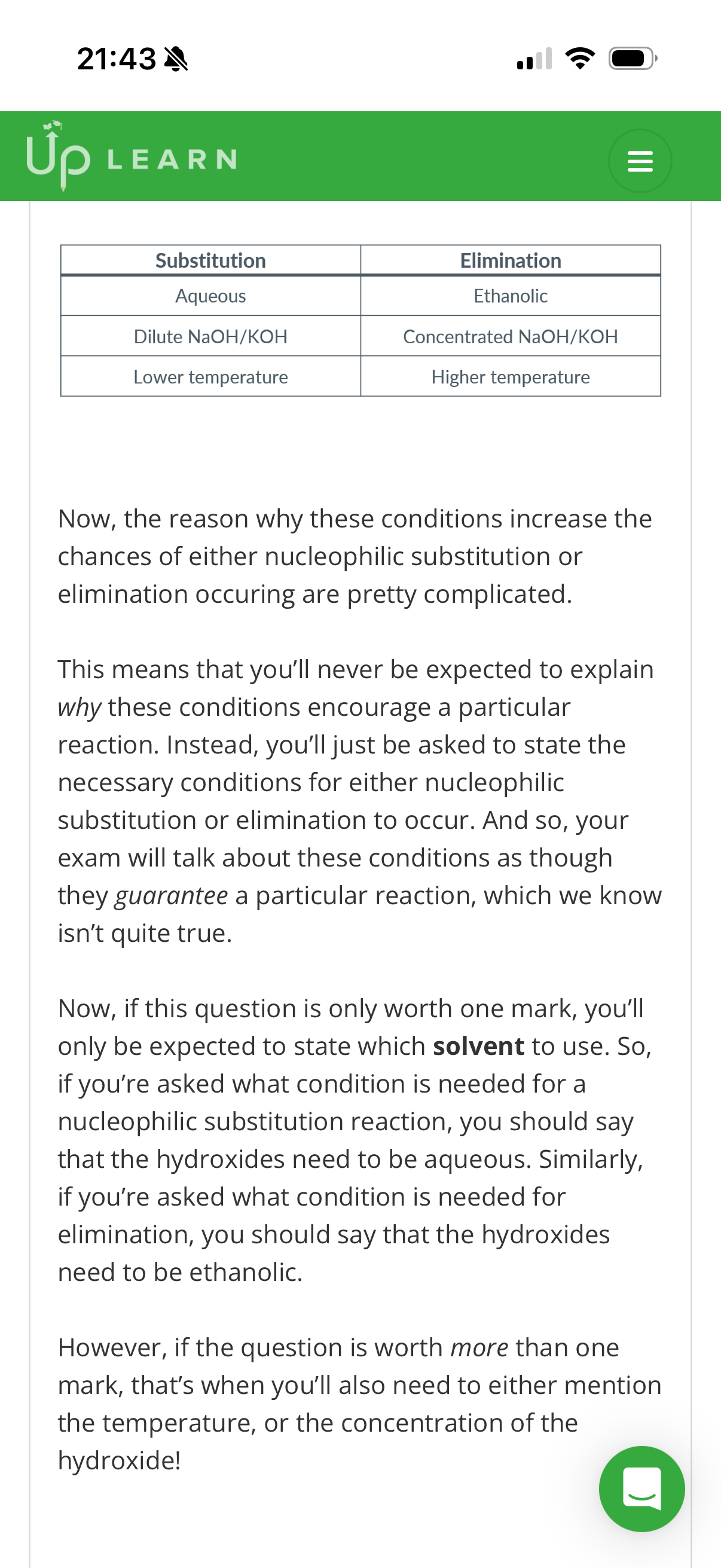
We’ve seen that when halogenoalkanes react with …1? hydroxides, they undergo nucleophilic substitution reactions.
And we’ve also now seen that when halogenoalkanes react with …2? hydroxides instead, they undergo elimination reactions.
However, the reality is actually a little more complex than that.
Normally, halogenoalkanes are reacted with hydroxides in a mixture of …3? and ethanol. And actually, it’s the …4? of each solvent that affects the type of reaction.
Also, there’s no proportion of water that absolutely guarantees that substitution will occur. And there’s no proportion of ethanol that guarantees that elimination will occur.
And to make these reactions even less certain, there are also other factors which affect which type of reaction will take place!
For example, reactions with …5?hydroxides increase the chances of nucleophilic substitution occurring. However, reactions with …6? hydroxides increase the chances of elimination occurring.
Additionally, while we typically heat the mixture for both reactions, …7? temperatures increase the chances of nucleophilic substitution occurring. However, …8? temperatures increase the chances of elimination occurring.
Now, the reason why these conditions increase the chances of either nucleophilic substitution or elimination occuring are pretty complicated.
This means that you’ll never be expected to explain why these conditions encourage a particular reaction. Instead, you’ll just be asked to state the necessary conditions for either nucleophilic substitution or elimination to occur. And so, your exam will talk about these conditions as though they guarantee a particular reaction, which we know isn’t quite true.
Now, if this question is only worth one mark, you’ll only be expected to state which solvent to use. So, if you’re asked what condition is needed for a nucleophilic substitution reaction, you should say that the hydroxides need to be …9?. Similarly, if you’re asked what condition is needed for elimination, you should say that the hydroxides need to be …10?.
However, if the question is worth more than one mark, that’s when you’ll also need to either mention the temperature, or the concentration of the hydroxide!
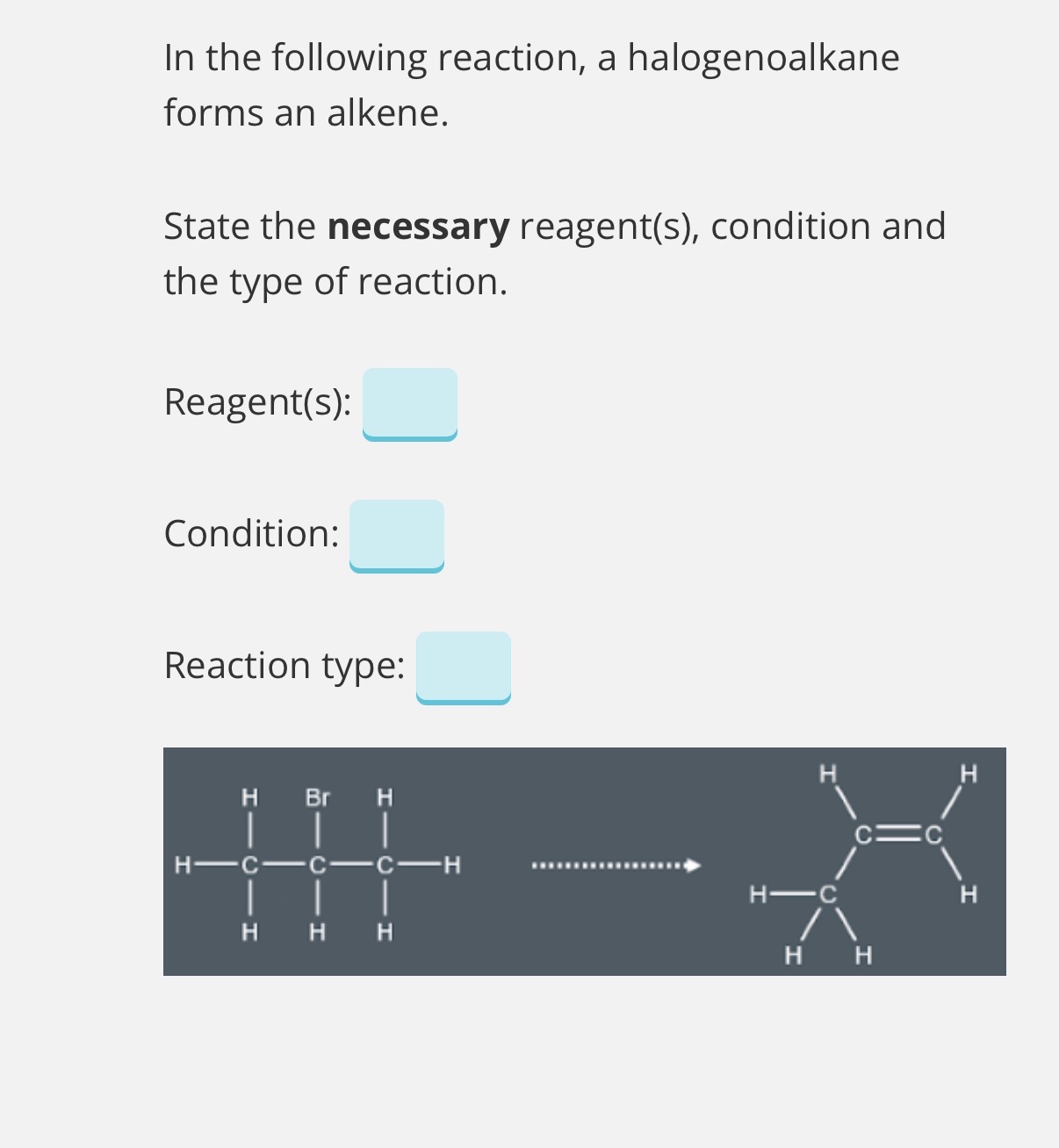
Answer image question?
Aqueous. 2. Ethanolic. 3. Water
Proportion. 5.dilute. 6.concentrated. 7.lower. 8.higher
Aqueous. 10.ethanolic
Reagent: KOH Condition: Ethanolic. Reaction type:Elimination
(Explanation
In this reaction, two sigma bonds are broken (C-H and C-Br) and a pi bond is formed (C=C). Therefore, this is an elimination reaction.
For a halogenoalkane to undergo elimination, they must react with an ethanolic hydroxide.
Elimination reactions
Bases in organic chemistry
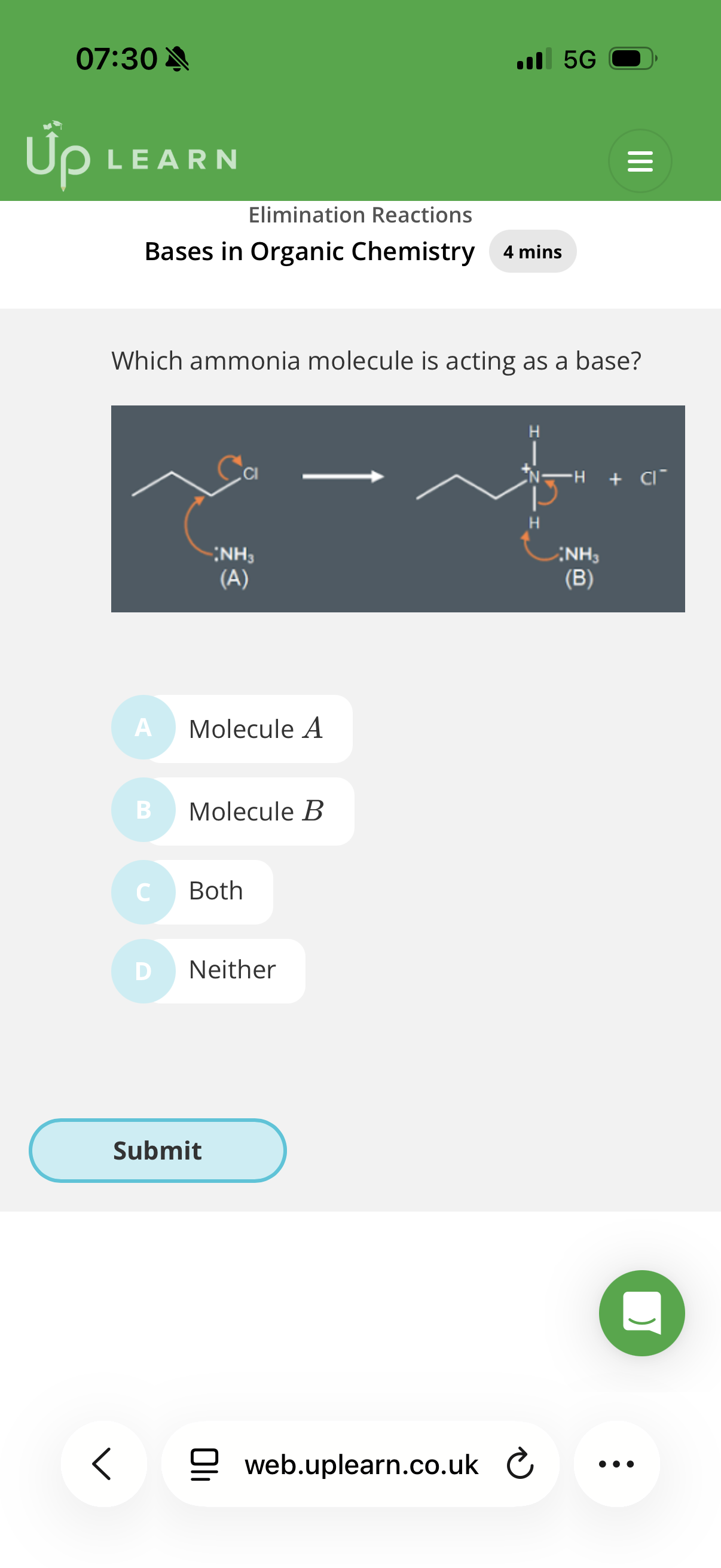
The Brønsted-Lowry model defines a base as a(n)...
Info:(If you see an arrow going from a lone pair to a Hydrogen, you can bet your life it's acting as a base. If that arrow points to a Carbon, it's acting as a nucleophile)
Answer image q?
• All bases are …3?, but not all Nucleophiles are …4?(because not all Nucleophiles accept a proton to form a new covalent bond,unlike bases)
Question 5 is in the back image
Note- if the exam asks to state the role of a species donating electrons to a proton(H), you will only get the mark if you state that it’s a base.
Proton acceptor (they have negative charges) (When a species accepts a proton, we say it is acting as a base)
B
Nucleophiles. 4. Bases
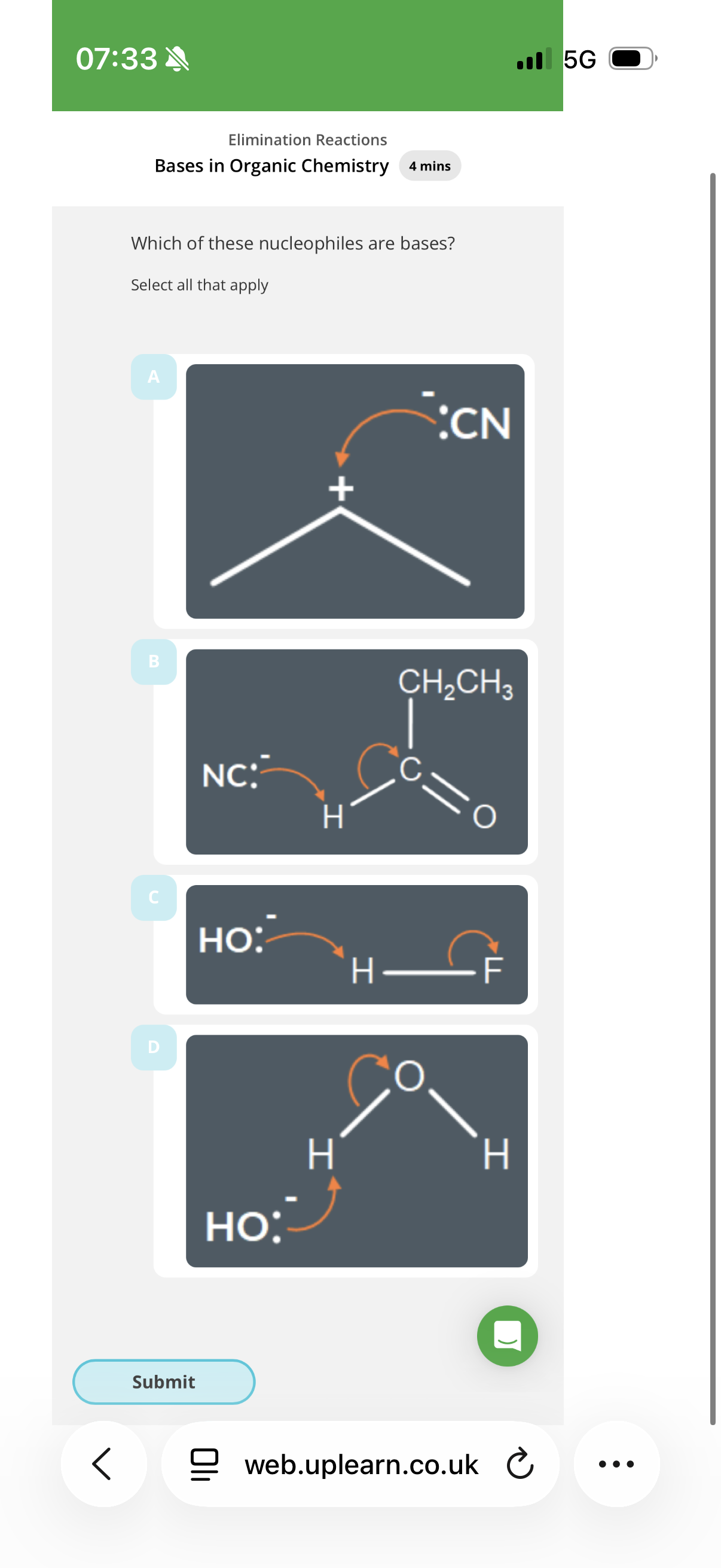
B,C,D
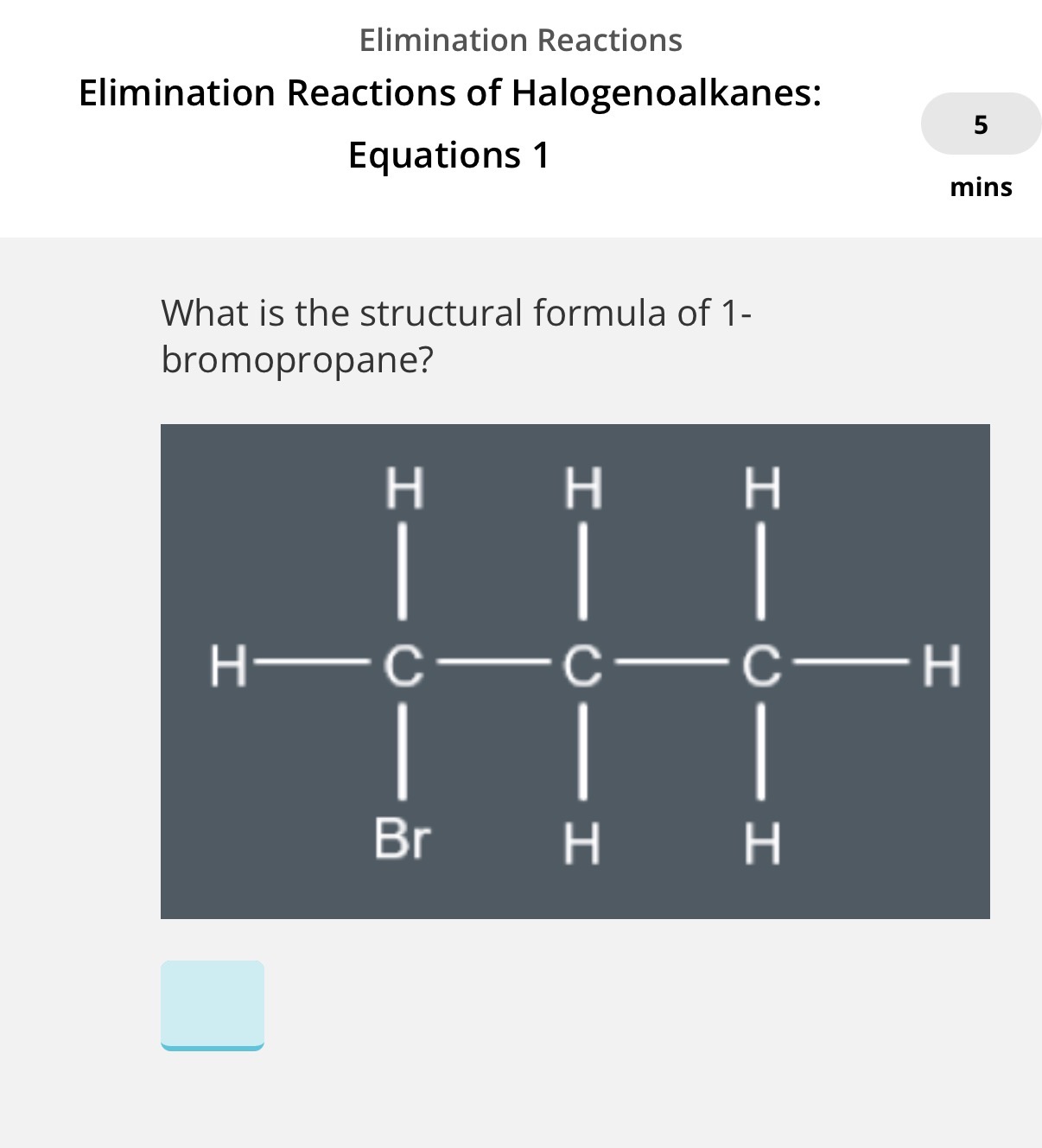
To show equation of a halogenoalkane reacting with Ethanolic KOH, we write the structural formula of the halogenoalkane that is reacting with KOH. Now answer the image question?
Info• In the image, we can only form 1 structural isomer which is propene. If only 1 structural isomer is able to be produced for the alkene(product) you can write the molecular formula.
What is the molecular formula of propene?
What does the ionic equation for this reaction look like?(q1 reaction)
Full equation:
CH3CH2CH2Br+KOH→...Info: When the alkene product does have a structural isomer, we have to write it in the equation in structural formula.
Whats the structural formula of but-1-ene?
What is the full equation for the conversion of 2-chlorobutane to but-2-ene using ethanolic KOH?
A: CH3CHClCH2CH3+ KOH→ C4H8+ H2O+ KCl
B: CH3CHClCH2CH3 + −OH → C4H8 + H2O +Cl−
C: CH3CHClCH2CH3. + −OH → CH2=CHCH2CH3 + H2O + Cl−
D: CH3CHClCH2CH3. + KOH. → CH3CH=CHCH3 + H2O + KCl
•When we write equations for elimination reactions of halogenoalkanes, we write the …7? formula of the alkene when it has position isomers.
Your answer was missing:CH3CH2CH2Br
C3H6
CH3CH2CH2Br + −OH → C3H6 + H2O + Br−
Your answer was missing:
C3H6+KBr+H2O
CH2=CHCH2CH3
D (D is the correct answer, not A
This equation doesn’t include the structural formula of the alkene.)
(B: This is the ionic equation and it doesn’t include the structural formula of the alkene.)
(C: This is the ionic equation and it includes the structural formula for but-1-ene.)
Structural

Answer image q?
Identify the correct ionic equation for the reaction of ethanolic KOH with 1-bromopentane.
Correct answersYour answers
A: CH3CH2CH2CH2CH2Br + KOH → CH3CH2CH2CH2CH2OH + KBr
B: CH3CH2CH2CH2CH2Br + KOH → CH3CH2CH2CH=CH2 + H2O + KBr
C: CH3CH2CH2CH2CH2Br + OH− → CH3CH2CH2CH2CH2OH + Br−
D: CH3CH2CH2CH2CH2Br + OH− → CH3CH2CH2CH=CH2 + H2O + Br−
E: CH3CH2CH2CH2Br + OH− → CH3CH2CH=CH2 + H2O + Br−
D