C3: The Chemical Building Blocks of Life
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
How are macromolecules built up?
Macromolecules are built up from far smaller compounds with simpler chemical structures
Macromolecules are ______ built by ______ ______.
polymers, linking monomers
What are the 4 classes of macromolecule?
Carbohydrates, Proteins, Lipids, Nucleic Acids
What process is used for formation of macromolecule?
Dehydration, which is the removal of a water molecule.
Explain how dehydration leads to formation of macromolecules.
An H atom is removed from one monomer, while a hydroxyl (-OH) is removed from the other, forming a covalent bond between the two monomers.
What is the process used for breakdown of macromolecules?
Hydrolysis, which is the addition of a water molecule.
Explain how hydrolysis leads to breakdown of macromolecules.
An H atom is attached to one subunit , while a hydroxyl is attached to the other, breaking a specific covalent bond in the macromolecule.
Carbohydrates is a group of molecules that all contain 1. ____, ____ and ____ in the 2. ____ ratio _:_:_, and has the 3._____ formula of ______, where n = number of ____ atoms.
1. carbon, hydrogen, oxygen
2. molar, 1:2:1
3. empirical, (CH2O)n, carbon
Carbohydrates contain many 1._____- _____ bonds, which 2. _____ _____ when 3. _____ occur.
1. carbon-hydrogen
2. releases energy
3. oxidation
Carbohydrates can be classified into 3 groups:
Monosaccharides, Disaccharides, Polysaccharides
What is a monosaccharide? How are they grouped?
A monosaccharide is the simplest form of carbohydrates, so it cannot be broken down into smaller sugars.
They are grouped by how many carbon atoms they have in their structure (3/5/6)
Give 3 examples for 6-carbon sugar, 2 for 5-carbon sugar, 1 for 3-carbon sugar.
Glucose, fructose, galactose
Ribose, deoxyribose
Glyceraldehyde
What are isomers, as well as its structural and stereo variants?
Isomers are organic molecules that have the same molecular structure or empirical formula.
Structural isomers are different in structure of carbon skeleton, while stereoisomers have the same carbon skeleton but differ in how the groups in the skeleton are arranged.
____ is the most important 6-carbon sugar for _____-storage, and contains ___ energy storing __ - __ bonds.
Glucose, energy , C-H
____ is a structural isomer of glucose; it differs in the position of the ___________(___) and is more _____ than glucose.
Fructose, carbonyl carbon, sweeter
_____ is a _____ of glucose, differing in the position of ___ and ___ relative to the ring.
Galactose, stereoisomer, -OH, -H
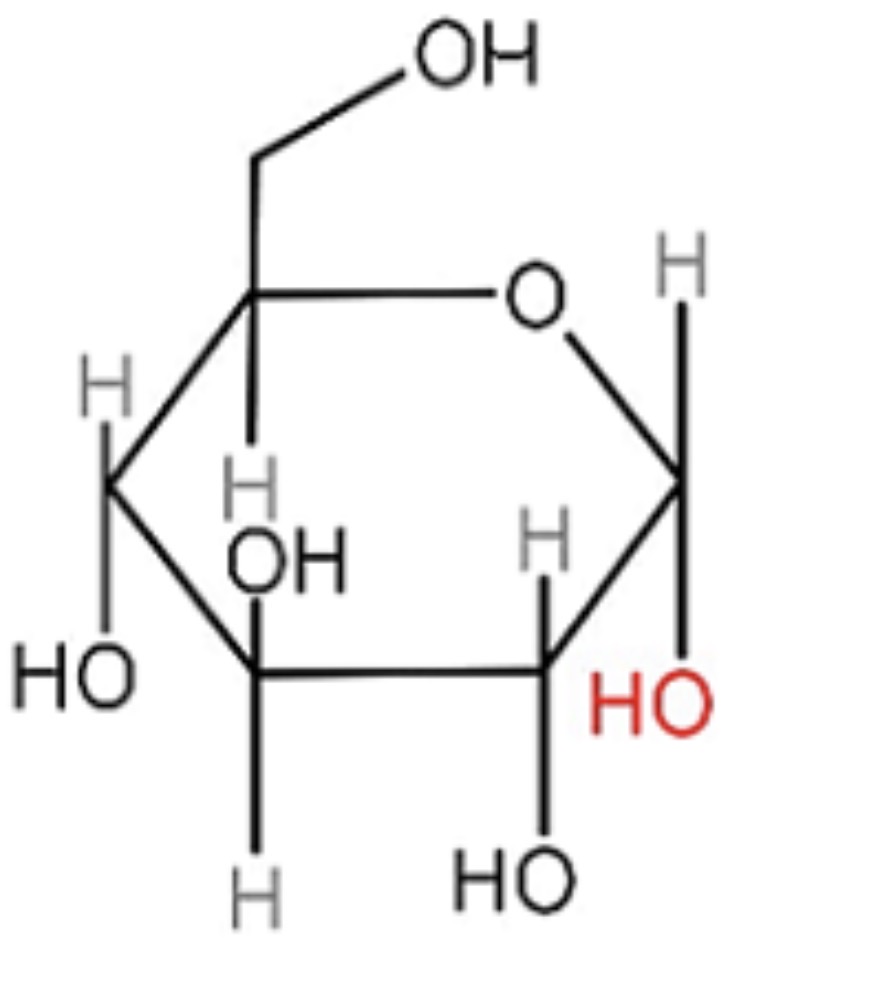
Identify this.
Alpha - glucose
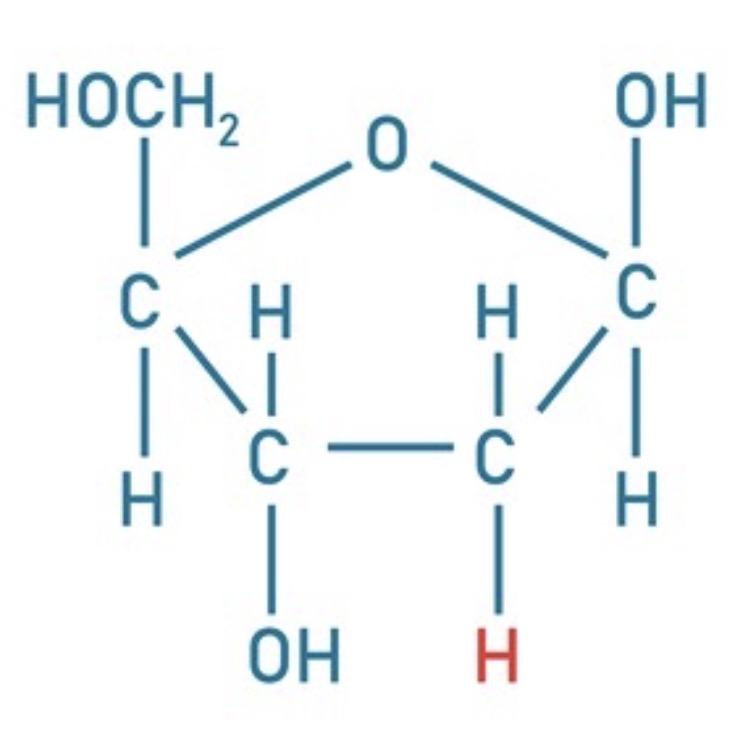
Identify this.
Deoxyribose
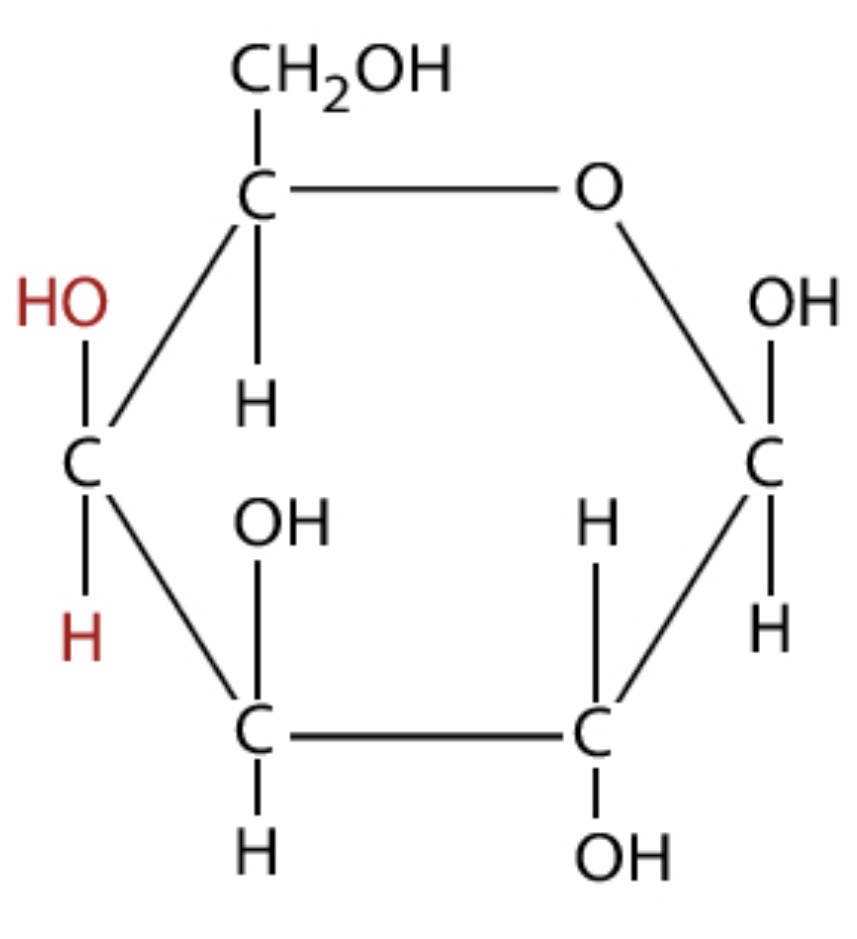
Identify this.
Galactose
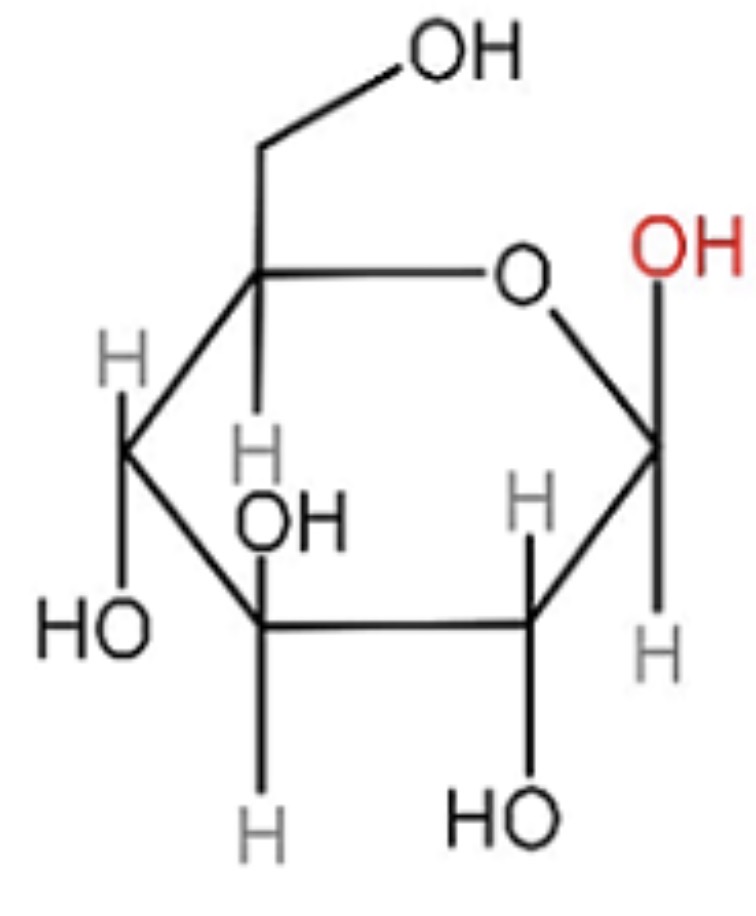
Identify this.
Beta-glucose
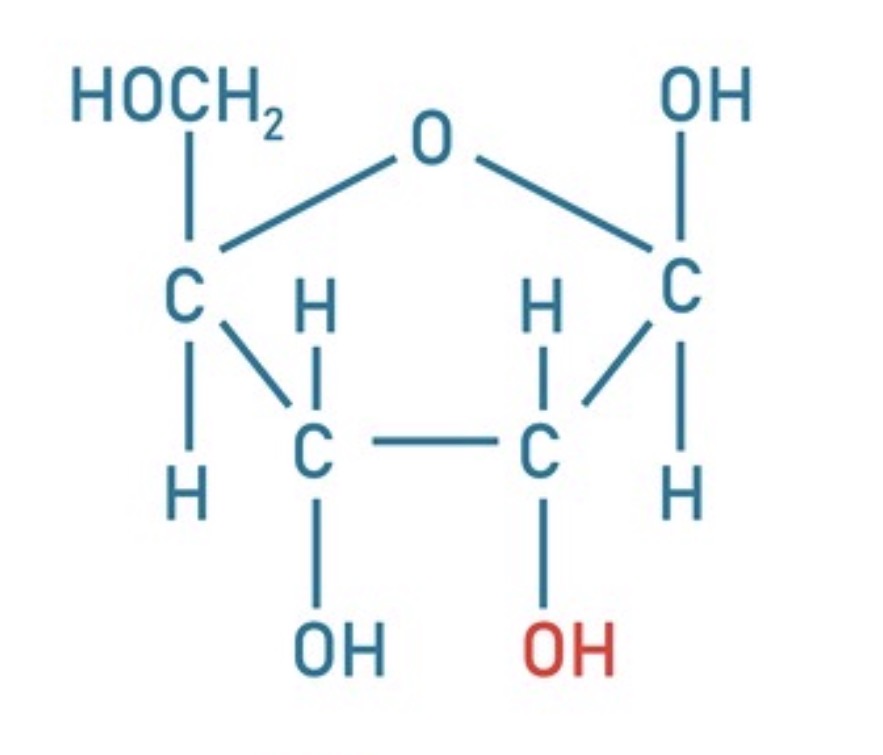
Identify this
Ribose
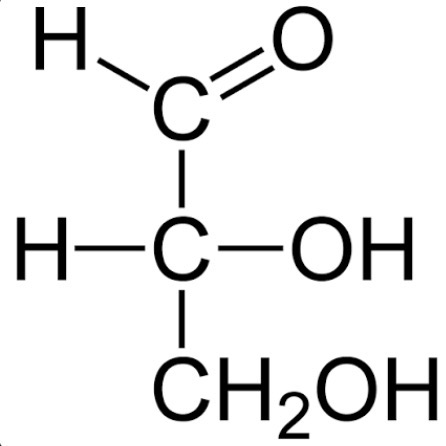
Identify this.
Glyceraldehyde
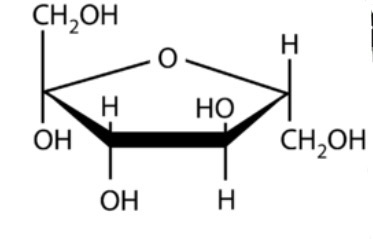
Identify this.
Fructose
What are disaccharides?
They are transport sugars made out of 2 monosaccharides linked by dehydration synthesis.
Why are disaccharides called “transport sugars”?
They are used to move energy around organisms as they are small enough to dissolve in water, are stable and non-reactive compared to glucose, and can be transported in blood or plant sap without being immediately used up.
Which disaccharide is commonly known as table sugar? Which two monosaccharides is it made out of and what is this form normally used for?
Sucrose. It is made out of fructose and glucose and it is the form most plants use to transport glucose.
Which disaccharide is commonly known as milk sugar? Which two monosaccharides is it made out of and what is this form normally used for?
Lactose; It is made out of glucose and galactose and is the form used by most mammals to supply energy to their young.
What is maltose used for and which two monosaccharides is it made out of?
Maltose is used in grain for storage, and it is made out of 2 glucose.
What are polysaccharides? What are they used for?
Polysaccharides are long chains of monosaccharides linked through dehydration synthesis. They are used for energy storage or structural support in animals and plants.
Both starch and glycogen are ______ of ______ molecules.
polymers, glucose
Starch is used as _____ _____ in _____, and is made up of ______ subunits.
energy storage in plants, alpha-glucose
There are two types of starch: _____ and _____.
amylose and amylopectin
What is amylose made out of?
It is made out of many hundreds of alpha-glucose molecules linked together in long, unbranched chains. Each linkage occurs between carbon 1 of one glucose molecule, and carbon 4 of another, making them a-1→4 linkages.
Amylose tends to ______ in water, thus making it _____ as it is _____ and its surface are _____ _____ to water.
coil-up, insoluble, compact, less exposed
Similar to amylose, amylopectin is (structure), but it ____ ____. Amylopectin is also ____ in water, forming ____ ____ in ____ ____.
made out of many hundreds of alpha-glucose molecules joined by a-1–>4 molecules; forms branches.
insoluble, starch granules, plant cells
Describe the branches of amylopectin. Each branches consist of ___ to ___ _____ subunits.
Branches occur at bonds between C-1 of one molecule and C-6 of another, forming a-1—> 6 linkages.
20-30 glucose
Most plant starch is made up of _____.
Amylopectin
Glycogen is a _____ form in _____ made out of _____ subunits (equivalent to starch in plants). It is ____ in water and is similar to _____ which contains _____ _____, but has a much _____ _____ _____ _____ and a lot more _____.
storage, mammals, a-glucose
insoluble, branched chains, longer average chain length, branches
Cellulose is a structural polysaccharides made out of _____ subunits. It is _____ and _____, linked by _____ linkages.
beta-glucose, long and unbranched, B-1–>4
Cellulose forms _____ _____, which is very _____ and quite _____ to _____ _____. It is also the chief component of _____ _____ _____.
long fibres, strong and resistant to metabolic breakdown, plant cell wall
Why can’t most organisms (humans,etc) break down cellulose?
Most organisms have enzymes that only recognise a linkages but not B linkages.
____ is the principal ____ ____ of many ____, such as lobsters. It is a ____ of _________, a derivative of ____. It forms ____, ____ ____ material when ____ - ____ by _____( ____ ____ of insects and crustaceans).
Chitin, skeleton element, invertebrates
polymer, N-acetylglucosamine, glucose
hard, resistant surface, cross-linked, proteins, hard exoskeleton
What are proteins?
Proteins are long, linear polymers of amino acids. They are also composed by one or more long, unbranched chains of amino acids linked by peptide bonds (chain is called polypeptide).
General chain of proteins?
Amino Acids → Polypeptides → Proteins
Proteins are ____ by ____ found in ____, and are ____ in the ____ by the process of ____ using ____ as a ____.
encoded, genes, DNA, synthesised, cell, translation, mRNA, template
Proteins are ____ to ____ ____ as they are involved in many ____ ____ and ____ like ____.
essential, cellular functions, cellular activities and processes like enzymes
____ is the ____ ____ of protein, and has a _____ _____ atom linked to ___ side groups: ___ group (__), ___ group (__), a ___ ___ (__), ___ side group (__).
Amino acid, building block, central carbon, 4, amino (-NH2), carboxyl (-COOH), hydrogen atom (H), functional (R)
The 20 common amino acids can be divided into groups based on their R group; ___ ___ amino acids, ___ ___ amino acids, ___ amino acids, ___ amino acids and amino acids with ___ ___. Explain what each groups mean.
(Non Polar Crabs Are Unique)
Groups: Have R groups that contain:
Non-polar: -CH2/ -CH3
Polar-unchanged: Oxygen/ Hydroxide (-OH)
Charged: Acids/ Bases that can ionise
Aromatic: Organic (carbon) ring with alternating single/double bonds
Unique: properties
20 common amino acids:
__ are non-polar (aromatic: __ each); Another __ are polar and charged each (aromatic: __ each); remaining __ special-function amino acids have ___ ___ that allow them to help ___ ___ between ___ ___ or ___ in proteins.
7,2
5,1
3, chemical properties, form bonds, protein chains, kinks

How is a peptide bond formed?
Amino acid ionises at physiological pH (~pH 7): Carboxyl group (-COOH) → negatively-charged carboxylate ion (-COO) at one end, while amino group (-NH2) → positive amino group (NH3+) at the other end.
The amino group of one acid can undergo dehydration reaction with the carboxyl group of another A.A to form a covalent peptide bond (-CO-NH-) joining the two amino acids together.
*the resulting dipeptide still has an amino end and carboxyl end


Why is the resulting peptide chain not able to rotate freely?
Because of the partial double-bond nature of peptide bonds

What are the 4 levels of protein structure?
Primary, Secondary, Tertiary, Quaternery
The primary structure of protein is its ___ ___ ___ in a ___ ___. Secondary structure results from ___ ___ forming between two ___ ___ ___. This produces two different types of ___: _____ & ___ called_____. Regions of these secondary structures are ___ ___ ___ ___ to form the ___ ___ of the protein, which is the tertiary structure. Thus, tertiary structure is ___ ___ ___ of the protein. ___ structure is only found in proteins with ___ ___: in this case, the ___ ___ of protein is the ___ of ___ ___ ___ in ___.
amino acid sequence, polypeptide chain, hydrogen bonds, amino acids, beta (B)-pleated sheets, coils called alpha (a) helices
further folded in space, final shape, final 3D shape
Quaternary, multiple polypeptides, final structure, arrangement, multiple polypeptides in space

One example of a quaternary structure is the ___ ___, which is composed of __ ___ ___ subunits: __ copies of “___” & __ copies of “___”. Each chain is associated with a ___ ___ that has a ___ ___ ___, which can ___ to an ___ ___.
haemoglobin molecule, 4 protein chain, 2 “alpha chain” & 2 “beta chain”
heme group, central iron atom, bind to oxygen molecule
Name all the interactions that can contribute to/stabilise a protein’s tertiary structure.
1.Hydrogen Bond: Form between different amino acids
2.Covalent disulfide bridge (strong bond:help protein keep shape): Form between two cysteine side chains
3.Ionic bond/Salt bridges: Form between R groups with opposite charges
4.Van der Waals forces: Weak attraction between atoms due to oppositely polarised electron clouds
5.Hydrophobic exclusion: Non-polar R groups of protein tend to gather in protein’s interior; Polar portions of protein tend to gather in protein’s exterior and interact with water
What is protein denaturation?
It is the change of shape and unfolding of protein due to altered environment (e.g.pH,temperature,ionic concentration), leading to loss of function (like enzyme activity).
Describe what happens to protein during high heat.
Heating imparts energy to protein molecules; this added energy is enough to break the relatively weak forces that hold protein in its refolded and helical tertiary and secondary configurations.
As the process of denaturation continues: protein molecule unfolds more and more, the internally directed hydrophobic regions become more exposed on outside of molecules. Hydrophobic regions will associate with other hydrophobic regions on other protein molecules instead of interacting with water, clumping together known as aggregation. The molecules aggregate into a very large water-insoluble collection that is randomly assembled.
As proteins continue to denature, latticework structures grow amorphously and attract solvent water molecules into cell-like structures. Self-associated water molecules will adhere to surface of hydrophilic regions of proteins, while hydrophobic regions dissolve into each other and provide the energy to retain the structure.
As this process continues irreversibly, all protein molecules are recruited to this large, insoluble mass in a randomly organised structural framework that contains entrapped water molecules.
State examples of denatured proteins assembling into 3-D structures.
Coagulation of egg whites: Assembly of irreversibly denatured protein molecules results in formation of solid gel. The gel entraps water molecules inside the whites into semi-solid structure, which holds its shapes under normal conditions.
Others: Baking of yeast-risen bread, coagulation of meat proteins by cooking in products like hot dogs, solidification of gelatinous upon cooling a solution
What is nucleic acid? What are the two types?
Nucleic acid is a polymer of nucleotides connected by phosphodiester bonds. The two types are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
DNA is found in the ___ ___ and contains ___ that ___ for ___.
cell nucleus, genes, codes, proteins
What are the 3 main types of RNA? What important role do they all play?
Messenger RNA (mRNA)
Ribosomal RNA (rRNA)
Transfer RNA (tRNA)
*They all play a part in a role of protein synthesis, known as translation
Describe the 3 basic components of a nucleotide (DNA/RNA).
5-carbon sugar: Deoxyribose sugar for DNA, Ribose sugar for RNA
Nitrogenous base: Adenine, Guanine, Thymine, Cytosine, Uracil
A phosphate group
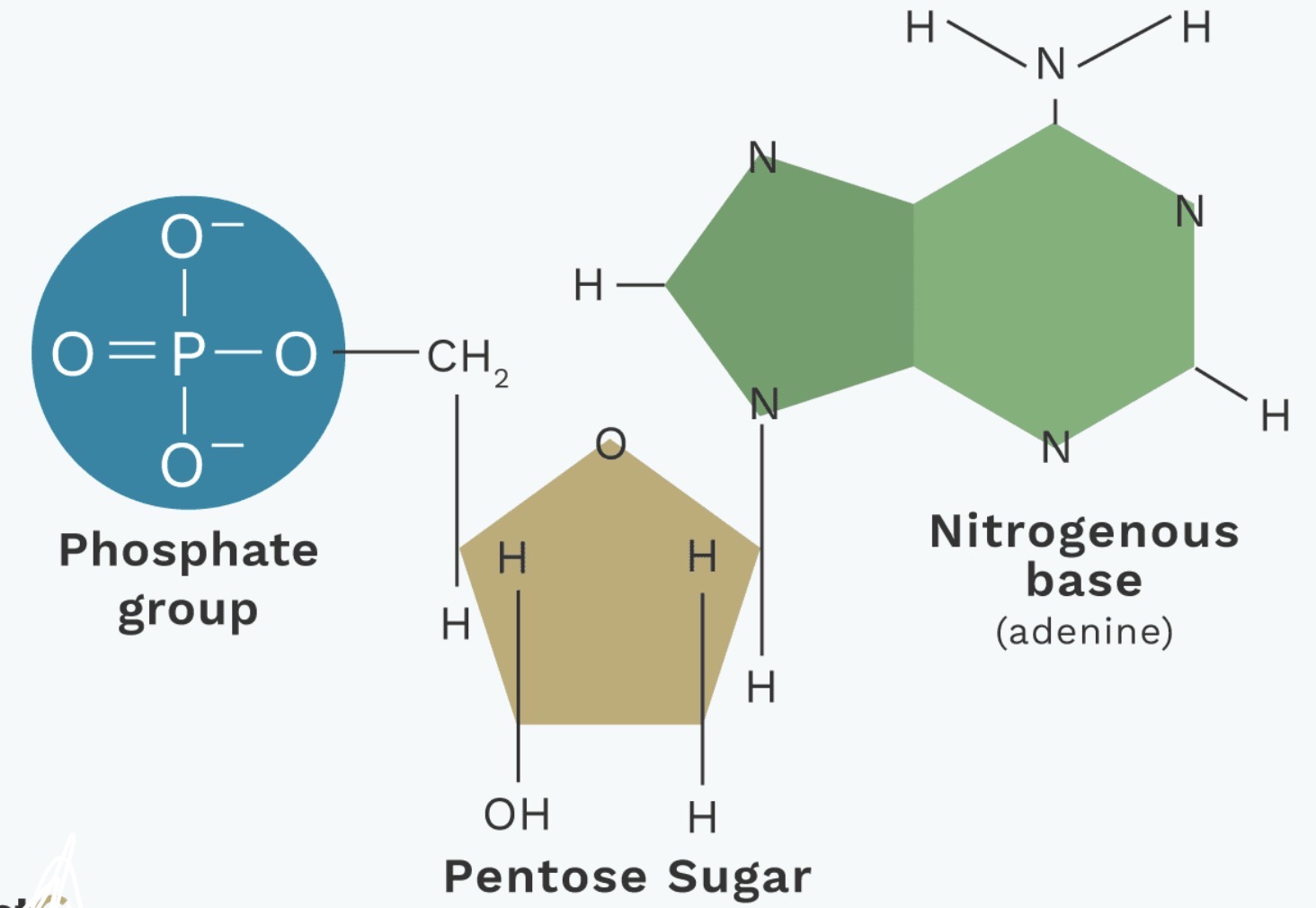
What are the two main types of nitrogenous base? Categorise them
Purine bases: Adenine (A), Guanine (G)
Pyrimidine bases: Cytosine (C), Thymine (T; only in) , Uracil (U)
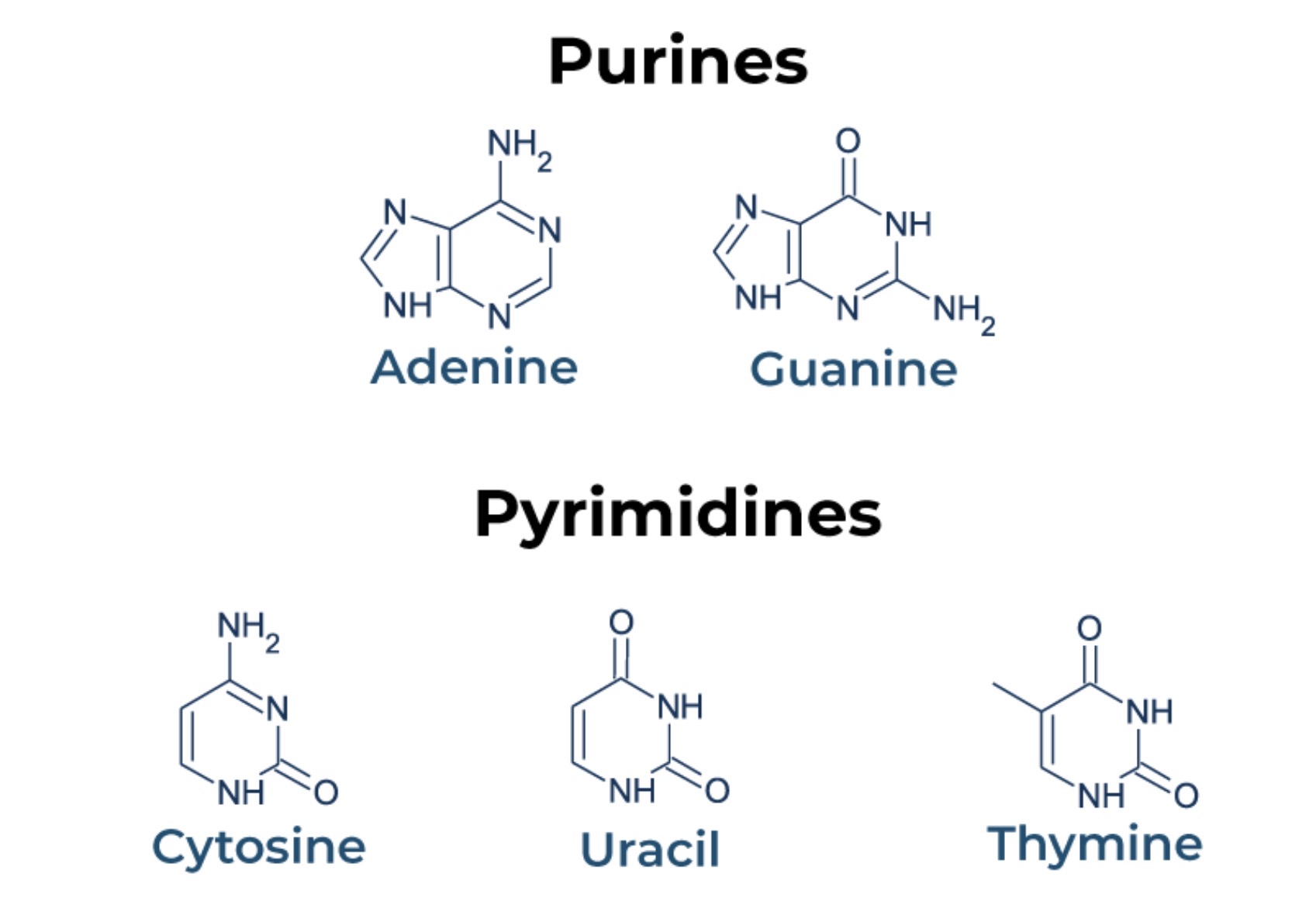
List the differences between DNA and RNA.
Double helix vs Single strand
Deoxyribose sugar vs Ribose sugar (in sugar-phosphate backbone)
Thymine vs Uracil (nitrogenous bases)
Describe the structure of nucleotides in-depth.
In a nucleic acid, nucleotides are linked to one another via phosphodiester bonds formed between the phosphate of one nucleotide and the sugar of the next nucleotide. The organic nitrogenous base protrude from this sugar-phosphate backbone; the backbone also has different ends: a 5’phosphate end and a 3’hydroxyl end (is from carbon atom no. in sugar). The organic nitrogenous bases can be either purines or pyrimidines; the base thymine is found in DNA, while the base uracil is found in RNA.
Describe the structure of DNA.
DNA contains genes that code for proteins. It is made out of two polynucleotide chains running in opposite directions/ anti-parallel to each other, which forms a double helix structure which are joined together by hydrogen bonds between complementary bases: Base pairing rule: A with T (2 hydrogen bonds), G with C (3 hydrogen bonds).
What are lipids? What is it excellent for and what are some examples?
Lipids are a loosely defined group of non-polar molecules that are insoluble in water that have a high proportion of C-H bonds. It is ann excellent form of energy storage. Examples include animal fat, olive oil, etc.
___ are a type of lipids found in ___ cells and are built from two kinds of molecules: ___ ___ & ___.
Fats, fat, fatty acids & glycerol
Fatty acids are ___ ___ ___ with a ___ (__) group at one end; glycerol is a __ ___ ___. A fat molecule consists of __ glycerol molecule and __ fatty acids attached by ___ ___, once to each ___ of ___ ___. Therefore, a fat molecule is also called a ___.
long-chain hydrocarbons, carboxyl (-COOH), 3-carbon polyalcohol, 1, 3, dehydration synthesis, carbon of glycerol backbone, triglyceride
The hydrocarbons chains of fatty acids can vary in length, the most common are ___-numbered chains of __ to __ ___.
even,14-20 carbons
Fatty acids can either be ___ or ___ . Explain.
Saturated: all internal carbon atoms are bonded to at least 2 hydrogen atoms
Unsaturated: contain double bonds between 1 or more pairs of successive carbon atoms
Unsaturated fatty acids can be further divided into ___ & ___.
Monounsaturated fatty acid: contains 1 double bond
Polyunsaturated fatty acid: contains 1> double bond
A saturated fat is composed of ___ that contain __ ___ ___ ___ (no ___ ___), therefore a saturated fat has the ___ number of ___ ___ bonded to its ___ ___. Most ___ fats are saturated, except some ___ oil.
triglycerides, 3 saturated fatty acids (no double bonds), maximum, hydrogen atoms, carbon chain, animal, fish
Unsaturated fat is composed of ___ that contain __> ___ ___ ___ (has 1 or more ___ ___). These have fewer than the ___ number of ___ ___ bonded to a ___ ___. ___ oils are typically unsaturated (EXCEPT: ___ & ___ oil). The many ___ of the ___ ___ prevents ___ from ___ ___, making them ___ oils at room temperature.
triglycerides, 1> unsaturated fatty acid (has 1 or more double bonds), maximum no. of hydrogen atoms, carbon atom
Plant, coconut and palm oil, kinks, double bonds, triglycerides, closely aligning, liquid
Most ___ ___unsaturated fatty acids have ___ ___ with a ___ configuration, if the ___ ___ is on the ___ side before and after the ___ ___.
naturally occuring, double bonds, cis, carbon chain, same, double bond
When fats are ___ ___ ___, this can produce ___ ___ with ___ configuration, if the ___ ___ is on the ___ side before and after the ___ ___. ___ fats are linked to ___ ___ of ___ ___ ___(__), which is ___ ___, while ___ ___ of ___ ___ ___, which is ___ ___. This leads to higher risk of ___ ___ ___.
partially hydrogenated industrially, carbon chains, trans, carbon chain, opposite, double bond, Trans, elevated levels of low-density lipoprotein (LDL), bad cholesterol, lowering levels of high-density lipoprotein (HDL), good cholesterol, coronary heart disease
What is a phospholipid molecule? What are the 3 components of it?
It is a triglyceride molecule with one of the fatty acids being replaced by phosphate group.
Glycerol, fatty acids (only 2 fatty acids attached to backbone), phosphate group (attached to one end of glycerol molecule)
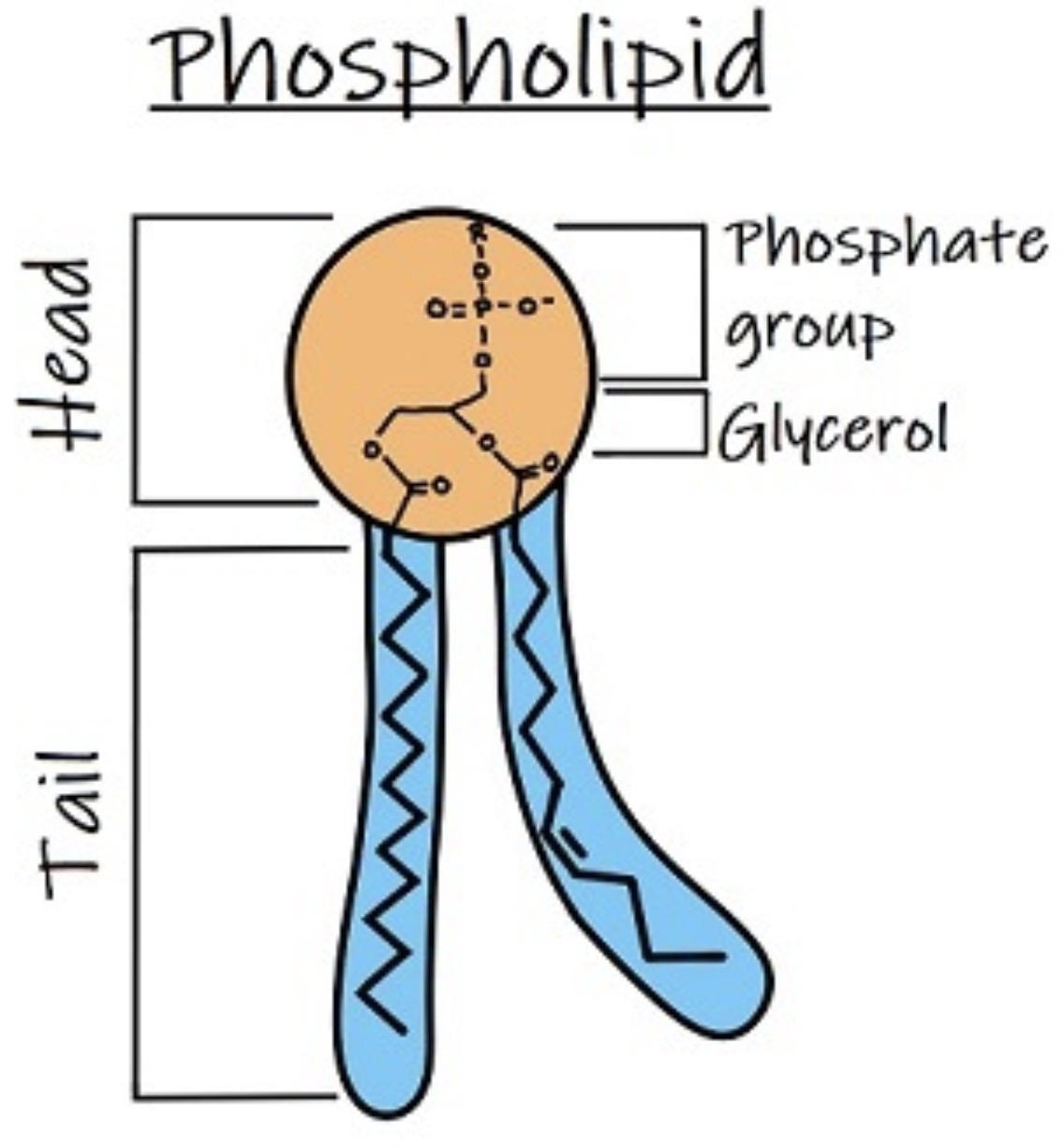
The phosphate group in phospholipid is ___ and usually has a ___ ___ ___ linked to it, such as ___, ___ or the ___ ___ named ___.
charged, charged organic molecule, chlorine, ethanolamine, amino acid named serine
Phospholipid is a ___ molecule with a ___ and ___ end, forming the ___ ___ in cell membrane.
amphipathic, polar and non-polar (polar head/end: phosphate group, non polar tail/end: fatty acids), phospholipid bilayer
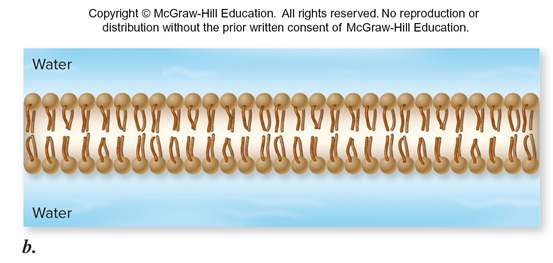
Phospholipids form a ___ ___ in water. In an aqueous environment, ___ ___ molecules ___ so their ___ heads are in the polar medium (___), while their ___ tails are held away from the ___. They can arrange themselves into two layers, known as ___.
lipid bilayer, amphipathic lipid molecules orient, hydrophilic, water hydrophobic, water, bilayer