3.1.1.2 - Mass Numbers and Isotopes
0.0(0)
Studied by 0 peopleCard Sorting
1/3
There's no tags or description
Looks like no tags are added yet.
Last updated 5:58 PM on 4/11/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
1
New cards
Define relative atomic mass
The average mass of an atom of an element
relative to 1/12th the mass of a Carbon 12 atom
2
New cards
How do you calculate RAM (relative atomic mass)?
Multiply each mass (m/z) by each relative intensity
Calculate the sum of them all
Divide by the sum of the relative intensity
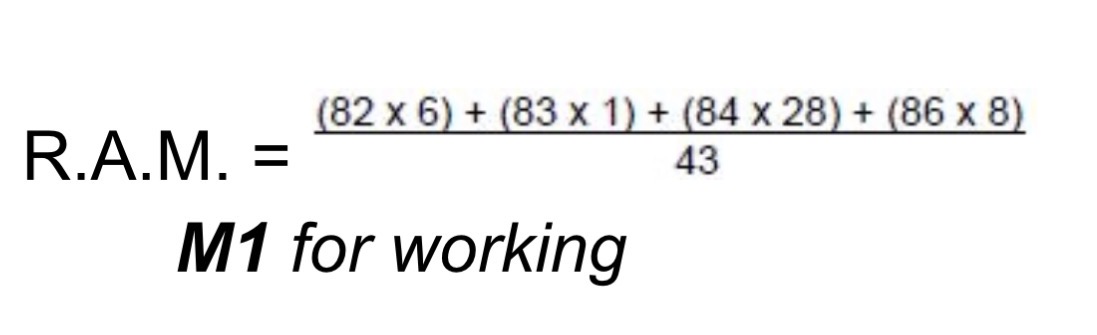
3
New cards
What is the TOF equation?
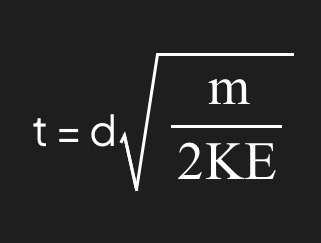
4
New cards
What unit does mass have in a TOF mass spectrometer?
Kilograms (kg)