Tricarboxylic Acid (TCA) Cycle
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Tricarboxylic acid (TCA) Cycle / Citrate Cycle
“hub” of cellular metabolism; captures energy using redox reactions
Functions of TCA cycle
Primary function: To oxidize acetyl-CoA
1. Generates the bulk of NADH and FADH2, which are used to produce ATP by oxidative phosphorylation
2. Links the oxidation of metabolic fuels (carbohydrates, fatty acids, proteins) to ATP through shared intermediates
3. It provides metabolites for other biosynthetic pathways (fatty acids, amino acids, hemes)
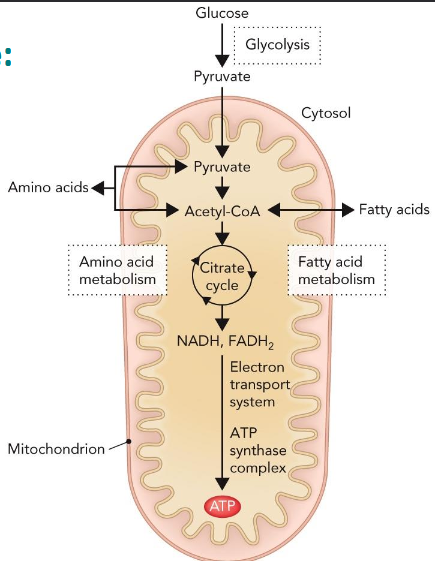
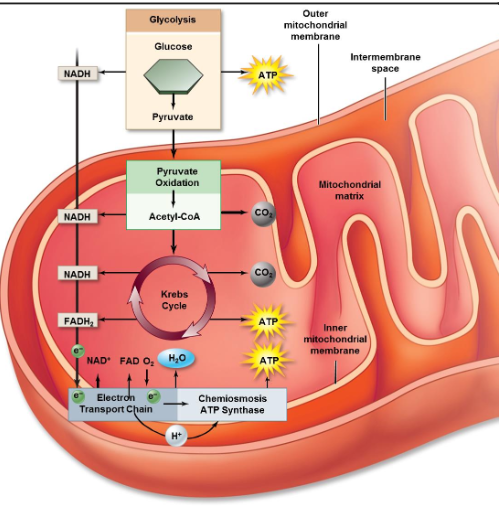
Metabolic pathways compartmentalization
Glycolysis takes place in the cytoplasm
Citric acid cycle takes place in the mitochondrial matrix
Oxidative phosphorylation takes place along inner mitochondrial membrane
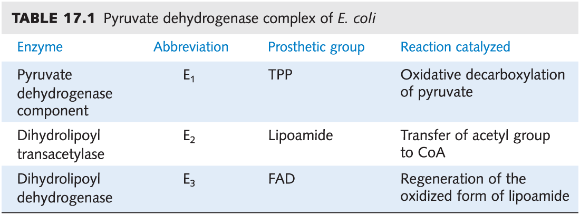
pyruvate dehydrogenase
Pyruvate + CoA + NAD = Acetyl-CoA + CO2 + NADH + H+
ΔG° = -33.4 kJ/mol
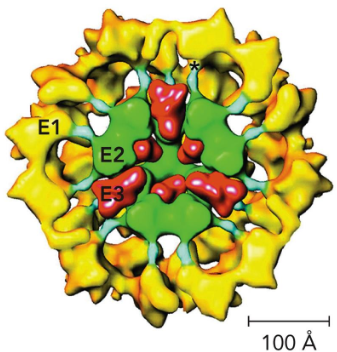
Contains 3 distinct catalytic enzymes (E1-E3)
Uses 5 different cofactors or co-enzymes
Essentially irreversible (commits pyruvate to aerobic respiration)!
pyruvate dehydrogenase complex
pyruvate dehydrogenase (E1) - decarboxylation w/ TPP
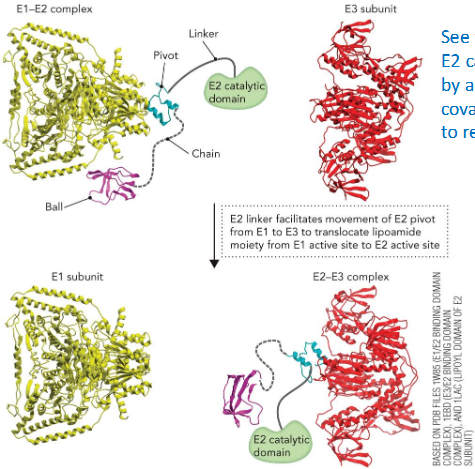
dihydrolipoyl transacetylase (E2) - transfer to CoA
dihydrolipoyl dehydrogenase (E3) - oxidation of lipoamide
Why use a complex?
Catalytic sites are close to one another, which allows channeling of substrates
Avoids side reactions with intermediates
Facilitates coordinated regulation of activity of different subunits
Pyruvate dehydrogenase steps
Decarboxylation - attaches to TPP cofactor, lose CO2!
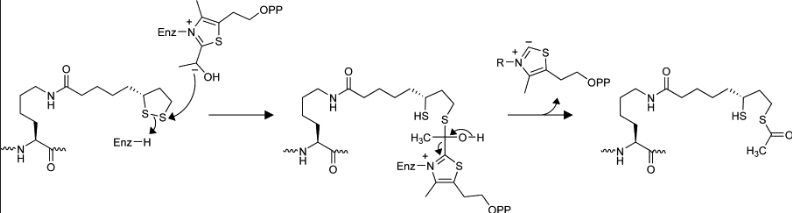
Activation of Ac - thioester formed; hydroxyethyl group is oxidized/ reacts with disulfide of lipoamide attached to Lys of E2, which acts as the oxidant and is itself reduced
Transthioesterification - Acetyl-CoA production; occurs in active site of E2 with CoA; dihydrolipoamide is now fully reduced
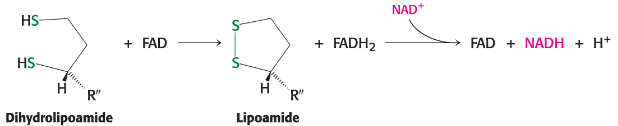
Oxidation of dihydrolipoamide - transfers 2e-, 2 H+ to E3 disulfide, which then gets re-oxidized and reduces FAD to FADH2
Oxidation of FADH2 to FAD by NAD+ to produce NADH + H+ - regenerates FAD
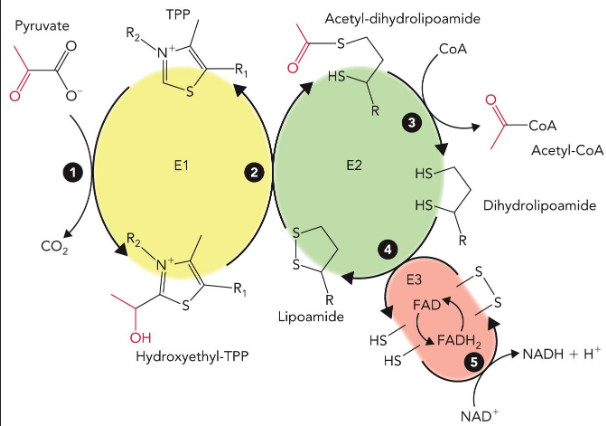
Net Reaction of one turn of TCA cycle
Acetyl-CoA + 3 NAD+ + FAD + GDP + Pi + 2 H2O → CoA + 2 CO2 + 3 NADH + 3 H+ + FADH2 + GTP
TCA cycle electron transfer
Each “turn” of citrate cycle produces 4 pairs of e- (8 e-)
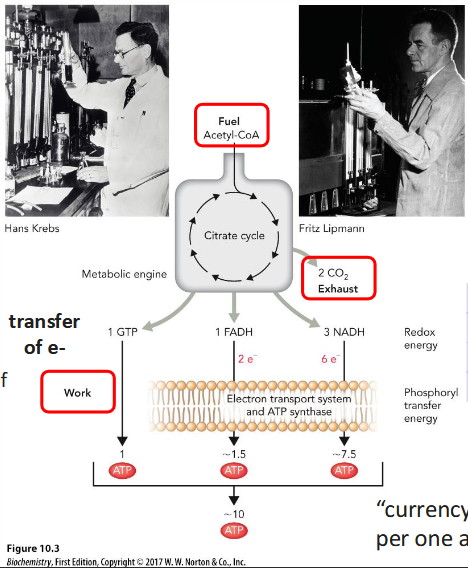
History of TCA cycle
Krebs described the cycle in 1937.
Krebs and Henseleit also discovered urea cycle (urea synthesized by amino acids and ammonia)
Lipmann discovered role of acetyl-CoA in metabolism (early 1940s)
Krebs and Lipmann share 1953 Nobel Prize in Physiology.

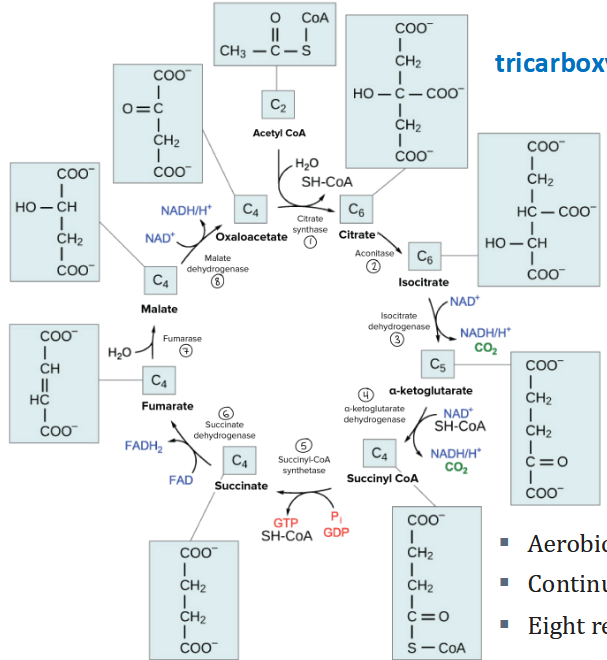
TCA Cycle steps
Aerobic pathway
Continuous (cyclic)
Eight reactions
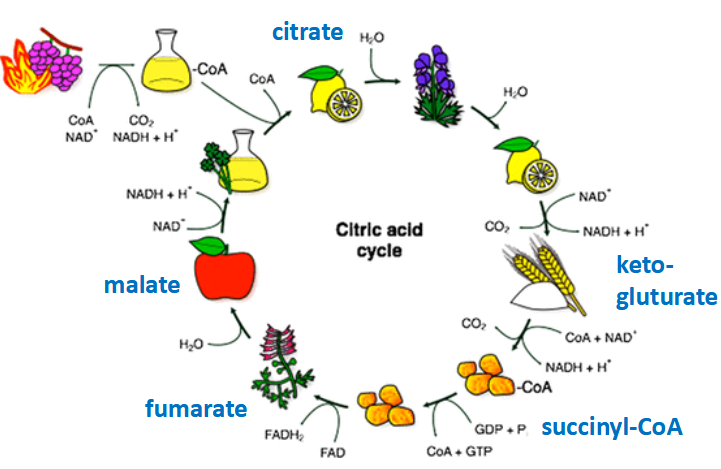
Naming the cycle
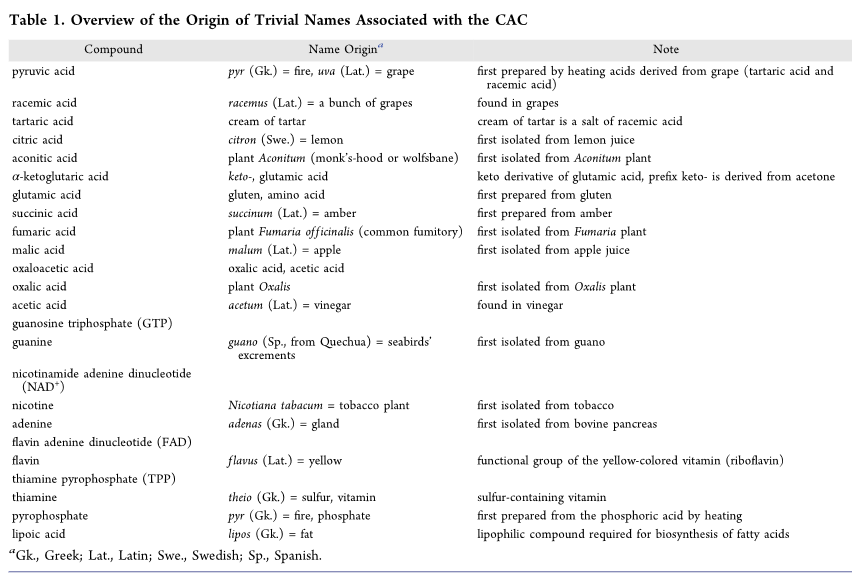
TCA cycle products and carbon backbones
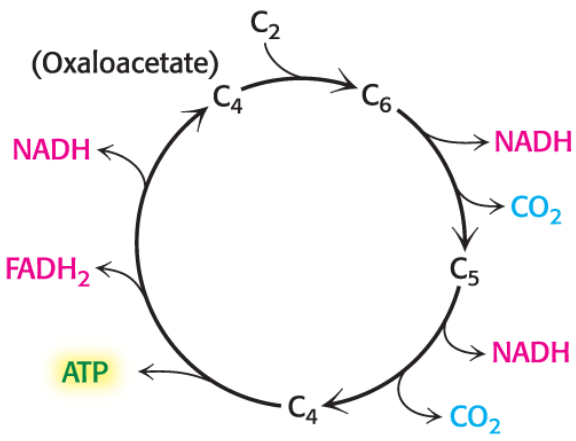
Lipoamide
extracts the acetyl group from TPP
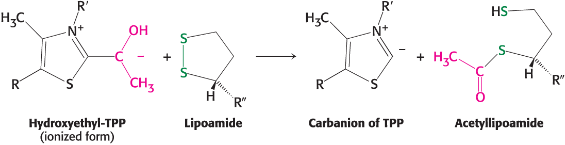
Lipoamide transfer mechanism
Lipoamide regeneration
regenerated by oxidation with FAD, which transfers e- to NAD+
dihydrolipoyl transacetylase “ball and chain” mechanism
arsenite
dihydrolipoyl transacetylase inhibitor; covalent binding to reduced lipoamide
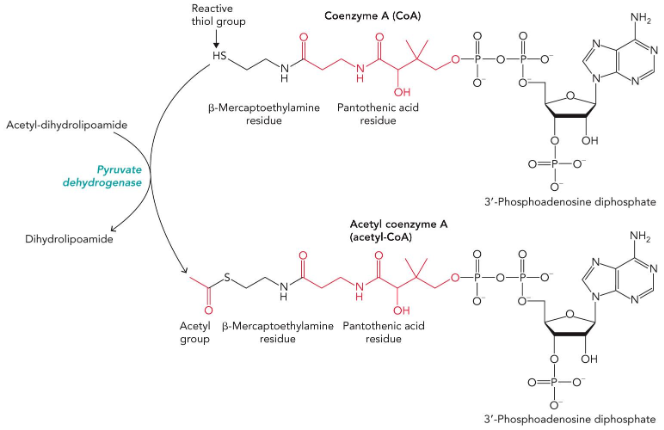
Coenzyme A (CoA)
common “acyl carrier compound”; half deprotonated at physiological pH
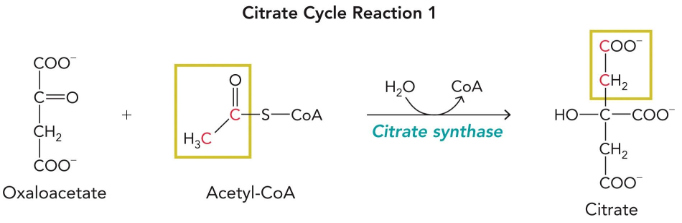
Citrate synthase
OAA + aectyl-CoA + H2O → citrate + CoA
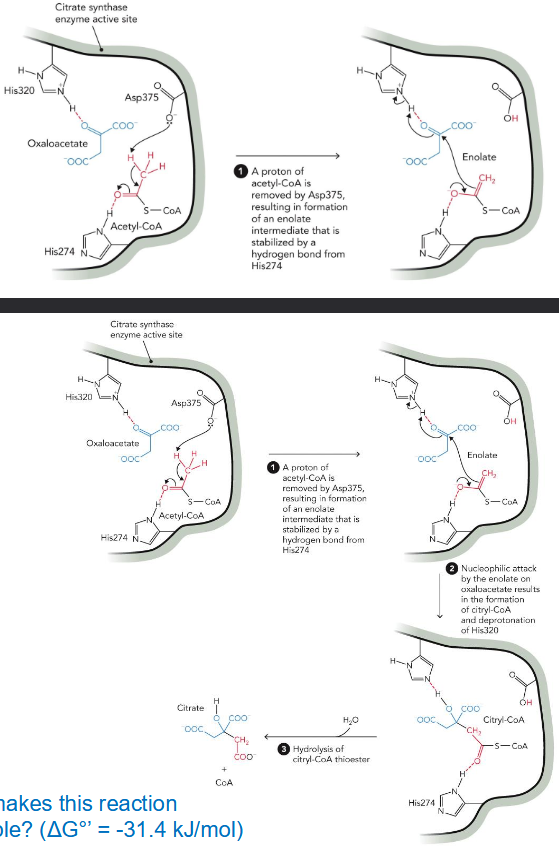
Why is citrate synthase reaction so favorable? (ΔG°’ = -31.4 kJ/mol)
CoA release - breaking thioester bond
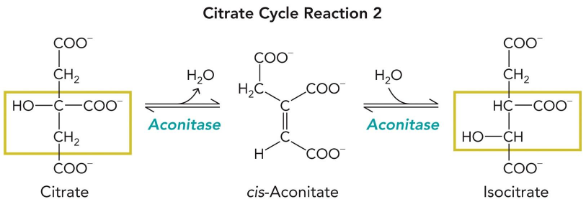
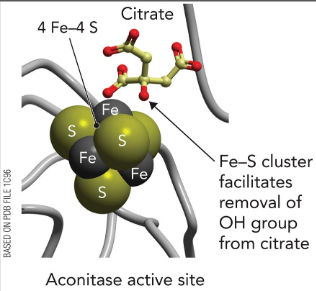
aconitase
Fe-S cluster facilitates removal of OH group from citrate
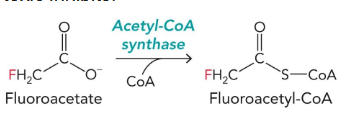
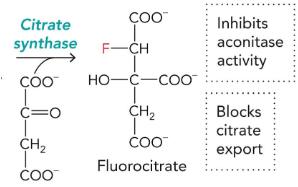
fluoroacetyl-CoA
can be turned into flurocitrate → inhibits aconiase activity and blocks citrate export
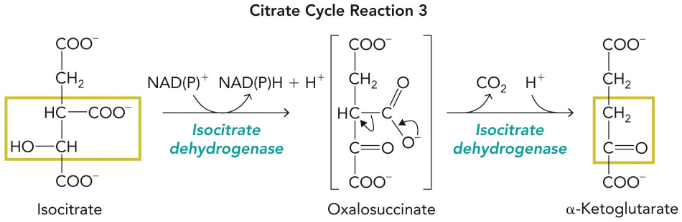
isocitrate dehydrogenase
first oxidative decarboxylation of the cycle (and first production of NADH)
oxidation helps stabilise decraboxylation
alpha-ketoglutarate dehydrogenase complex
Second oxidative decarboxylation of the cycle, which also produces NADH
similar to pyruvate dehydrogenase
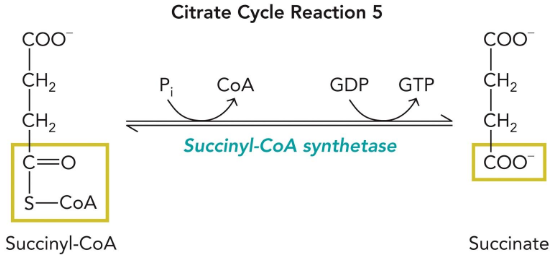
succinyl-CoA synthetase
Goes through high energy intermediates (succinyl-phosphate and phospho-His)
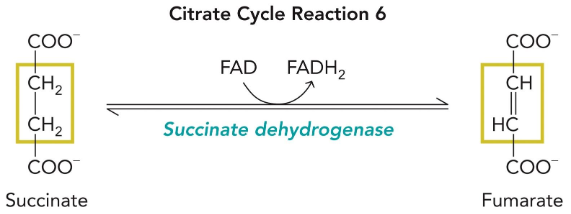
succinate dehydrogenase
This is complex II in the ETC (direct link!)
Produces FADH2 (used directly in ETC)
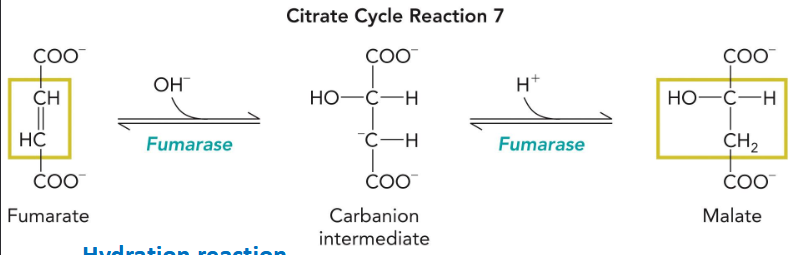
fumarase
hydration rxn
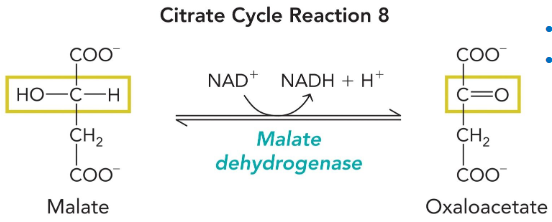
malate dehydrogenase
Redox reaction
Produces final NADH!
decarboxylation reactions
lead from 6-carbons to 4-carbon molecules
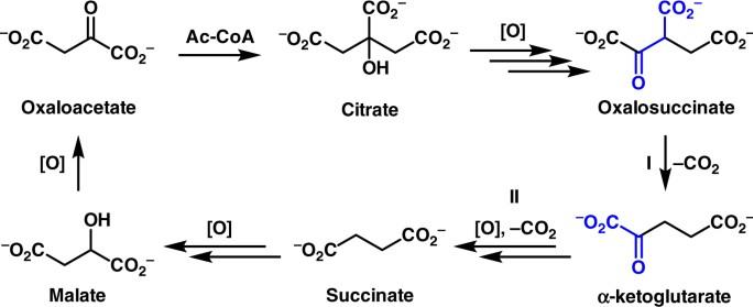
biochemical standard free energy changes for TCA reactions
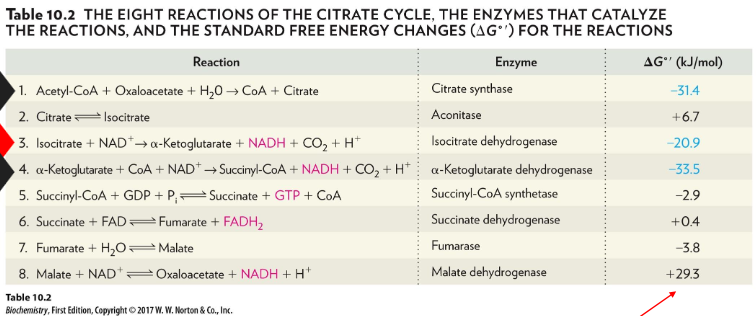
Why is this high ΔG°′ tolerated?
citrate synthase very favourable + low concentrations of OAA
TCA cycle regulation points
pyruvate dehydrogenase
pyruvate carboxylase
citrate synthase
isocitrate dehydrogenase
alpha-ketoglutarate dehydrogenase
TCA cycle regulation
Product inhibition by acetyl-CoA, NADH, ATP, other carbon products
excepy pyruvate carboxylase, which is allosterically upregulated by acetyl-CoA
ADP regulation
can activate enzymes by relieving allosteric inhibition by ATP
Ca2+ regulation
signals need for ATP during muscle contraction
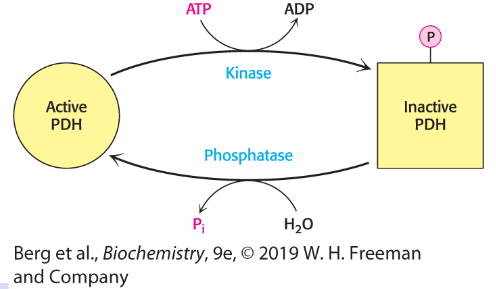
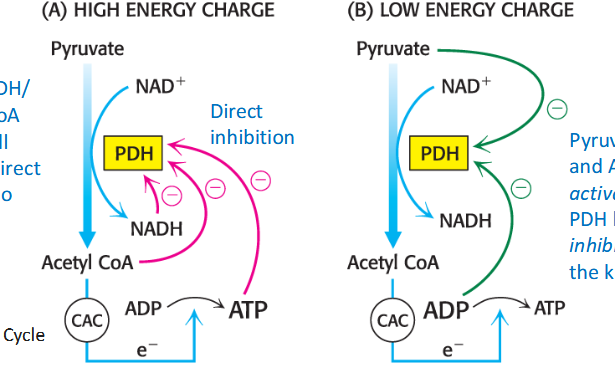
Regulation of pyruvate dehydrogenase (PDH) by PTM
regulated by phosphorylation; relates to cellular energy charge
E1 is phosphorylated by KDH kinase/ dephosphorylated by the phosphatase
Kinase is activated by NADH, acetyl-CoA, ATP to block PDH function; NAD+/CoA/ADP inhibit kinase