BIMM 134: Biology of Cancer
1/58
Earn XP
Description and Tags
Directly from notes
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
[start wk.1] Cancer is the _____ leading cause of death in the US.
Cancer is the second leading cause of death in the US.
Note: All diseases show a statistically significant decrease in 2023 compared to 2022, except CANCER…
Men: most likeky
Prostate
Lung Cancer
Lung Cancer is the deadliest, yet the most avoidable (not smoking).
Colon Cancer
Pancreatic Cancer
Pancreatic (survival rate is low, hard to detect, and very aggressive cancer)
Cancer Death Incidence in Male
|
Cause of Lung Cancer
Approximately ___ of lung cancer related deaths attributed to tobacco use
Approximately ___ of all cancer related deaths attributed to tobacco use
Approximately 90% of lung cancer related deaths attributed to tobacco use
Approximately 1/3 of all cancer related deaths attributed to tobacco use
Female: most likely
Breast Cancer
Lung Cancer
Colon Cancer
Pancreas
Cancer Death Incidence in Female
Lung Cancer is the most deadliest, yet the most avoidable (not smoking).
Breast
Pancreatic (survival rate is low, hard to detect, very aggressive cancer)
Colon
Education - Lung Cancer
*Other cancer declining rates: Colorectal, Breast, and Prostate…..why???
A decrease in lung cancer deaths correlates to decrease of cigarette consumption
Note: Females in US lagging in this compared to male
Other cancer declining rates: Colorectal, Breast, and Prostate…..why???
Education
Sceening
Treatment
Not much change in the other ones…………
Metastasis leads to ___% of cancer-related deaths
Metastasis leads to 95% of cancer-related deaths
At the time of diagnosis, approximately what % of cancers have already metastasized?
Up to 70% of patients with invasive cancer have metastases at diagnosis.
Approximately what % of US Budget has been spent on cancer research (pre-Trump)?
0.1%
What is Cancer?
“Cancer” actually > >>>100 forms of disease
PATIENT SPECIFIC mutations; Genetically unstable; Intratumor variations/variations in metastases
Basic processes/pathways that produce this diversity are the same
CELL DIVISION/CELL DEATH/CELL DIFFERENTIATION/Metabolism
Cancer development is a multistep process; Cells must acquire multiple mutations (4-7) for malignant transformation
ONE “RENEGADE CELL”
DECADES TO DEVELOP
How can we combat this diversity in cancer among patients?
GENOMICS;PROTEOMICS; PHARMACOGENETICS
“New proteomics (proteins) and genomics (DNA and RNA) arrays can distinguish between cancer severities and predict the most effective treatments.”
Development of patient/tumor-specific diagnosis/treatments
“If you overtreat patients, it can piss off the tumor.”
You don’t want to overtreat; cancer can be “sticky”
Basic processes/pathways that produce this diversity the same: Cancer
Increase proliferation/decrease senescence
senescence: the condition or process of deterioration with age.
Increase survival/decrease apoptosis
Decrease differentiation
Increase cell motility (metastasis)
“Cancer development is a multistep process; Cells must acquire ____ (4-7) for malignant transformation”
Cancer development is a multistep process; Cells must acquire multiple mutations (4-7) for malignant transformation
Malignant vs. Benign Tumors
Benign (not cancer) tumor cells grow
only locally and cannot spread by invasion or metastasisMalignant (cancer) cells invade neighboring tissues, enter blood vessels, and metastasize to different sites
Cancer Classification
Carcinomas
The most common cancer type with >80% of cancer-related deaths
Two-types (not needed to know):
Squamous cell carcinomas
derived from cells forming protective layer from underlying cells (ex skin,cervix)
Adenocarcinomas:
Derived from cells lining secretory cells (ex. Mucous producing cells within lung,colon, prostate)
Some tumors have mixture of these and several are separately classified
Sarcomas
Derived from connective tissues like bone,fat,etc
Hematopoietic
Derived from blood producing cells
Leukemias are circulating malignancies
Lymphomas derived from B and T lymphocytes
Usually solid masses in lymph tissue
Tumor Grade vs. Stage
Tumor Grade: Appearance of cells in biopsy
Tumor Stage: How far the cancer has spread
What does a pathologist look for?
Mitotic rate
How many/fast cells are dividing
Increased mitotic rate = worse grade
Nuclear grade
abnormal nuclei = worse grade
Cellular differentiation
Loss of cell specialization=worse grade
Surgical margins
How close are tumor cells to the surgical edge
Positive=cells at edge=worse prognosis
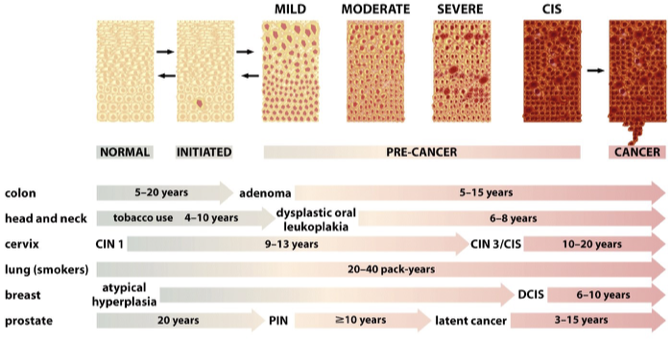
Cancer Progression Stages
Many cancers have somewhat predictable mutational patterns
Colon cancer highly characterized
TNM STAGING
PRIMARY TUMOR
ABSENCE/PRESENCE REGIONAL LYMPH NODES
ABSENCE/PRESENCE DISTANT METASTASIS

Stage | Description | Example of TNM |
I | localized | T2N0MO (Patient 1) |
II or III | Regional spread | T3N2MO (Patient 2) |
IV | Distant Metastasis | T3N2M1 (Patient ) |
^Need to know!!
SIMPLIFIED TNM EXAMPLES
Patient 1 Biopsy: well-differentiated;low mitotic index; good surgical margins (LOW GRADE);
Gene screen Estrogen receptor +; Her2+; progesterone receptor +
Patient 2 Biopsy: poorly differentiated; high mitotic index good surgical margins (High GRADE)
Gene screen Estrogen receptor +; Her2+; progesterone receptor +
Patient 3 Biopsy: poorly differentiated; high mitotic index poor surgical margins (High GRADE)
Gene screen Estrogen receptor neg; Her2 neg; progesterone receptor neg(TRIPLE NEGATIVE)
Stage | Description | Example of TNM |
I | localized | T2N0MO (Patient 1) |
II or III | Regional spread | T3N2MO (Patient 2) |
IV | Distant Metastasis | T3N2M1 (Patient ) |
Obesity vs. Cancer
Fat produces factors that increase the likelihood that cells will develop mutations.
These factors represent Carcinogens: Cancer Promoters
Exercise vs Cancer
IN CONTRAST, exercise decreases the risk of cancer and cancer progression
Decreases circulating cancer promoters
Increases immune response against cancer.... "poisons the tumor-promoting microenvironment
Carcinogens
Mutagens: Directly mutate DNA/RNA
Include tobacco; UV radiation
Promoters: Increase the likelihood of mutations
Substances that increase proliferation, cell damage, inflammation e.g., Alcohol;hormones
A combination often results in a synergistic increase in cancer incidence
Alcohol + Smoking
Alcohol caused cell damage and stimulates cell proliferation
Smoking leads to DNA mutation
Work synergistically to produce greater effect than acting singly
Oncogenes vs Tumor Suppressors
Oncogene
Gain of function (stuck accelerator)
Dominant phenotype
Tumor Suppressors
Loss of function
Recessive phenotype
Generally, a combination of activation of oncogenes and inactivation of tumor suppressor genes lead to lack of growth control and unrestrained proliferation
Proto-oncogene vs Oncogenic Ras
Proto-oncogene: Ras with normal function
Oncogenic Ras: Increase Ras activity, increase in proliferation
Telomeres, telomerase, and cellular “immortality
Telomeres are a repeated sequence at the ends of the chromosomes that protect the chromosomes from damage
Cancer cells re-express telomerase leading to unlimited cell division: Endless tank of gas
Apoptosis
Cell death
Cancer cells decrease apoptosis via several mechanisms including
Gain of anti-apoptotic proteins (e.g. Bcl-2)
loss of pro-apoptotic proteins (e.g.BAX or p53)
Angiogenesis
Tumors are limited in growth to around 1-2mm due to limited diffusion of oxygen/nutrients and removal of waste
In response, cancer cells initiate an “angiogenic switch”
Increasing release of pro-angiogenic molecules (e.g. VEGF)
Decreasing release of anti-angiogenic molecules (e.g. thrombospondin)
Angiogenesis results in…
Resulting in new blood vessel growth
Growth of cancer
Invasion into the bloodstream
Metastasis
Summary of main hallmarks of cancer
Gain of an oncogene
Example: RAS
Car analogy: stuck accelerator
Loss of tumor suppressor
Example: Loss of p53
Car analogy: cutting the brakes
Cell immortalization
Example: Loss of telomerase
Car analogy: running out of gas (or electric charge..)
Loss of apoptosis
Example: Loss of p53
Angiogenesis: growth of blood vessels into tumor
Example: Increase in VEGF
Metastasis: Spread to distant sites
Example: loss of E-cadherin
Warburg effect: Aerobic glycolysis
The Warburg effect is when cells preferentially use glycolysis to produce energy even in the presence of oxygen, leading to lactate production (aerobic glycolysis).
[end of wk.1] Reverse Warburg effect
Instead of cancer cells doing glycolysis themselves, nearby stromal cells (like fibroblasts) do glycolysis and feed the cancer cells.
[start of wk.2] Enzyme-linked Receptors: Tyrosine Kinase receptors
Many growth factors (insulin, epidermal growth factor, fibroblast growth factor) use this type of receptor
Important for control of cell division and growth
These are often mutated in cancer cells
Signal Transduction: Phosphorylation Cascade
Adding or removing P to molecules is used to activate/deactivate molecules
Protein Kinases: Add phosphates
Protein phosphatases: Remove P
Tyrosine Kinases: Add phosphates to tyrosines
Serine/threonine Kinases: Add phosphates to serine/threonines
Dual kinase:has both tyrosine kinase and ser/thr kinase activity
Lipid kinase: Add phosphates to lipids
Tyrosine Kinase receptors
Family of related receptors
Share common extracellular and intracellular protein domains
INCREASED COMPLEXITY
Some signals can activate several receptors
Some receptors activated by multiple ligand Intracellular pathway convergence and divergence
Intracellular pathways can have numerous cellular effects depending on pathways that are activated
EGF-RTK-RAS-MAPK Pathway
EGF binds to EGF Receptor
Binding causes conformational change in receptor exposing extracellular dimerization domain
RTK-RAS-MAPK Pathway
EGF binds to EGFR
Binding causes conformational change in receptor
Exposing extracellular dimerization domain
Receptor dimerization
Autophosphorylation of key tyrosines needed for recruitment of signal molecules
Phosphorylation of key tyrosine levels result in binding/activation of numerous intracellular molecules
Helps explains activation of multiple downstream pathways/cellular effects
INCREASED COMPLEXITY
Some signals can activate several receptors
Some receptors activated by multiple ligand Intracellular pathway convergence and divergence
Intracellular pathways can have numerous cellular effects depending on pathways that are activated
Intracellular protein interactions
Intracellular protein interactions Mediated by specific protein modular domains
SH2 (Src homology 2) 100 amino acids
Interact with specific amino acids located near P tyrosine
SH3 (Src homology ) 50 amino acids
Interact with proline and hyrdophobic residues on proteins
Protein recruitment to membrane/receptor
Protein recruitment to the membrane/receptor Adaptor GRB2 binds to EGFR via the SH2 domain
SH3 domains of GRB2 bind SOS (son of sevenless), which recruits SOS to the membrane
SOS is a GEF( Guanine-nucleotide exchange factor)
Transfers a GTP to Ras: activates Ras
Results in RAS binding/activation
RAS is subsequently inactivated by GAP (GTPase) proteins, which hydrolyze GTP to GDP
RAS Modifications
Addition of a farnesyl lipid and methylation to RAS targets thought to be necessary for full RAS activity
Farnesyl transferase inhibitors have been tested in clinical trials
RTK-RAS-MAPK Pathway
Ras now recruits RAF and other proteins to membrane
RAF is a serine/threonine kinase
RAF (MAPKKK) adds P and activates
MEK (MAPKK) MEK is a tyrosine and ser/thre kinase
MEK adds P and activates MAPK (also called ERK: extracellular signal-related Kinase)
Activated MAPK translocates to nucleus and activates transcription factors
These include AP-1 (Fos/Jun) and Myc:Max
Activate many genes (e.g. cyclins) promoting cell cycle/cell division
Alternative MAPK pathways
JNK and P38 MAPK pathways
Respond to cellular damage/stress
Usually trigger apoptosis
Type and severity of signals can lead to either
Cellular survival or Cellular death
RAS-PI3K-AKT Pathway
RAS can also activate other pathways that often mutated in human cancers, including PI3K/AKT pathway
RAS activated Phosphatidylinositol-3-kinase (PI3K:Lipid Kinase):This kinase attaches phosphates to lipids,in this case, to PIP2, producing PIP3
PIP3 binds a serine/threonine kinases and PDK-1 and AKT(PKB): PDK1 activated AKT
Phosphorylation of AKT/PKB leads to phosphorylation/activation other substrates resulting in
Decrease apoptosis/increased survival
Increase in protein synthesis
RAS-PI3K-AKT-mTOR Pathway
Note: mTOR is also a serine threonine kinase
Activation of the serine/threonine kinase,mTOR, (mechanistic/mammalian target of rapamycin),
leads to activation of many cancer hallmarks
proliferation
motility/metastasis,
angiogenesis, and decreased apoptosis
Studies suggest that the disruption of the PI3K pathway is one of the most common pathways mutated in human cancers
loss of PTEN (a phosphatase) the most abundant tumor suppressor mutation
PTEN converts PIP3 back to PIP2 inhibiting pathway
Point mutations vs Gene Amplification
Point mutations
deletions of the receptor causing ligand-independent dimerization/activation
Gene amplification
overexpression of receptors; ligand-independent dimerization/activation
Conversion from paracrine to autocrine signaling pattern
Cancer likes autocrine; to be independent
EGF Receptors and Ligands
Two main receptors
(HER1)(erB1)
HER2(erB2)
HER2
Over expression of HER2 common in breast cancer
Expression associated with poor prognosis
Over expression of HER2 results in ligand-independent HER2 dimerization/activation of Ras-MAPK and PI3K-AKT pathways
RAS mutation
RAS normally only sends signals when a positive growth signal is sent to the cell
Then RAS becomes quickly deactivated
Ras point mutations prevent Ras interaction with GAPs (GTPase activating proteins)
Prevents (decreases) hydrolysis of GTP
Ras remains in activated state for extended periods of time
most RAS mutations are within codons
12,13,61
Codon 12 mutations predominate in lung,colorectal and pancreatic cancers
High prevalence of a G12C (Glycine to cysteine) mutation is associated with Lung adenocarcinoma (LUAD)
High prevalence of a G12D (Glycine to Aspartate) mutation is strongly associated with pancreatic (PDAC) and colorectal cancer (CRC)
⭐RAS therapeutics : Undruggable target…???…️
Ras has been considered “undruggable” because
GTP binding pocket relatively inaccessible
High affinity for GTP
High levels of cytoplasmic GTP
⭐ Two inhibitors targeted G12C mutations in NSCLC have gained FDA approval
Sotorasib (2021) and Adagrasib (2022)
RAS therapeutics RAS G12C mutations in NSCLC
These small molecules covalently bind to the cysteine in the G12C mutation in the switch-II GTP binding pocket preventing/decreasing activation
They do not greatly affect wild-type RAS or other RAS mutations
RAS therapeutics RAS G12D mutations in Pancreatic cancers
Development of RMC-6236