polymers, amino acids, DNA, proteins
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
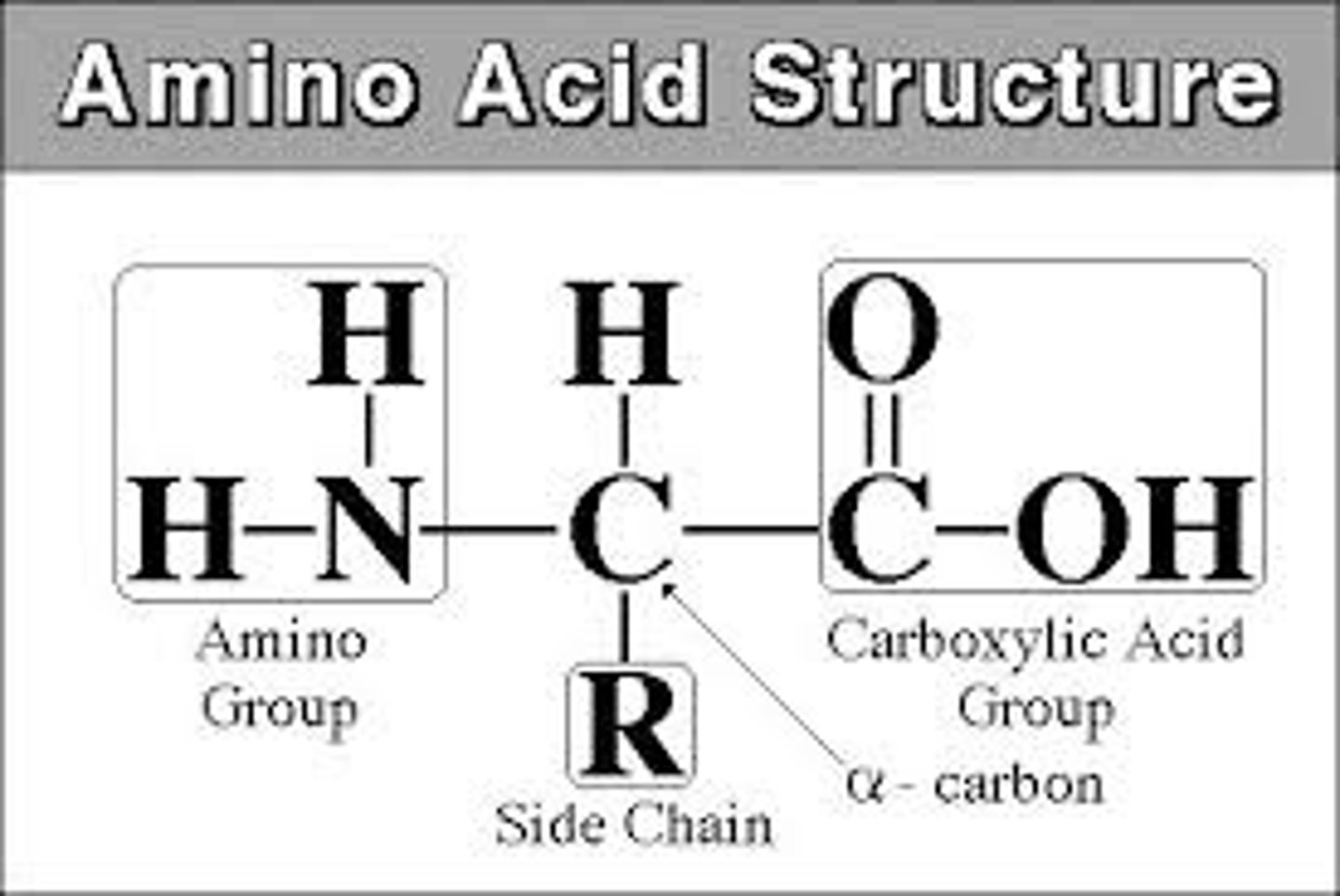
amino acid structure, general name for amino acids
2-amino carboxylic acids (name starts from the cooh group, nh2 is on second carbon)
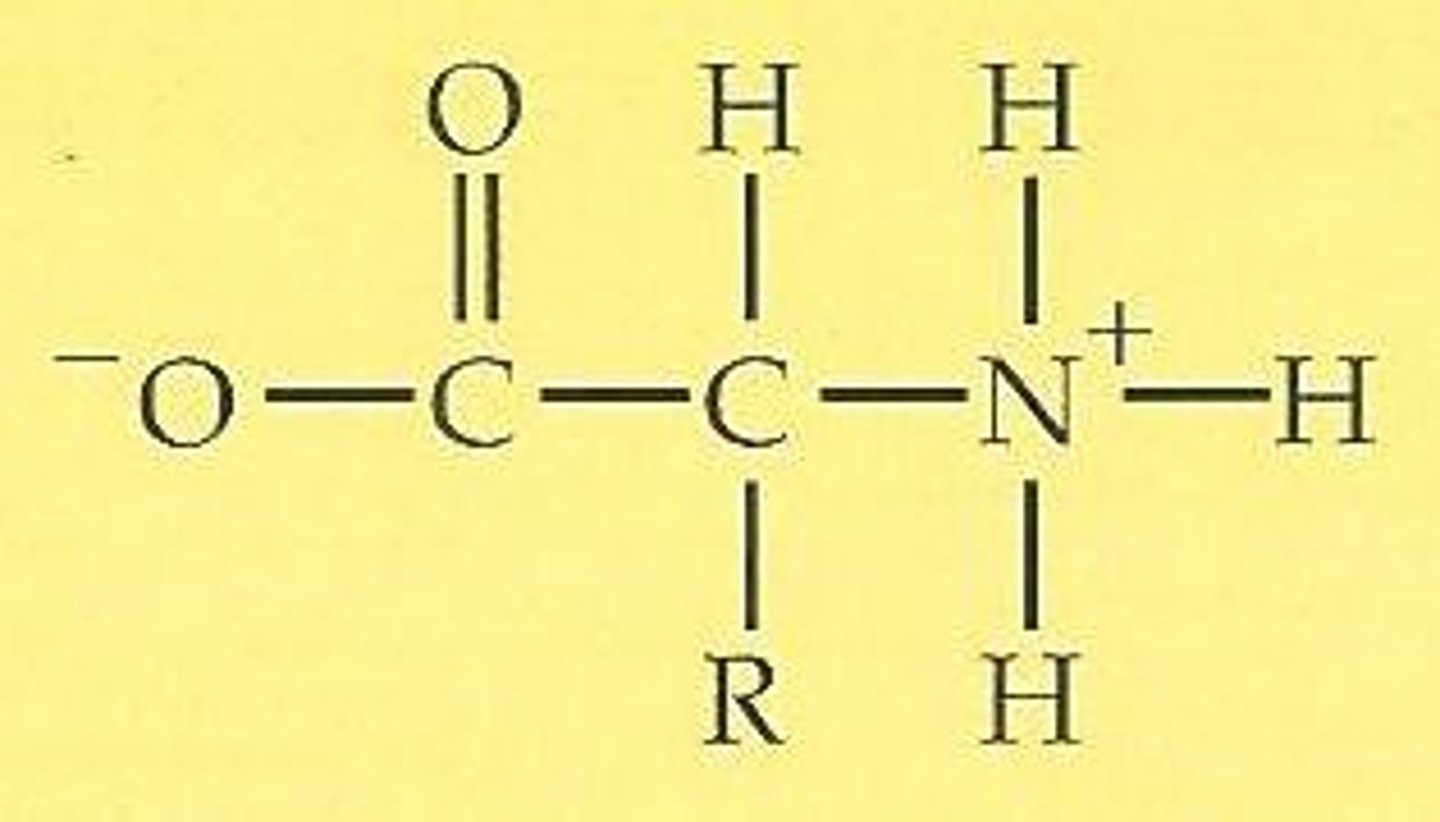
Zwitterions at intermediate pH and room temperature, what are they, overall charge, state etc
-positive (-NH3+) and negative charge (COO-) at physiological pH (overall neutral charge)
- solid at room temp because, even though overall charge zero, due to ionic bonding (form giant crystal lattices)
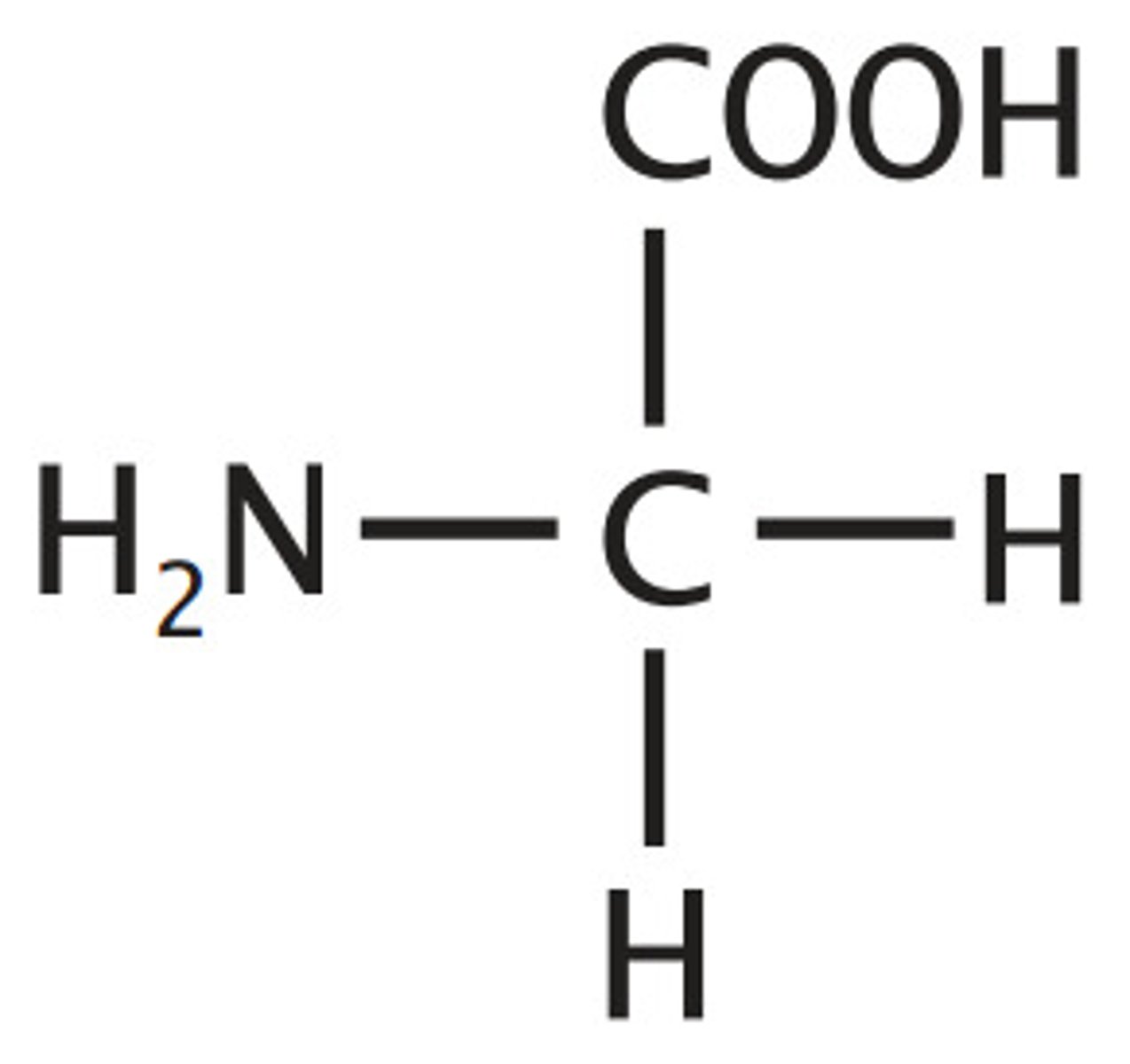
which amino acid doesnt contain a chiral carbon, and how can you tell
glycine
use plane polarised light, optical isomers rotate in opposite directions
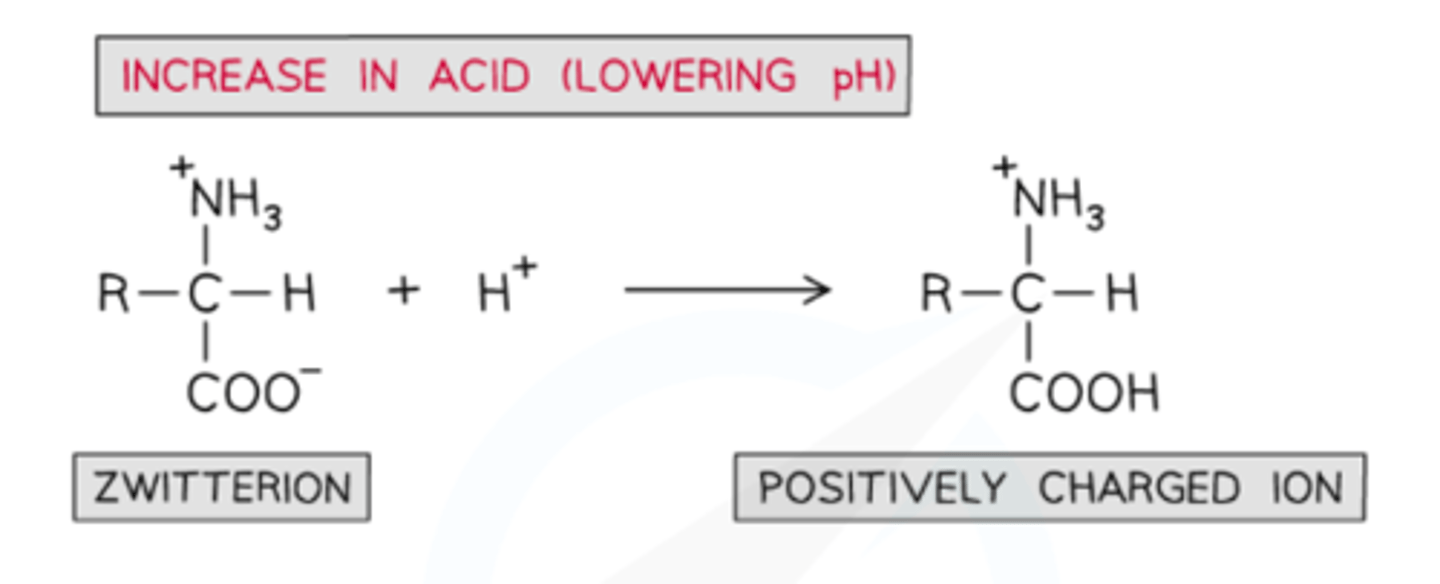
zwitterion at
zwitterion gets further protonated, just NH3+
in 'excess acid' any other NH2 in the R group also becomes NH3+
zwitterion at >pH 9, and what happens in 'excess alkali'
zwitterion gets further deprotonated, just COO-
in 'excess alkali', any other COOH groups in the R group also becomes COO-
how can amino acids react with other molecules (NOT includign polymerisation)
amino acids are both carboxylic acids and amines, so
as a carboxylic acid:
-react with alcohol to ester (conc H2SO4)
(any amino acids eg serine which have alcohol groups can react with THEMSELVES!! to form esters so cool)
-nucleophillic addition elimination (used as the thing being added onto)
as an amine:
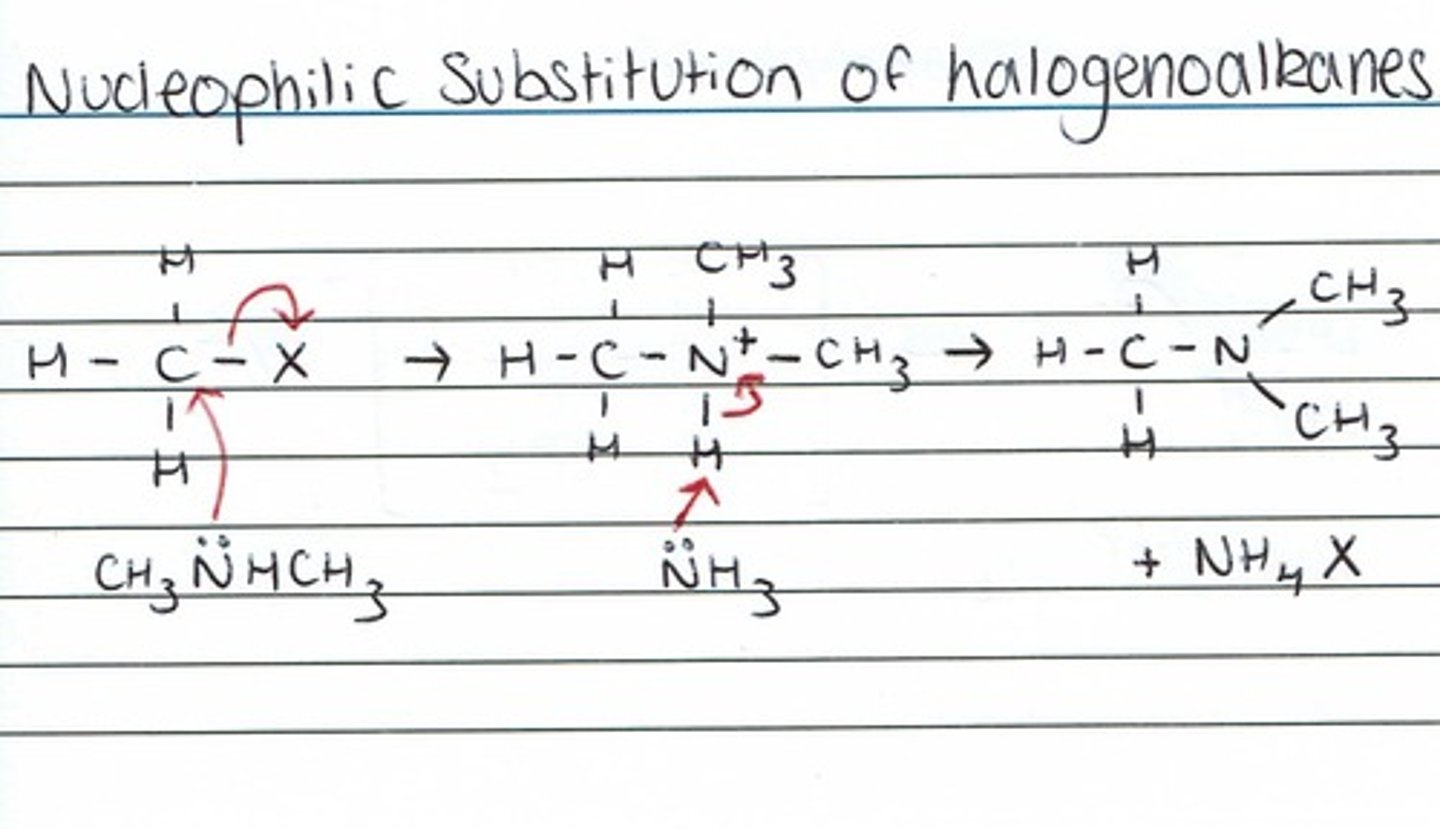
-react in nucleophillic substitution with halogenoalkanes, if haloalkane is in excess will produce a quarternary ammonium salt (cationic surfactant)
-nucleophillic addition elimination to form amide bonds (amine as nucleophile)
condensation polymerisation to form polypeptides
OH from COOH and H from NH2 form water, and these two groups form a peptide/amide bond
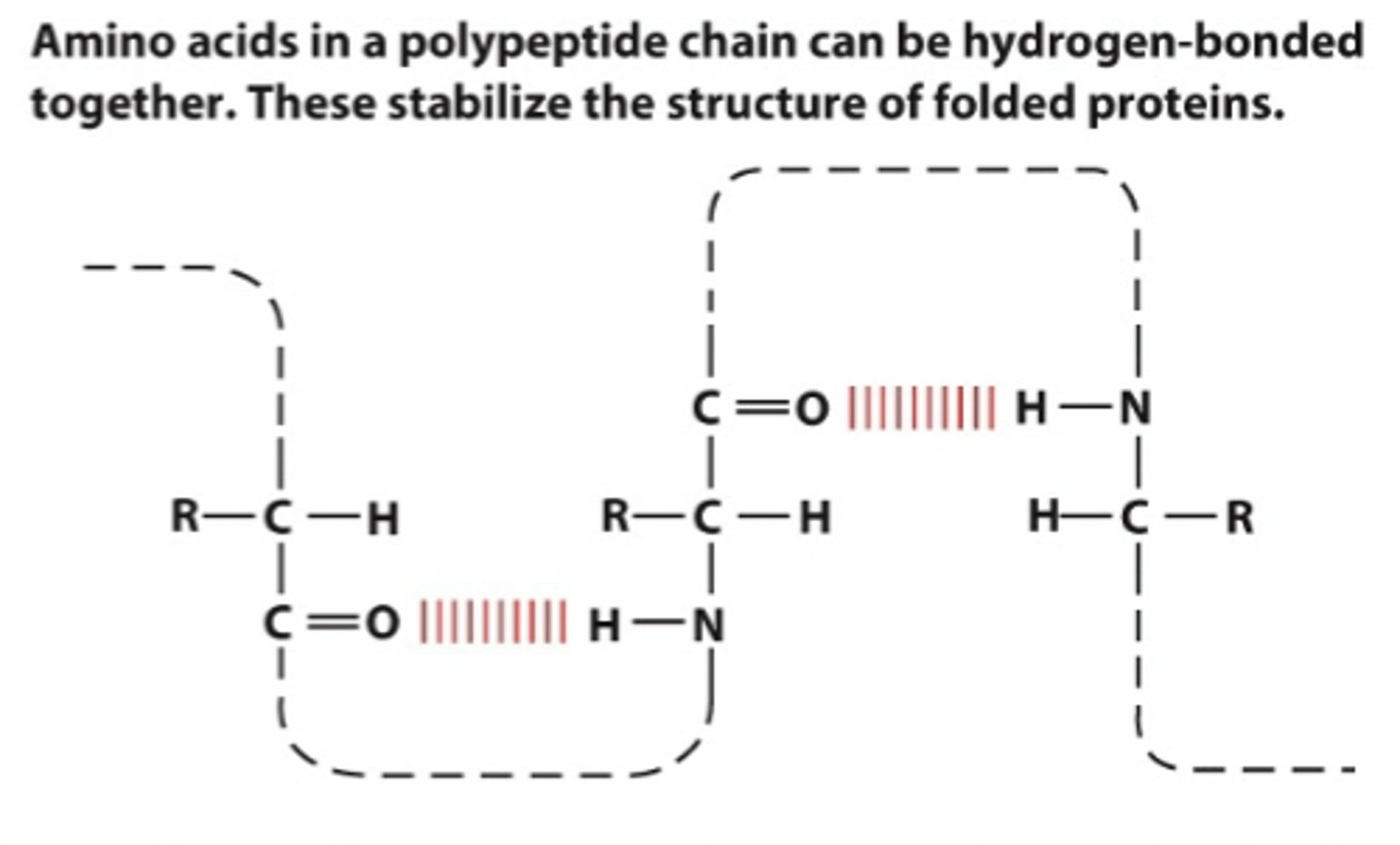
hydrogen bonds between amino acids
O in C=O has lone pair, attracted to the delta + H in the NH2
h bonds can also happen between R groups
what is a disulfide bridge
S-S (usually between two cysteines)
how do ionic bonds form in tertiary structure
They form between carboxyl and amino acid groups that are a R group. These ionise into NH3+ and COO-
alpha helix and beta sheets
secondary structure of proteins, held by h bonds
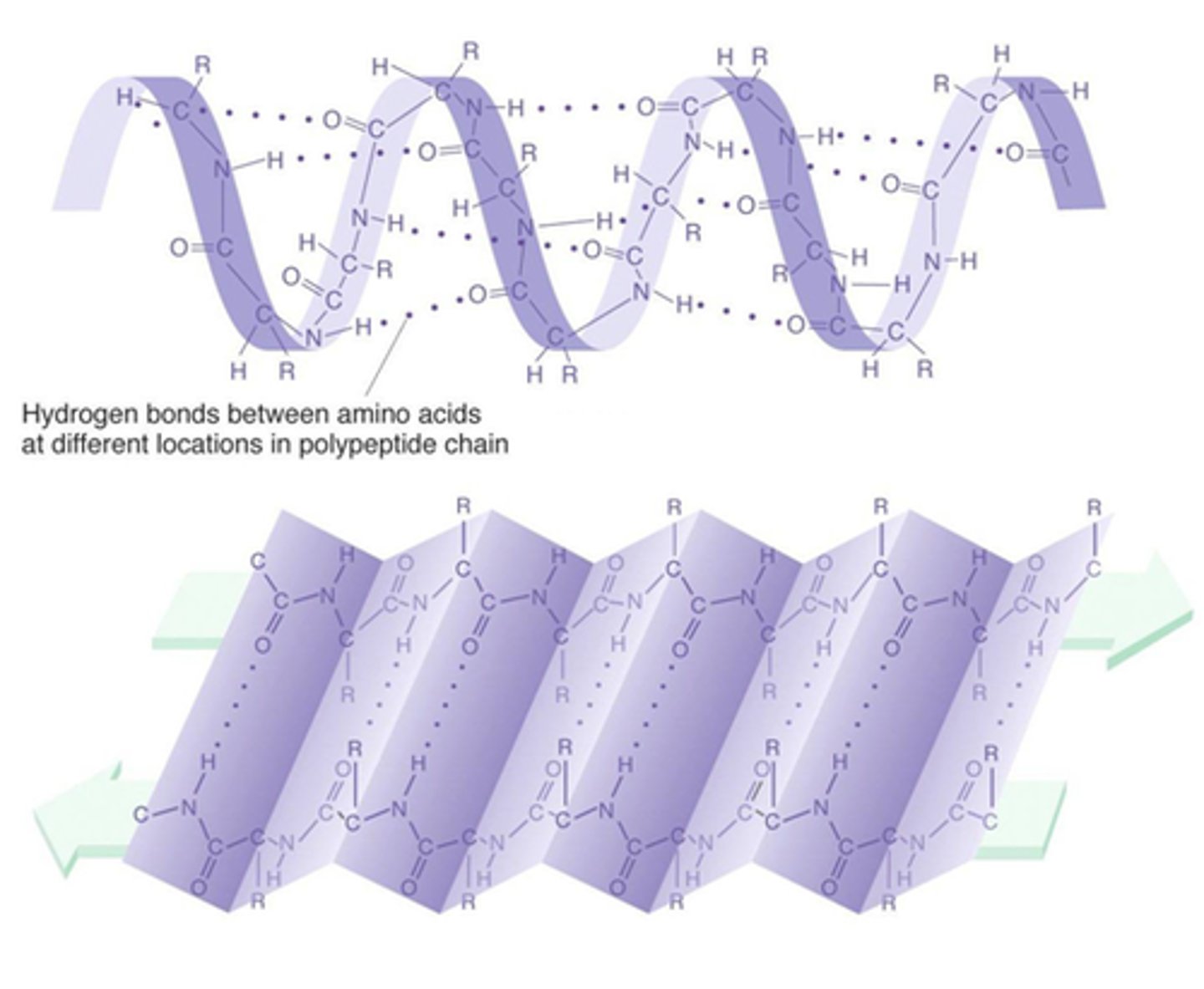
forces in tertiary and what alters them
VAN DER WAALS, ionic, h, disulfide
weird pH/ temperature denatures protein by unfolding them
active site and how they act with eantiomers
Enzymes are proteins. The action of enzymes as catalysts, including the concept of a STEREOSPECIFIC active site that binds to a substrate molecule
only stereospecific to ONE eantiomer in a pair
how do drugs work to prevent E-S complexes forming and what helps deisgn these
-act as competiive inhibitor, bind to active site by having a similar shape so that the actual substrate cant
-computers help to design drug+ ensur
hydrolysis of peptide bond, conditions, catalyst, products
concentrated HCl, high temperature+ under reflux, forms constituent amino acids
TLC method to identify amino acids
1. draw a line in pencil near the bottom of the TLC plate
2. using a capillary tube place a drop of each mixture on the line, allow the spots to dry and do more spots to increase concentration, then allow to dry
3. place the plate into a beaker with a small volume of solvent (mobile phase), with a lid to prevent evaporation
4. solvent level must be below the baseline so it does not dissolve the samples
5.leave the beaker until the solvent has moved almost to the top, carrying pigments
6. remove from the beaker before solvent reaches the top
7. draw a pencil line to mark solvent front (before it evaporates)
8. place paper in fume cupboard to dry, preventing toxic/flammable fumes escaping into the room
9. place under ninhydrin/ UV to make amino acids visible, and calculate Rf values of the amino acids, and compare to KNOWN VALUES
safety measures of TLC method (2)
- wear plastic gloves to prevent contamination
- work in a fume cupboard as solvent may be toxic
why do amino acids have different Rf values?
amino acids have different R groups, so different polarities
this means that those that are more polar will have more attraction/ affinity for polar solvent and vica versa
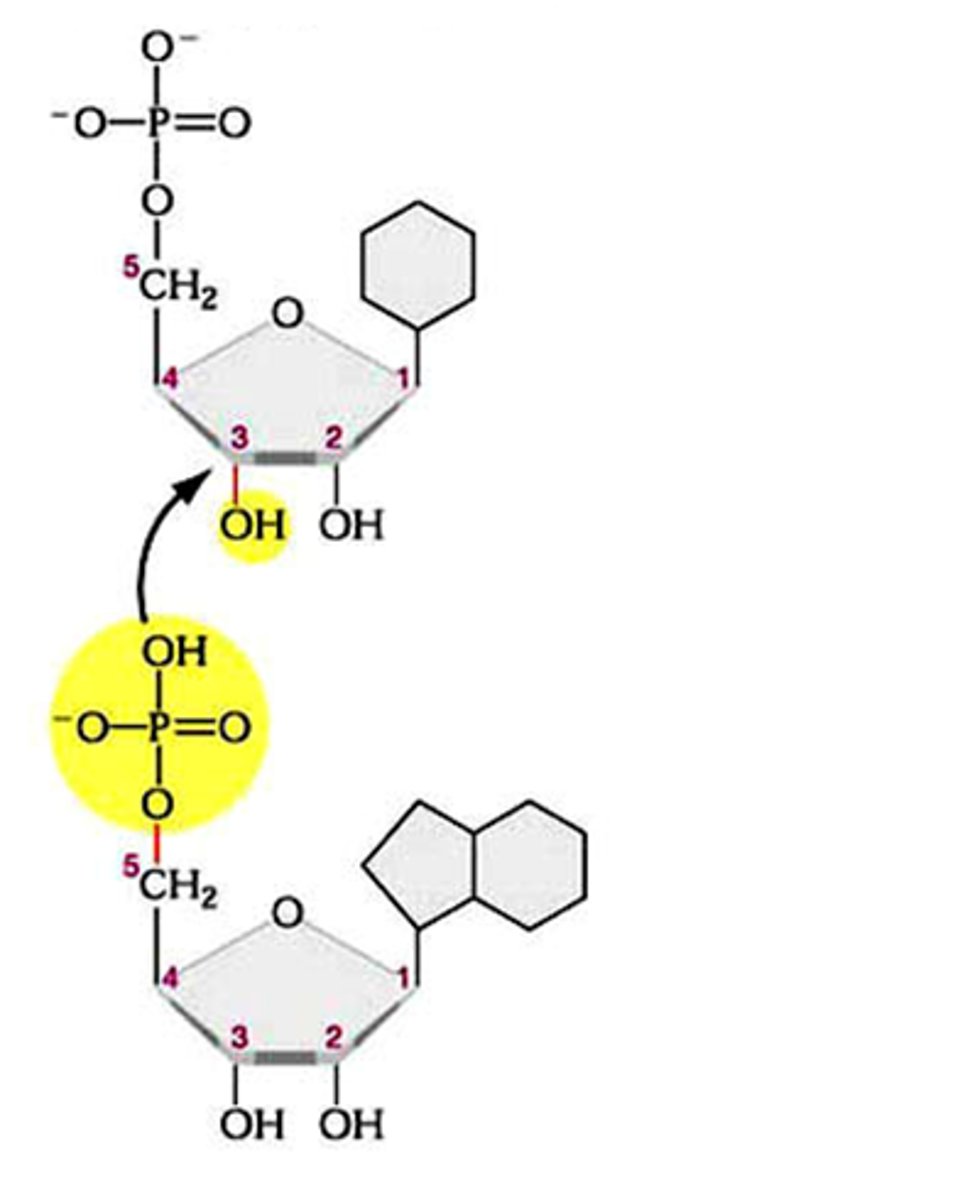
phosphodiester bond
the type of bond that links the nucleotides in DNA or RNA. joins the phosphate group of one nucleotide to the hydroxyl group on the sugar of another nucleotide in condensation
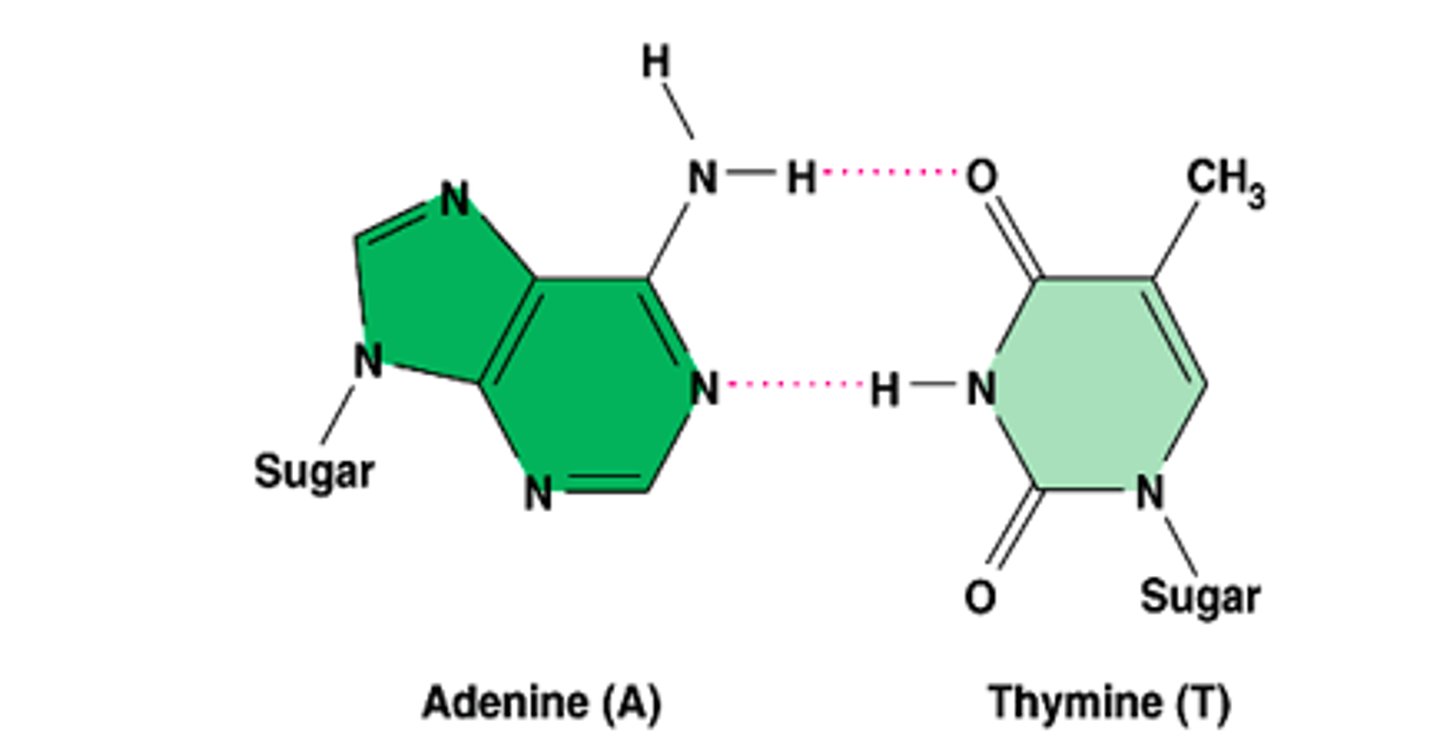
hydrogen bonds between A and T
NH2 bonds to O on C=O
NH bonds to N
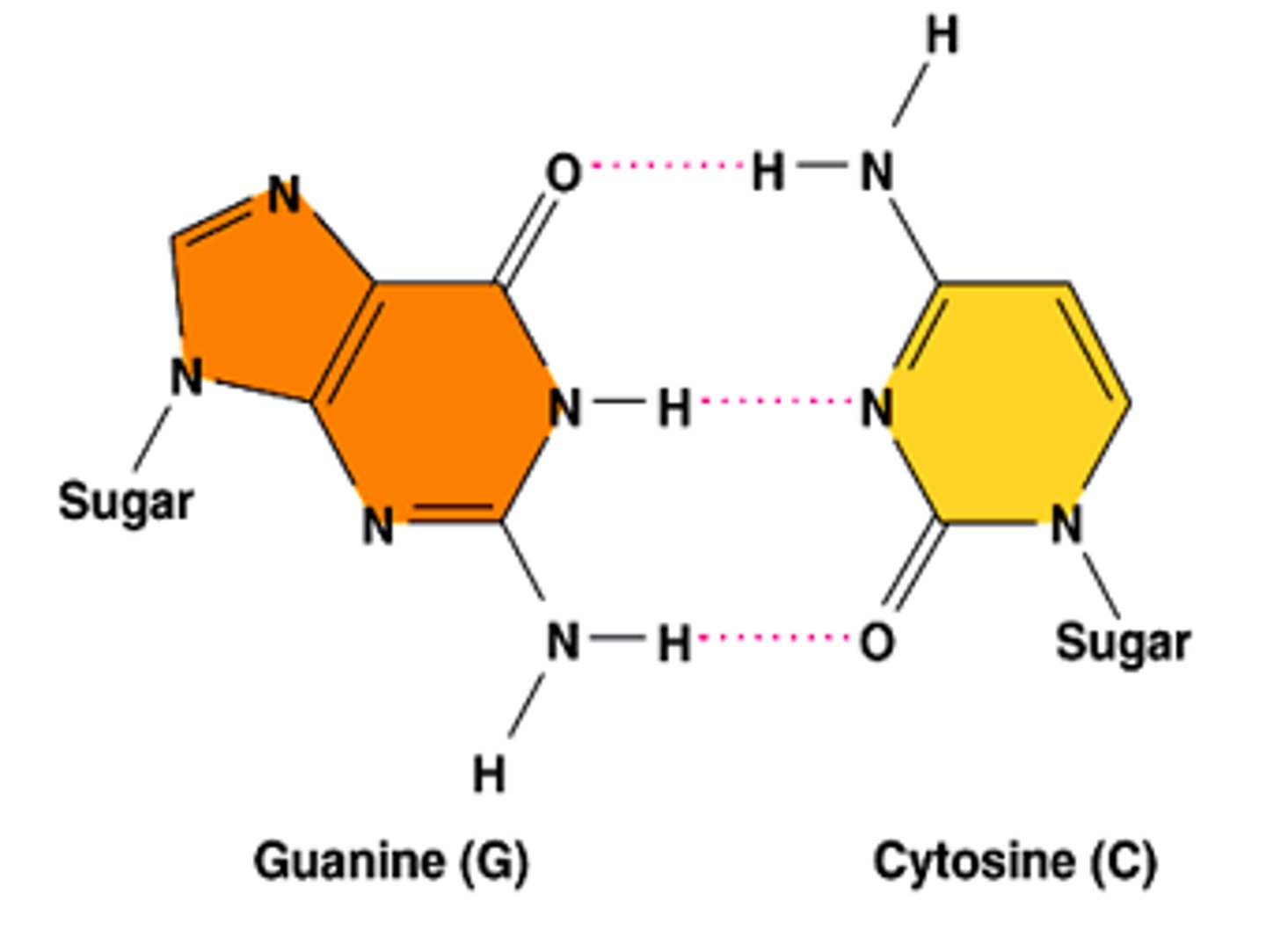
hydrogen bonds between C and G
2x NH2 bonds to O on C=O
NH bonds to N
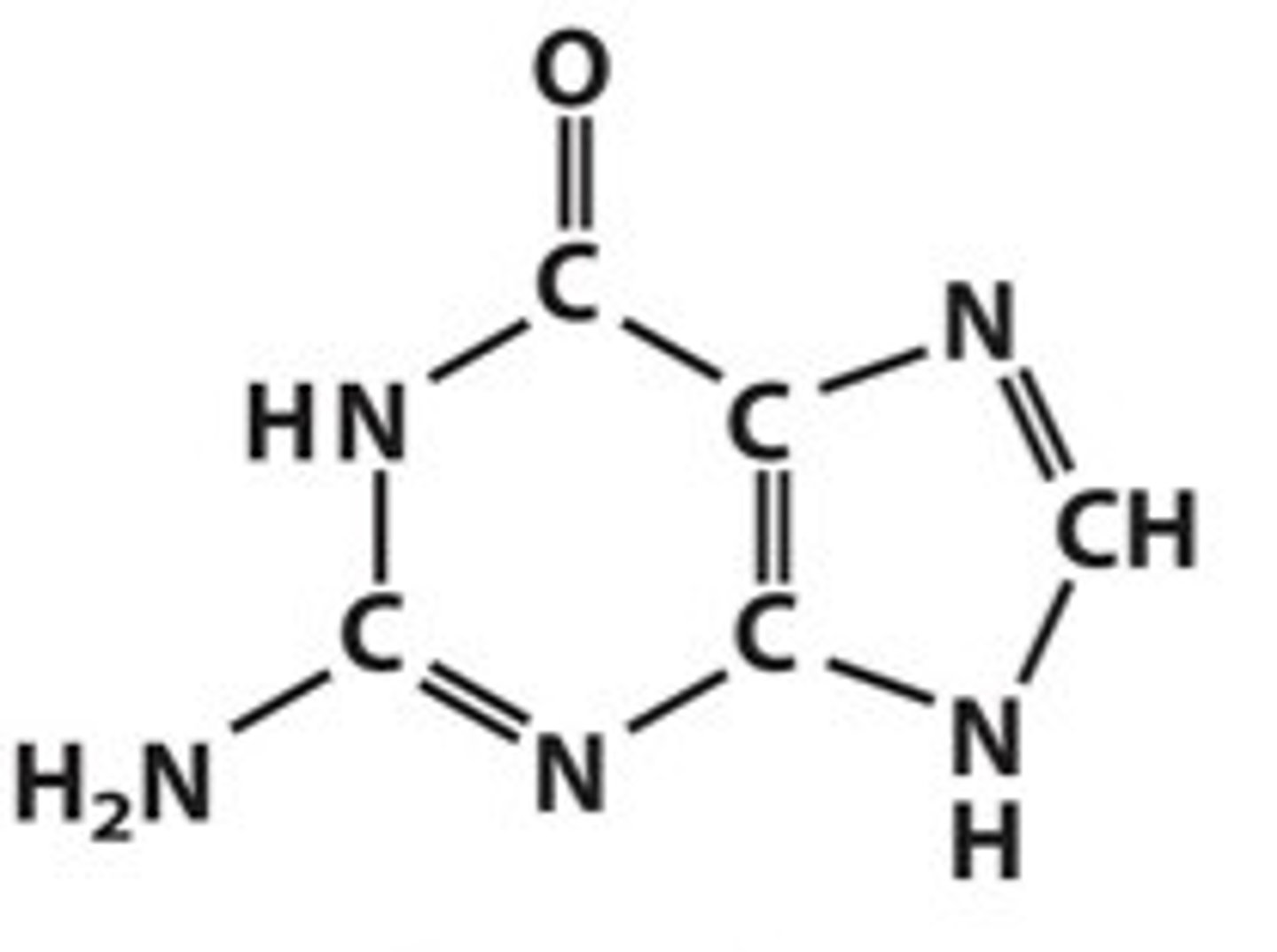
how does cisplatin act as an anti cancer drug, and what are its negatives
1) one of its Cl- ligand is replaced by water
2) the remaining h2O and Cl- ligands each form a coordinate bond with the nitrogen on the double bond in guanine in the SMALLER ring
3) this stops the DNA from replicating uncontrollably
-toxic and has horrible side effects, so can only be used in small amounts to make sure the benefits outweigh the risks
condensation polymer
ong polymer chains through the joining of monomers, accompanied by the elimination of small molecules like water (H2O) or hydrogen chloride (HCl) (if an acyl chloride is used)
Common types of condensation polymers are:
Polyesters
Polyamides
Polypeptides
Polyesters
condensation polymer formed from the reaction between dicarboxylic acids (or derivatives such as acyl chlorides) and diols. The carboxyl (-COOH) and hydroxyl (-OH) groups react
forms water as byproduct
Polyamides
formed from dicarboxylic acids (or derivatives such as acyl chlorides) and diamines.
The carboxyl and amine groups react, forming amide links
can form H2O if with COOH or HCL if with COCL
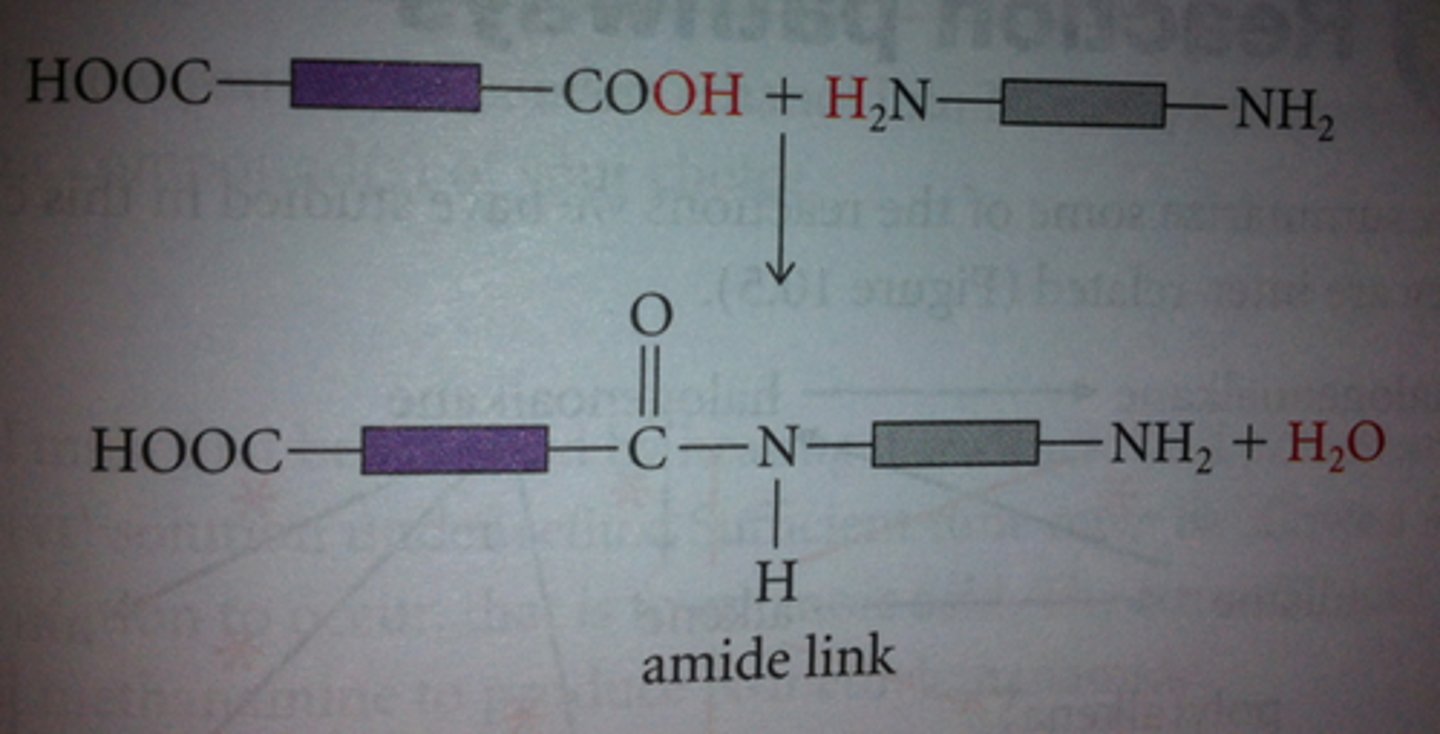
naming polyamides
nylon-x,y
x = number of carbons in the diamine
y = number of carbons in the dicarboxylic acid
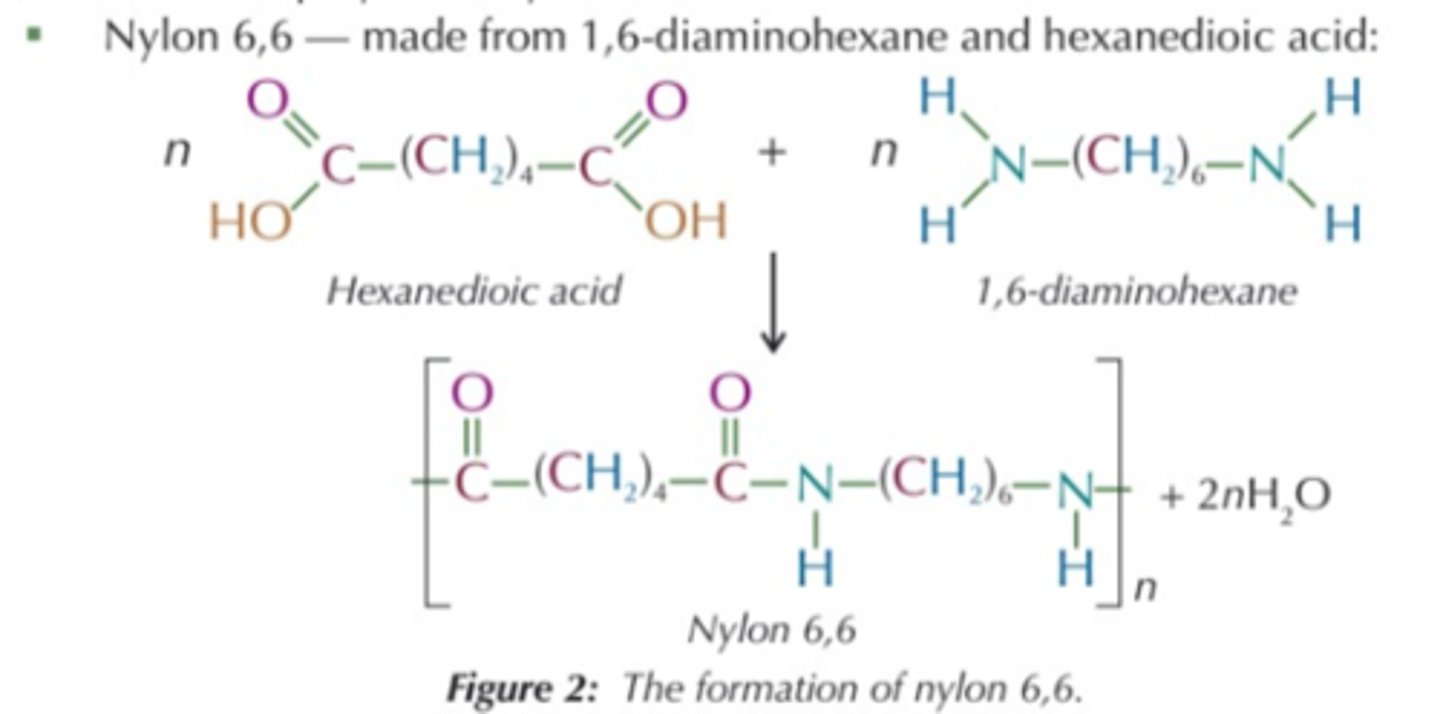
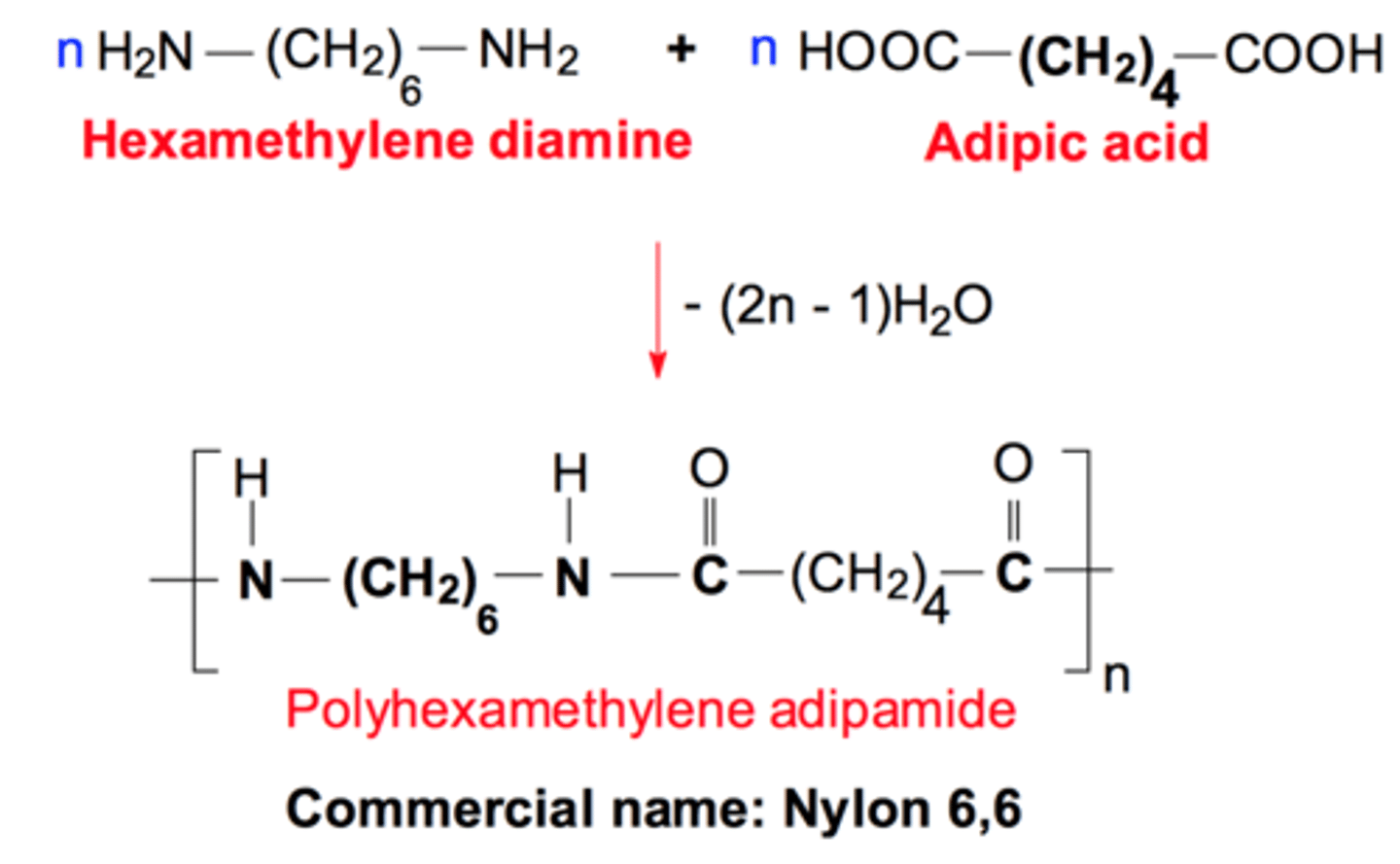
Nylon 6,6
1,6-diaminohexane and hexane-1,6-dicarboxylic acid
used for fibres in clothing, ropes and parachutes
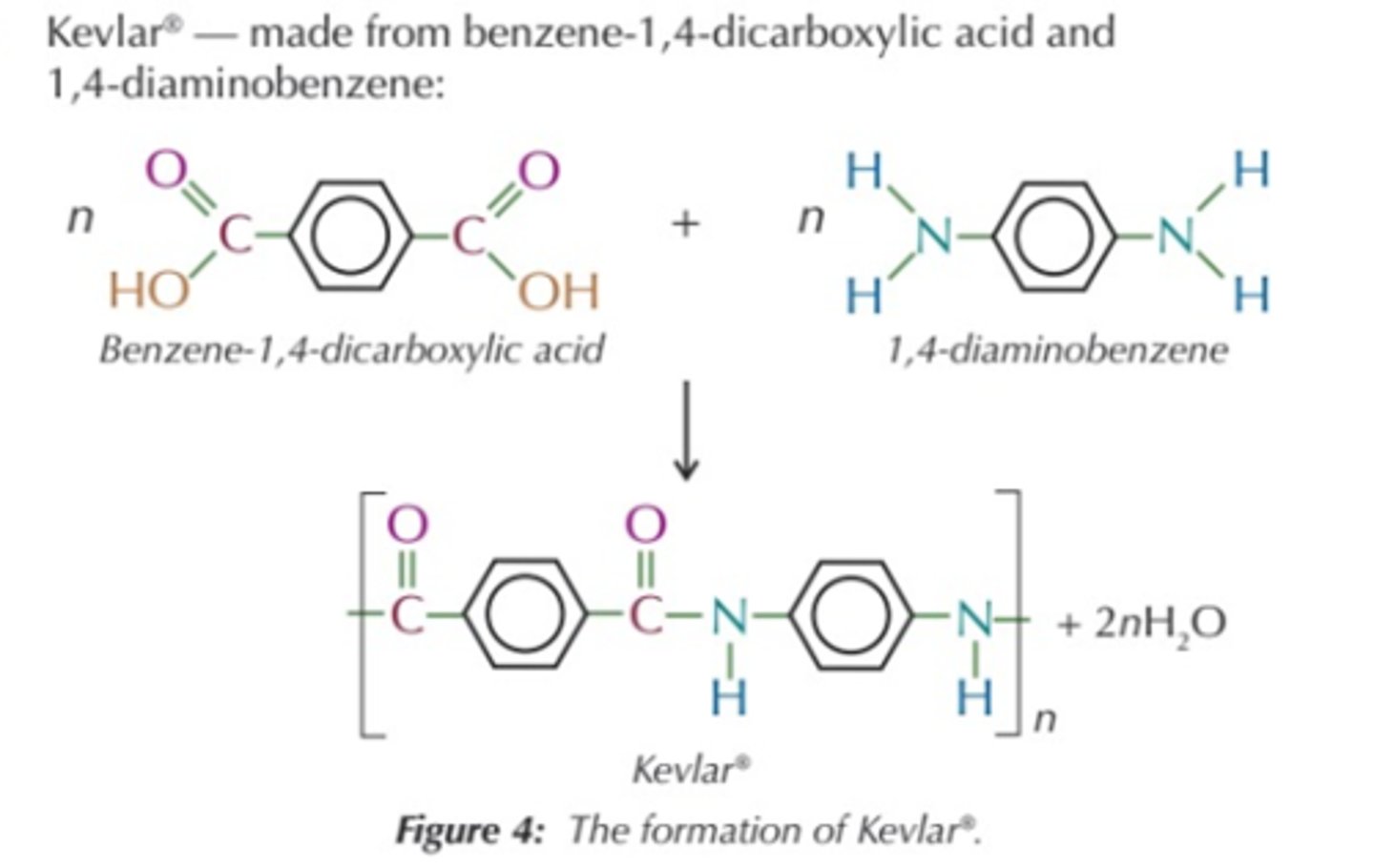
kevlar
benzene-1,4-dicarboxylic acid and 1,4-diaminobenzene
(basically a diamine with the middle being just a benzene, and a dicarboxylic acid with a benzene in the middle)
Kevlar is used for bulletproof vests and sports equipment.
Polypeptides
ondensation polymers formed from amino acids. The carboxyl group of one amino acid reacts with the amine group of another, forming a peptide (amide) link.
bonding in condensation polymers and how this determines properties
intermolecular forces:
Permanent dipole-dipole forces between polarised groups.
Hydrogen bonding between C=O, N-H, and O-H groups.
The presence of stronger intermolecular forces makes condensation polymers:
More rigid and less flexible.
Stronger, with higher melting and boiling points compared to addition polymers.
disposability of each of the 3 condensation polymers and why
polyalkenes non biodegradable as they are non-polar, saturated hydrocarbons
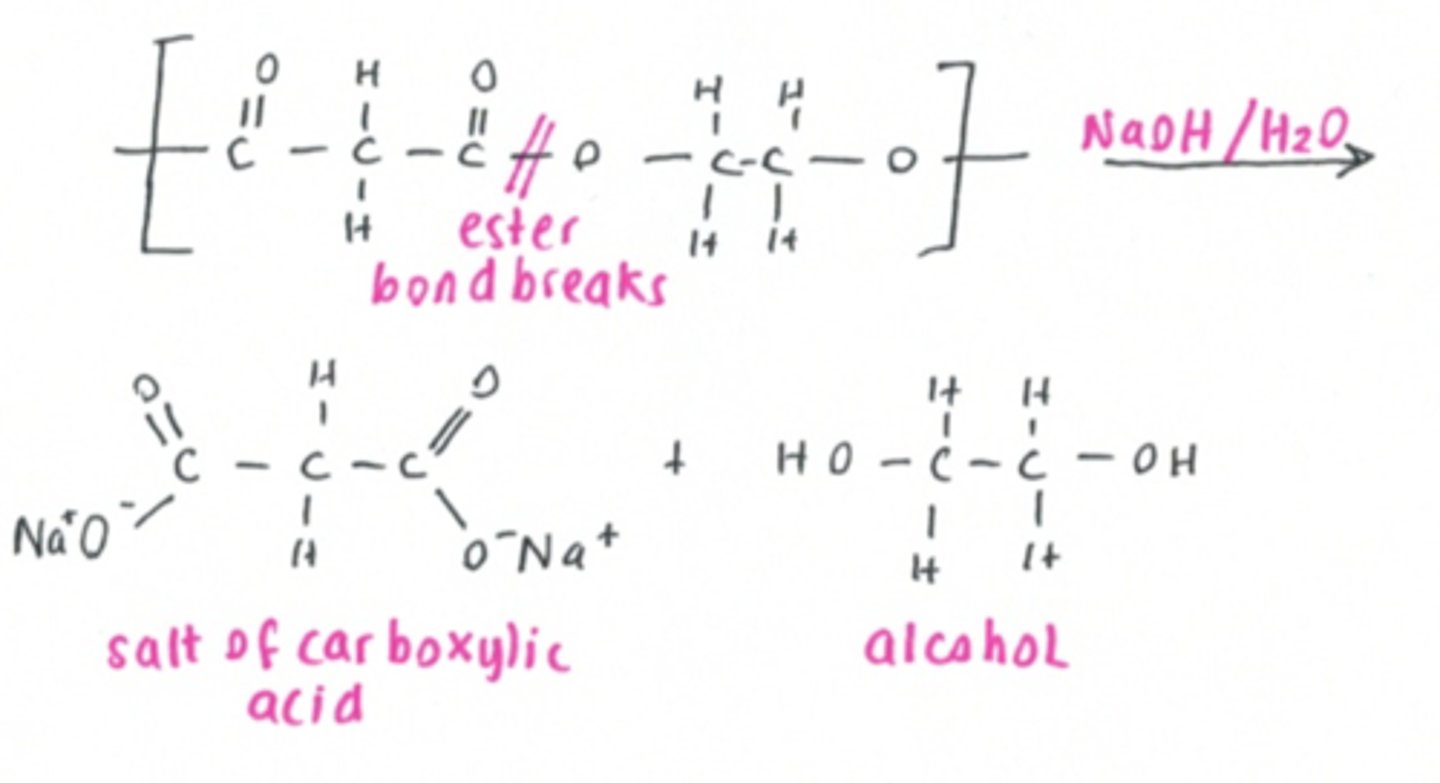
Polyesters and polyamides undergo hydrolysis- makes them potentially biodegradable ( very slow )
The amide bonds can be hydrolysed, breaking the polymer chain into the original dicarboxylic acid and diamine monomers.
landfill pros and cons
nvolves burying plastic waste alongside other types of rubbish
+cheap and quick
-emit methane, a potent greenhouse gas as waste decomposes
why are disulfide bridges stronger than hydogen bonds
disulfide bridges are covalent bonds
educe the type of interaction that occurs between a lysine R group and an aspartic acid R group (NH2 AND COOH)
ionic bonds
PVC and TERYLENE
PVC is poly(chloroethene)
terylene is an ester