Calcium Aluminate Cement
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Hydration reactions for CA (monocalcium aluminate) - long
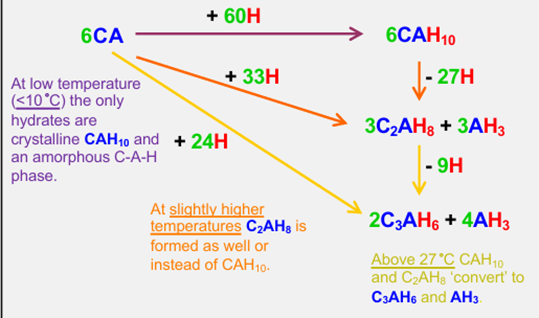
Properties of CAC (4)
Faster strength developement than CEM 1 (PC) but sets in similar time
Good chemical resistance (eg sulfates)
Can use at low temps
Used w refractory aggregate to form refractory castables (good mechanical strength and dry heat resistance)
Conversion reactions
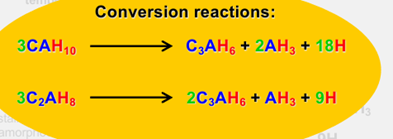
above 27C
explain strength changes as a result of conversion
strength ↓ over time THEN ↑
C3AH6 is denser than CAH10 and C2AH8
low w/c = ↓ water & space BUT conversions ↑ porosity (& release water) so ↓ strength
new space can be filled w the other hydration products so ↑ strength
CAC-GGBS mixes
Stratlingite forms instead of CAH10 & C2AH8
No conversion reactions so no porosity increase
Stratlingite strength constant over time.
compare CAC to PC properties
sets in similar time
CAC strength development faster
1 day CAC strength > 28day PC
heat of hydration lower
CAC-OPC property & why
reduced setting times (flash set can occur)
CAC setting accelerated by gypsum & CH from OPC
OPC setting accelerated by gypsum. Aluminates from CAC react to from ettringite.