3.1.1.3 electronic configuration
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
explain how electrons are arranged?
in principal energy levels (electron shells) which themselves have sub-levels (sub-shells)
what letters are the sub-levels assigned?
s, p, d + f
what sub-levels do each of the principal energy levels contain?

describe the energy of the sub-levels
each of the sub-levels has a different energy
the energy of them increases from s → p → d → f
give the order of increasing energy in the sub-levels
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s…
a. what are orbitals?
b. how are orbitals represented?
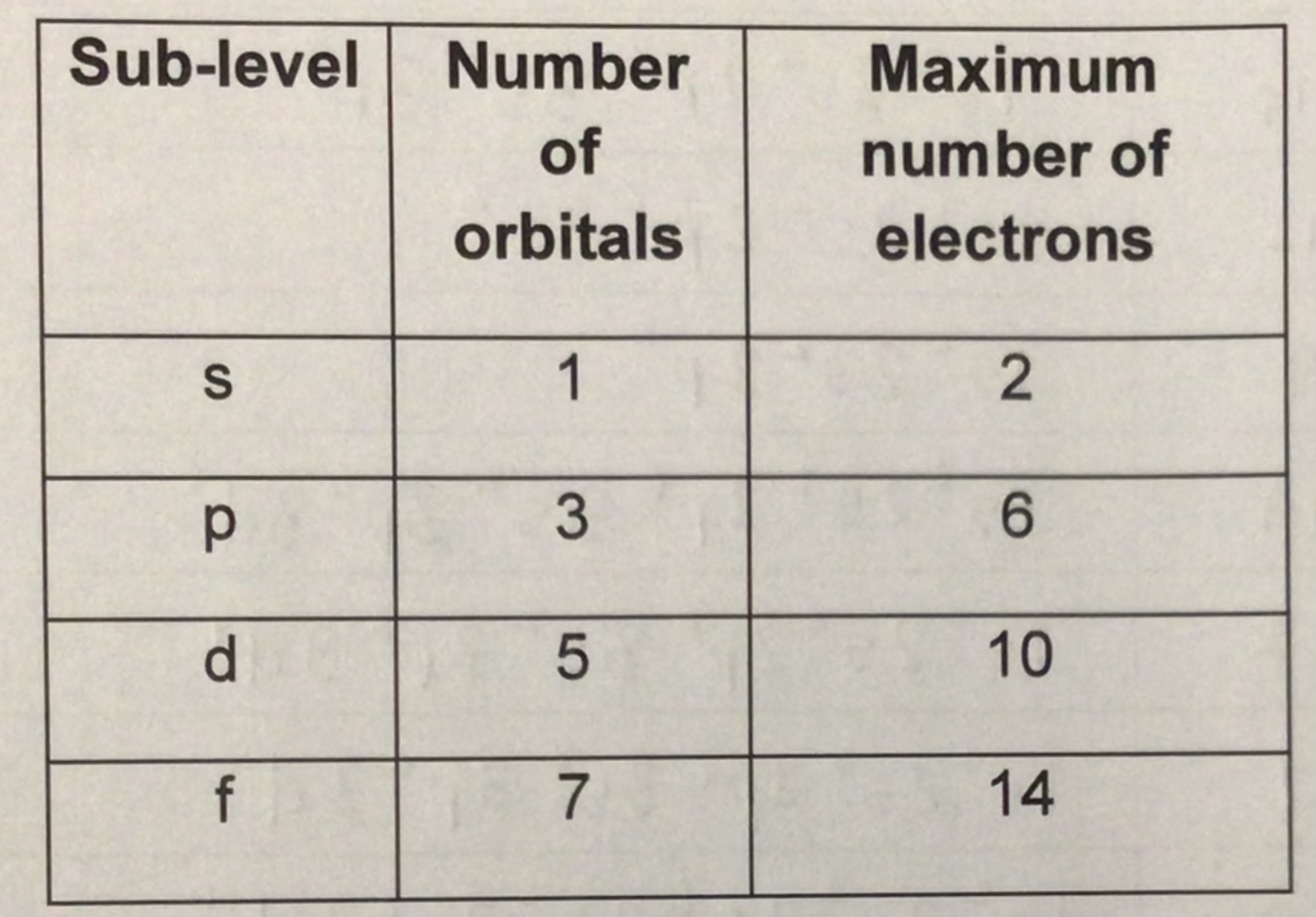
c. how many electrons does each orbital hold?
a. they are sublevels within sublevels → sub-levels are divided into orbitals
b. by boxes
c. each one holds a maximum of 2 electrons
give the number of orbitals in + maximum number of electrons held in each sub-level
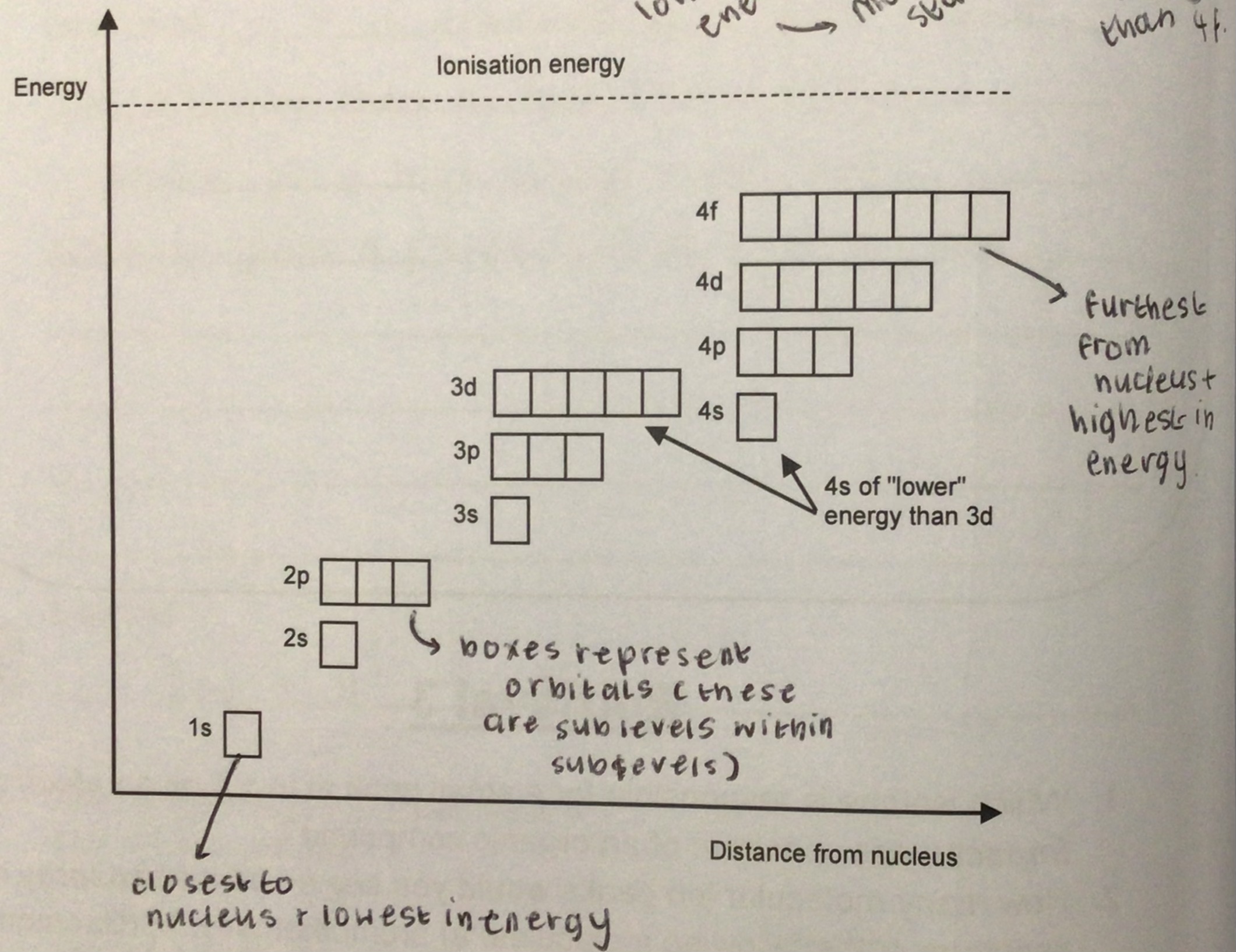
a. why does 4s fill before 3d?
b. but what happens regarding 4s + 3d when atoms are ionised?
a. because it is lower in energy → electrons fill the orbital of lowest energy first
b. electrons are removed from 4s before they’re removed from 3d
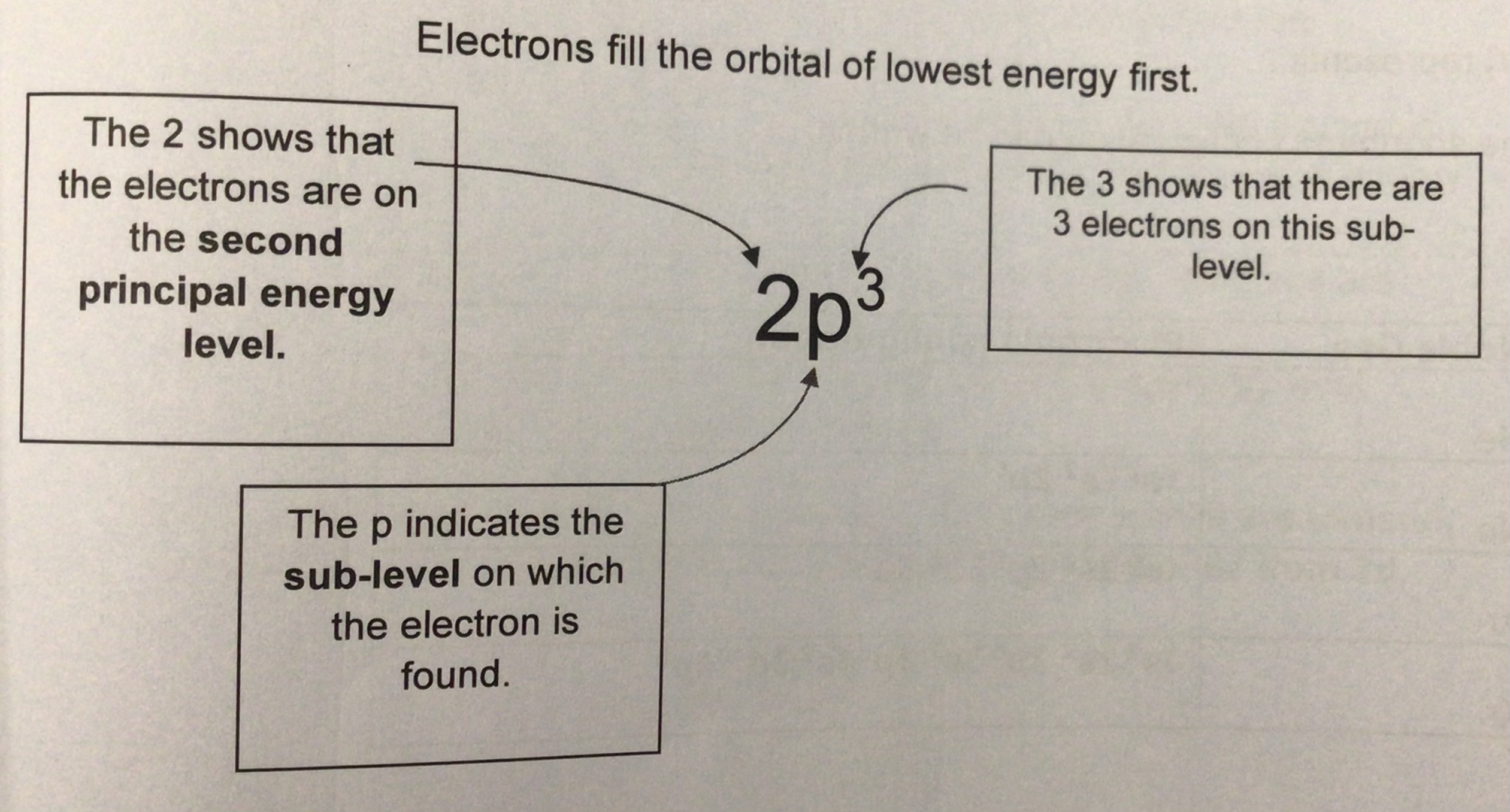
what does each number/letter represent in electronic configurations eg 2p³
write the electron configuration for:
Na
Oxygen
1s²2s²2p⁶3s¹
1s²2s²2p⁴
explain what noble gas configuration is
a short hand electronic configuration that uses the electronic configurations of the noble gases
the electron configuration of a noble gas can be used to represent part of any other elements’ configuration

give the electronic configuration of the noble gases:
a. He
b. Ne
c. Ar
d. Kr

what are the two exceptions when it comes to writing the electronic configuration of elements? + explain the reasons for these exceptions
chromium (Cr)
copper (Cu)

what’s the rule regarding 4s + 3d when atoms ionise? + why?
4s fills before 3d as it is lower in energy → removal of electrons occurs from the highest principle energy level
a. what is an energy level diagram?
b. draw one
a. a pictoral representation of an electronic configuration
b. look at image
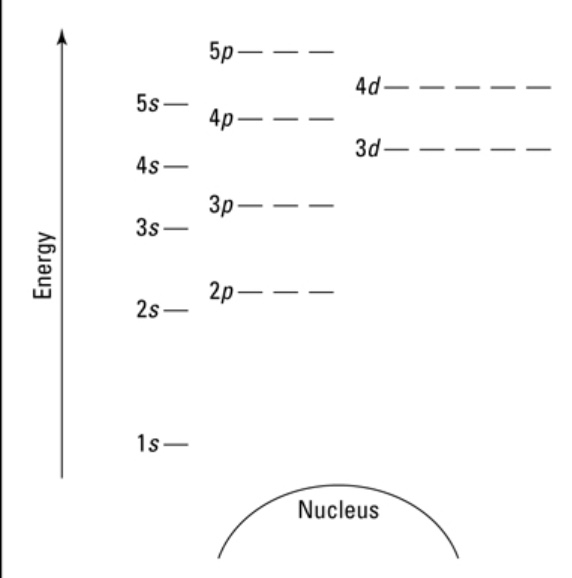
in an energy level diagram…
a. how many electrons does each orbital contain?
b. how are electrons represented/drawn in each orbital?
c. what represents the spin of the electrons?
d. how do the electrons occupy each orbital + why?
a. a maximum of 2
b. arrows represent electrons
c. direction of arrows represents spin of electrons
d. the electrons occupy orbitals as unpaired electrons rather than paired electrons until they have to be paired up → paired electrons have opposite spin as this reduces the repulsion between the paired electrons
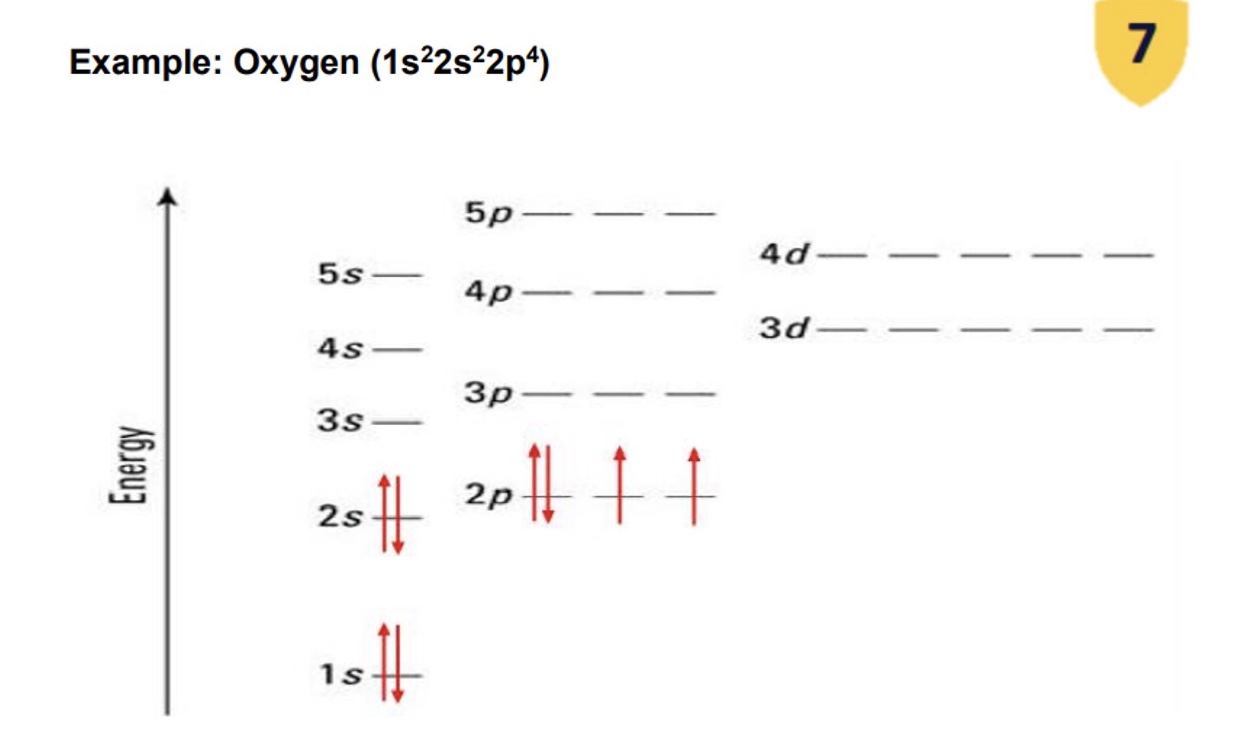
what are the three principles/rules of electron configuration?
Aufbau principle → electrons fill the lowest energy orbital first
Pauli principle → electrons in the same orbital have the opposite spin
Hund’s rule → electrons will be unpaired wherever possible
what can the periodic table be used to do regarding electron configuration? how?
to find the location of the outer electron in any atom → there is a link between the position of the atom on the table + its principal energy level + sublevel
what does each row on the periodic table represent?
a principal energy level
the block each element is in determines what?
which sub-level their outer most electron will be in
for each block on the periodic table, give the groups it contains the the sub-level it represents
s-block → group 1 + 2 → s sub-level
p-block → group 3 to 8 → p sub-level
d-block → transition metals → d sub-level
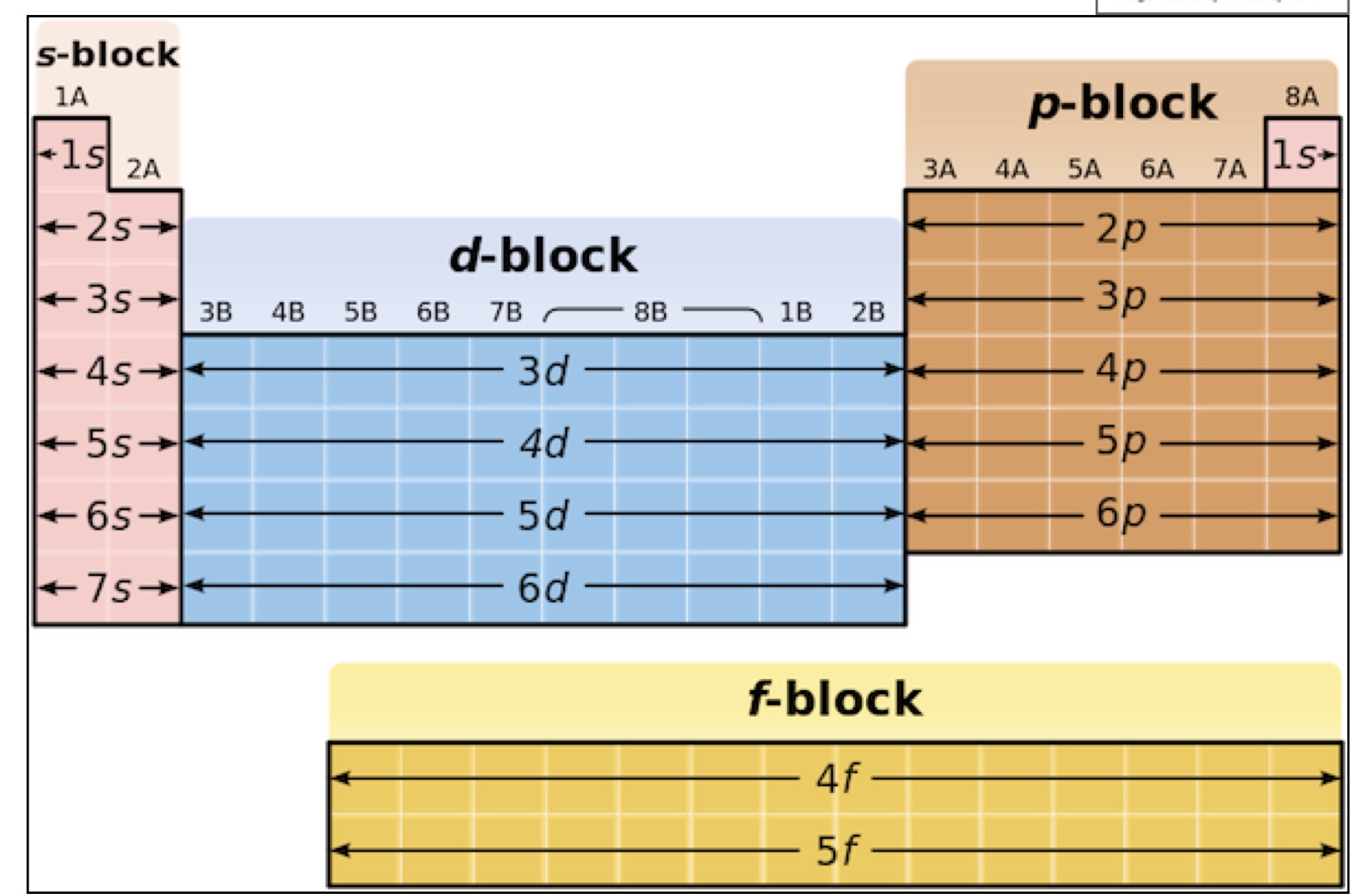
what will the columns within each block show? + give the no. of columns in each of the 3 blocks
it will give number of electrons on the sub-level ie Mg is in the second column of s-block so it had 2 electrons in its last sub-level
s block = 2
p block = 6
d block = 10
so how can you determine the electronic configuration of an element using the periodic table?
find which period its in (becomes principal energy level) *for d-block elements this number is ‘period minus one’
which block is it in (s, p, d or f)? (this becomes your sub level)
which column is it in within that block? (this is your no. of electrons/the electron ‘power of’ number)
fill the rest of the configuration before it
what do ionisation energies give evidence for?
for the existence of principal energy levels + the sub levels
what does the ionisation of an atom involve?
the loss of an electron to form a positive ion (not talking about the gain of an electron!!)
define first ionisation energy
the minimum amount of energy required to remove an electron from a gaseous atom
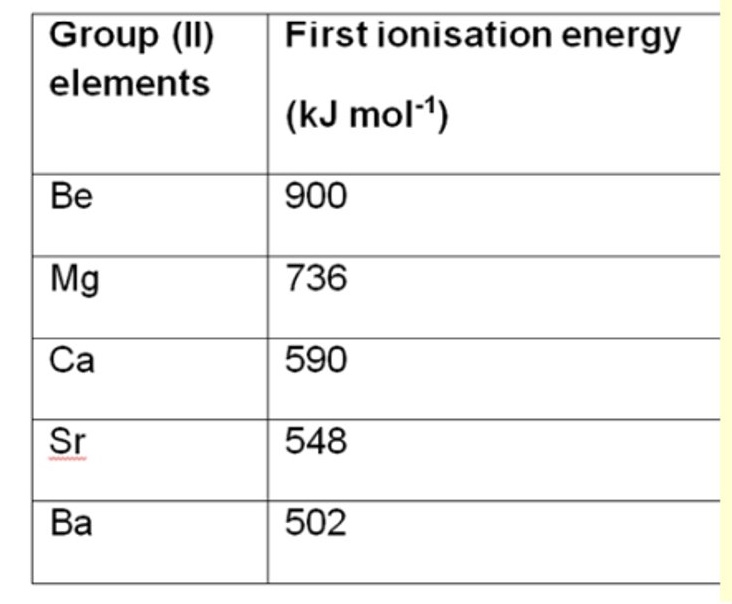
a. what is the trend in the first ionisation energies when you go down the groups eg group 2?
b. explain why
a. it decreases
b.
the electron is removed from a higher principal energy level so its removed further from the nucleus
there is more shielding
less energy is required to remove the electron as there is a weaker attraction between the nucleus + outer electron
a. describe the trend of the first ionisation energies across the periods ie period 3
b. explain the reason for this trend
a. ionisation energy increases
b.
the number of protons increase + so does the positive charge of nucleus
atomic radius decreases but shielding stays constant
more energy is required to remove the electron as there is a stronger attraction between nucleus + outer electron
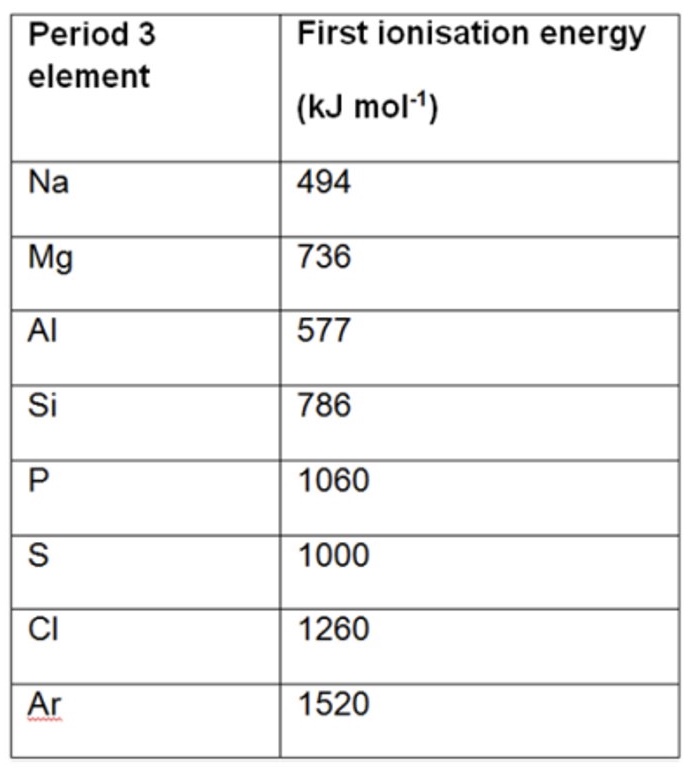
what can we see in the ionisation energies when we go across the period?
deviations in the energies increasing
explain two reasons for deviations in ionisation energies increasing across the period using the examples of Al + Mg and S + P
the first ionisation energy of Al is less than that of Mg because:
when you look at the electronic configurations of the two elements → Mg: 1s²2s²2p⁶3s²
Al: 1s²2s²2p⁶3s²3p¹
the electron is removed from a higher energy sub-level (p) in Al
which means less energy is required to remove the outer electron as there is a weaker attraction between nucleus + outer electron
the first ionisation energy of S is less than that of P because:
when you look at the electronic configurations for the two elements →
S: 1s²2s²2p⁶3s²3p³
P: 1s²2s²2p⁶3s²3p⁴
there is a pair of electrons in the p-orbital in P
this extra repulsion between the paired electrons means less energy is required to remove the electron
write the equations for first, second + third successive ionisation energies
X(g) → X⁺(g) + e⁻
X⁺(g) → X²⁺(g) + e⁻
X²⁺(g) → X³⁺(g) + e⁻
*always in the gaseous state
explain why successive ionisation energies will always increase
the positive charge on ion increases
the ionic radius decreases + shielding stays constant
nuclear attraction on the outer electron increases
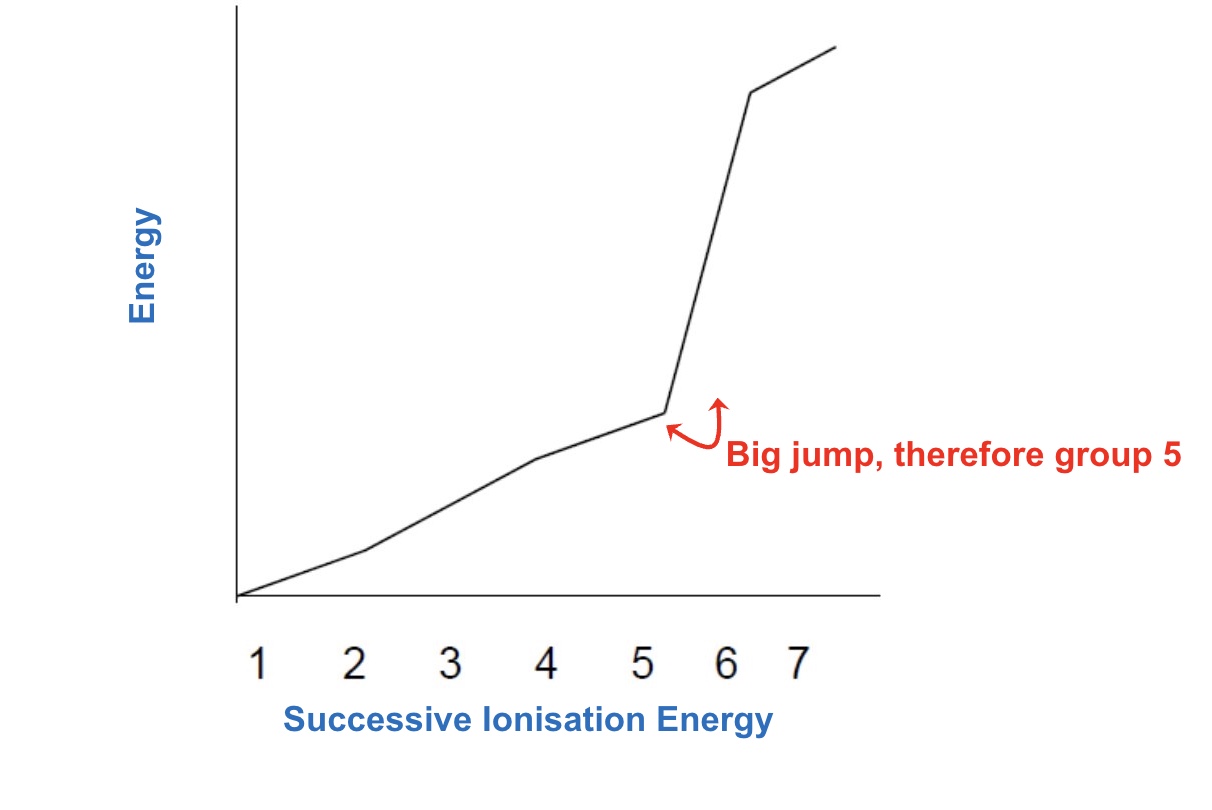
when you look at the successive ionisation energies of an element what will there always be? + why?
a significant jump in ionisation energy between two of the successive IEs eg between the 3rd + 4th one → due to the electron now being removed from a lower principle energy level (one that is much closer to the nucleus)
what does this large jump in ionisation energy be used to do + how?
can be used to find the group in which an element belongs
eg if the large jump is between 3rd + 4th IE there must have been 3 outer electrons hence the group must be in group 3