BIO 2 Functional Group vocabs and structure
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
DEFINE functional groups
specific repeated combination of atoms (usually parts of molecule) that have distinct properties (responsible for a characteristic of that molecule) and are often found in many biomolecules
DESCRIBE functional groups (give list of example functional groups)
Amino Group
Carboxyl Group
Hydroxyl Group
Methyl Group
Carbonyl Group
Thiol (a.k.a Sulfanyl) Group
Phosphate Group
EXPLAIN Hydroxyl Group
OH group covalently bonded to another atom (R — OH)
often, but not always bound to C atom; but, both C and H atoms will be attached towards O atom b/c of its large EN → making hydroxyl groups polar covalent bonds
EXPLAIN Carboxyl Group
combination of carbonyl and hydroxyl group attached to the same carbon → new characteristic recognized as carboxylic
carboxyl can ionize (likely to give up e-), which means it can act as a ACID and release H+ atom from the hydroxyl group as a free proton (H+) → results in delocalized negative charge on remaining O atoms
What kind of interaction does carboxyl have with protons (H+)?
Carboxyl is a proton DONOR and ACCEPTOR
What does it mean for a functional group to be both a proton donor and acceptor?
When a functional group serves as both a proton acceptor and donor, it means that it can flip-flop b/w protonated (R-COOH) and deprotonated (R-COO-) after donating H+ depending on the pH of the solution
EXPLAIN Amino Group
organic compound containing an amino group is called an amine (R — NH3)
like O, N is more EN than C and H → results in the amino groups displaying some polar characteristic
What kind of interaction does the Amino Group have with protons (H+)?
Amino is a proton ACCEPTOR
can act like a BASE, bond w/ a fourth H atom → N atom becomes positively charged and form ionic bonds
EXPLAIN Phosphate Group
the O atom bonded w/ the P atom is more EN, making the phosphate group are polar covalent bonds
commonly in nucleic acids and on phospholipids (make up the lipid bilayers in cells)
How else does the O atom’s high EN interact with its surrounding atoms?
the polar characteristic and large EN value of the O allows the O atoms to form HB (INTERmolecular (like a elastic force, not magnetic) w/ near by H atoms with a 𝛅+ (from forming a intramolecular (covalent) bond with another atom with a high EN
EXPLAIN Methyl Group
In a NON POLAR bond or molecule, e- and charge will be relatively evenly distributed
derived from methane molecule, consist of one C atom and 3 H atom (R — CH3)
How does the methyl group interact with its surrounding atoms/materials
b/c the C-H3 bonds are effectively NP bond, methyl groups can’t form HBs and won’t interact with/ polar compounds (i.e. H2O)
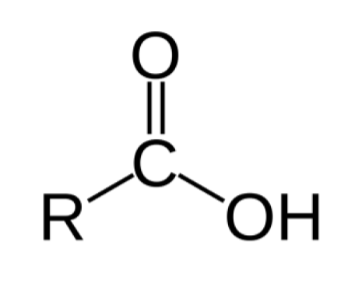
What functional group is this?
carboxyl group
combination of a carbonyl and hydroxyl group
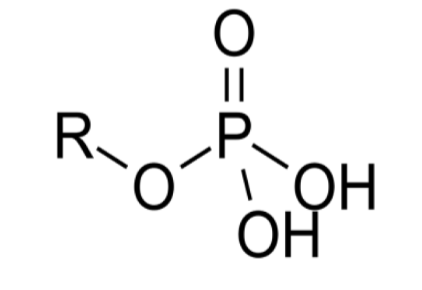
What functional group is this?
phosphate group
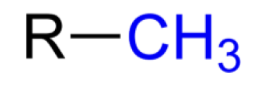
What functional group is this?
methyl group
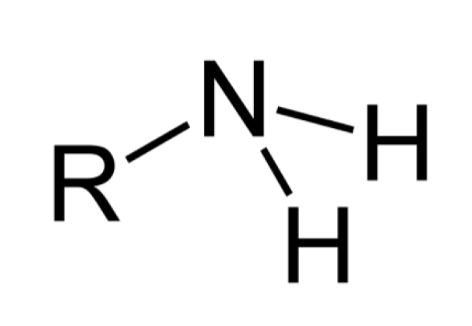
What functional group is this?
amino group: amine
EXPLAIN amine group
has a nitrogen atom directly bonded directly to alkyl groups
NO carbonyl group attached
BASIC
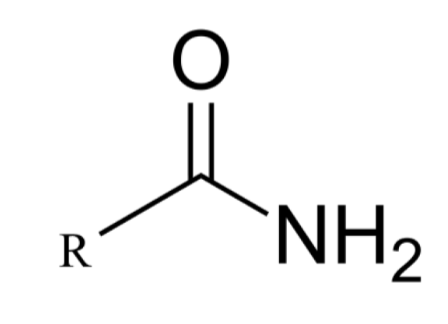
What functional group is this?
amino group: amide (carboxyamide)
EXPLAIN amide group
derivatives of carboxylic ACID
ACIDIC
nitrogen atom directly attached to a carbonyl group (C=O)
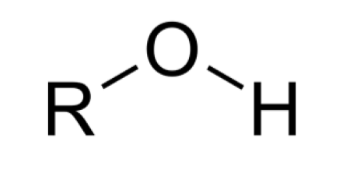
What functional group is this?
hydroxyl group