chemistry- periodicty draft
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Periodicity definition
Repeating trends of physical or chemical properties
What is a period
Horizontal rows of elements. Period one starts at H, period starts at Li…
All elements in the same period have the same number of principle energy levels and there are trends in the properties as w move along a period
What is a group
A vertical column
All elements in a group have the same number of outer electrons and similar chemical properties.
How does atomic radius change down a group and why
Increases down a group
As more shells
So more shielding
So outer electrons are further from the nucleus and have a weaker attraction to the nucleus
How does atomic radius change down a period and why
Atomic radius decreases down a period
As number of protons increases and shielding stays the same
So stronger nuclear charge and stronger attraction between outer electrons and nucleus
What effect is more significant shielding/ distance or number of protons
Shielding/ distance
What is the first ionisation energy
The energy required to remove 1 mol of electrons from 1 mol of gaseous atoms
Write an equation showing the process of removing the first ionisation energy
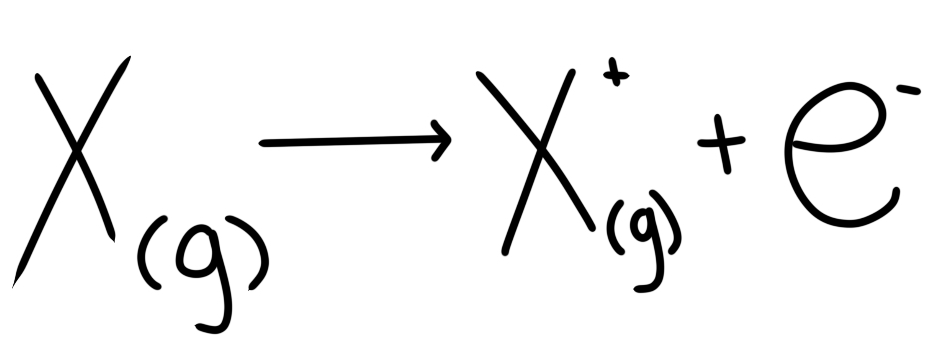
Describe and explain the trend in the first ionisation energy in group 1 elements
Ionisation energy decreases.
As the electron is removed from a higher principle energy leaves.
So distance between the outer electrons and the nucleus increases.
Shielding increases. Weaker attraction between nepucleus and outer electron so less energy required to remove electron.
Describe and explain the general trend of ionisation enerypgy across a period.
Ionisation energy increases as number of protons increase and atomic radius decreases. Shielding remains the same so there is a stronger attraction between the nucleus and the outer electrons, so more energy is required to remover electron.
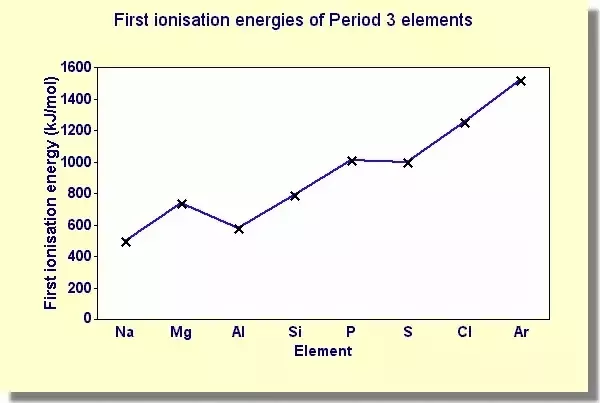
Draw a graph to show ionisation energy across a period
Increases. Except for between group 2 and group 3 elements where it decreases slightly, and between group 5 and 6 where it also decreases but by less.
Why is AL first IE lower than Mg
Mg: 1s2, 2s2, 2p6, 3s2
Al: 1s2, 2s2, 2p6, 3s2, 3p1
Electron is removed from a higher energy p sub shell so there is a weaker attraction between nucleus and outer electrons. Less energy required to remove Al.
Why is S first IE lower than P
P: 1s2, 2s2, 2p6, 3s2, 3p3
S: 1s2, 2s2, 2p6, 3s2, 3p4
There is a pair of electrons in P orbital which repel each other so less energy required to remove electron.
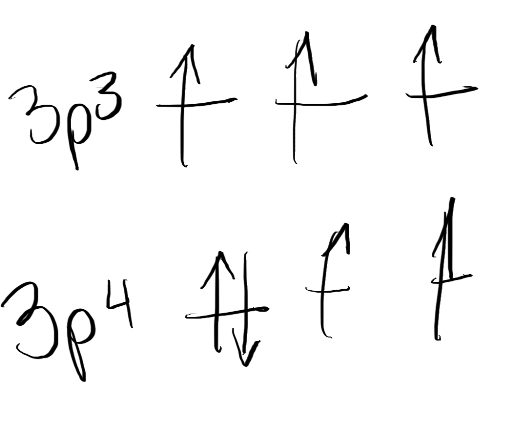
What are successive ionisation energy’s
Measured when additional electrons are removed, always removing outermost electrons.
You can as many successive ionisation energies as there are electrons in an atom
Explain why successive ionisation energies increase
Successive IEs will always increase
As positive charge in ion increases
So ionic radius decreases .
So nuclear attraction increases.
So more energy is required to remove outer electron
Write the first four SIE for Al

How can ionisation energies be used as proof for principle energy shells.
IE will always increase by a larger margin when an electron is being removed from a lower shell that is closer to the nucleus..
What is metallic bonding
String electrostatic forces of attraction between positive ions and delocalised electrons
How many delocalised electrons are in a metallic lattice
The number of delocalised electrons is proportional to those lost by mega, atoms in their attempt to gain a noble gas configuration. E.g. Na will donate 1 per atom, Mg will donate 2
What factors influence the strength of a metallic bond and why
Ionic charge on the metal (higher charge stronger bond)
Number of delocalised electrons (more e- stronger bond)
Ionic radius (smaller radius stronger bond)
This is because the positive ions will be more strongly attracted to the delocalised sea of electrons
Explain the conductivity of metals
Good conductors as the delocalised electrons are free to move and carry a charge throughout the structure.
Explain the melting point of metals
High melting a boiling points due to strong electrostatic forces of attraction between positive ions and delocalised electrons
Explain the malleability and ductility of metals
They can be hammered into shape as the layers on ions can slide over one and other
What are the giant covalent structures in Period 2 and 3
Diamond, graphite, graphene, silicon, silicon dioxide
What is the melting point of giant covalent structures
High as string covalent bonds between atoms require a lot of energy to break
Describe the bonding and conductivity of diamond
Each carbon atom covalently bonded to 4 other carbon atoms.
Not conductive as no delocalised electrons
Describe the bonding and conductivity of graphite
Each carbon atom covalently bonded to three other C atoms forming hexagonal layers. Weak London forces between layers.
Yes as one delocalised electron per atom free to carry charge by moving and flowing
Describe the bonding and conductivity of graphene
1 layer of graphite, each carbon atom covalently bonded to 3 other forming a single hexagonal layer.
Conductive as delocalised electrons are free to move and flow.
Describe the bonding and conductivity of silicon
Each silicon atom covalently bonded to four other silicon atoms.
No conductivity as no delocalised electrons.
Describe the bonding and conductivity of silicon dioxide
Each Si atom bonded to four O atoms.
No as no delocalised electrons
Period 2 what is bonding and crystal structure of Li and Be
Bonding - metallic
Crystal structure - giant metallic
Period 2 what is bonding and crystal structure of C
Bonding - covalent
Crystal structure- giant covalent (diamond, graphite, graphene)
Period 2 what is bonding and crystal structure of N, O and F
Bonding, covalent
Crystal structure, simple molecular. All form diatomic and are non polar so only London forces.
Period 2 what is bonding and crystal structure of Ne
Bonding, monoatomic (single atom)
Crystal structure, covalent bonds, nonpolar atoms
Period 3 what is bonding and crystal structure of Na and Mg and Al
Bonding, metallic
Crystal structure, giant metallic lattice
Period 2 what is bonding and crystal structure of Si
Bonding, covalent
Crystal structure, giant covalent
Period 3 what is bonding and crystal structure of P, S Cl
Covalent
Simple molecular
Period 3 what is bonding and crystal structure of Ar
Monoatomic
Simple molecular, non polar atoms so only London forces between molecules.