Thermodynamics
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
1st law of thermodynamics
energy cannot be created or destroyed - only changed from one form to another
how are system and surroundings related
delta Esystem = - delta Esurroundings
internal energy U
energy associated with random disordered motion of molecules in a system
how can U be changed
heat
work
work
w = -P deltaV = -P(Vf-Vi)
transfer of energy from system to surroundings
work = pos
surroundings are doing work on our system
i.e. system gains energy
work = neg
system doing work on surrounding
i.e. system loses energy
entropy S
a measure of how dispersed the energy is inside a closed or isolated system
higher S
more energy dispersed
lower S
energy less dispersed
second law of thermodynamics
entropy S of an isolated system always increases
deltaS>0
ex: ice cube melting
is the inital or final state of an isolated container more random
final state is less ordered and more random
system has become more disordered and entropy has increased
can we revert back to original state
no unless we do work on the system
reversible process
both system and surrounding can be reverted to original state after process has occured
irreversible process
system and surrounding cannot be reverted to original state after process has occured
how can we measure change of entropy delta S for a reversible process
delta S = qrev/T
q rev
heat supplied reversible to the system
microstate
when we look at a molecular system we want to know the number of diff ways molecules can be arranged
each individual arrangement = microstate
how is microstate and entropy related
greater no. of microstates = greater entropy
S = kB ln(W)
W
no. of microstates
kB
boltzman constant
-> 1.38 x 10^-23
how can we increase no. of microstates
increase volume
add heat
increase no. of molecules
change in entropy of a system from one state to another
delta S = kB ln(W final) - kB ln(W initial)
enthalpy H
heat content of a system at constant pressure
H = U + PV
change in enthalpy delta H
amount of heat a system loses or gains
delta H = delta U + P delta V
exothermic reaction
delta H is negative
endothermic reaction
delta H is positive
enthalpy vs internal energy
IE: sum of all molecular energies in a system
E: combo of internal energies + energy required to make space
so when heat is added enthalpy changes stays the same but if gas can expand some energy goes into work so internal energy increases less
total entropy change of the universe
ΔSuniverse=ΔSsystem+ΔSsurroundings
entropy change of surroundings
ΔSsurroundings = −ΔHsystem/T
Gibbs free energy of a system
∆𝑮𝒔𝒚𝒔𝒕𝒆𝒎 = ∆𝑯𝒔𝒚𝒔𝒕𝒆𝒎 − 𝑻∆𝑺𝒔𝒚𝒔𝒕𝒆m
neg delta G
spontaneous
pos delta G
not spontaneous
delta G = 0
equilibrium
Clausius-Clapeyron equation
OR
P2 = P1e
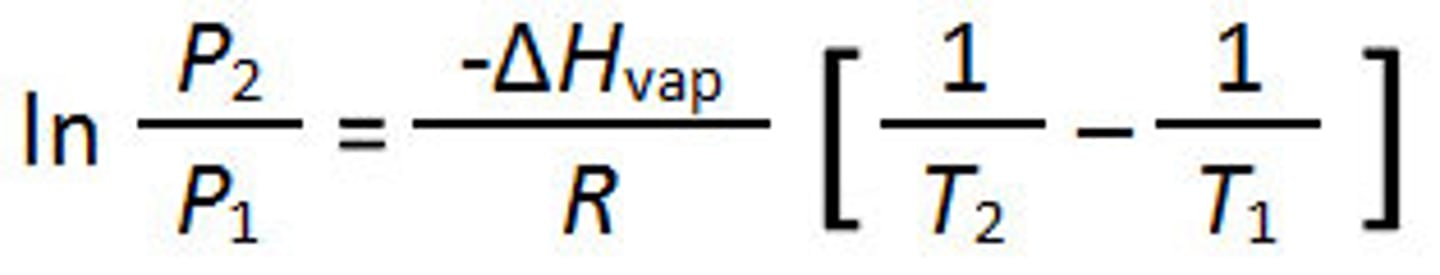
equation for gibbs free energy under non-standard conditions
𝚫𝑮 = 𝚫𝑮 𝟎 + 𝑹𝑻𝒍𝒏 (Q)
What is the relationship between ΔG° and K
𝚫𝑮 𝟎 = −𝑹𝑻𝒍𝒏 (K)
If products are favoured at equilibrium
K>1
delta G0 < 0
reactants are favoured at equilibrium
k < 1
delta G0 > 0
neither reactants or products are favoured
K = 1
delta G0 = 0