Chem final
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
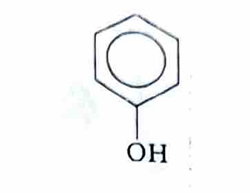
phenol
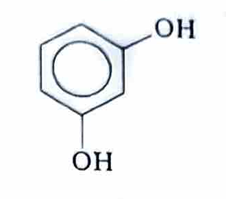
Resorcinol
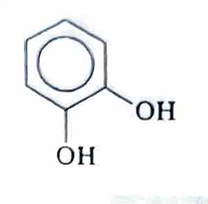
Catechol
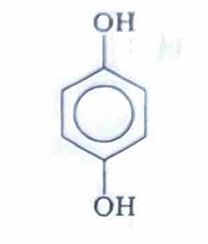
Hydroquinone
O-cresol
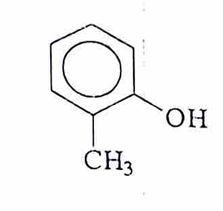
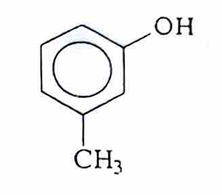
M-cresol
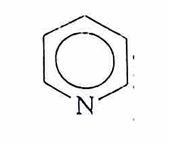
pyridine
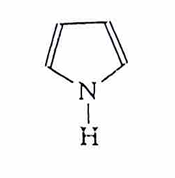
pyrrole
quinoline
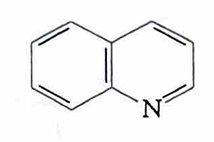
pyrimidine
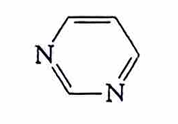
purine
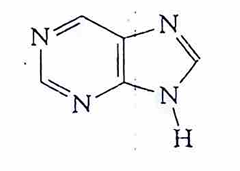
adenine
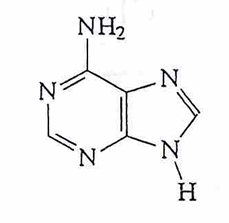
guanine
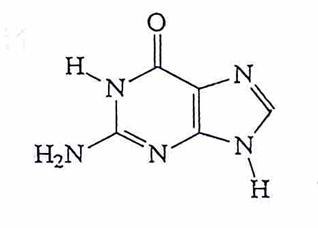
cytosine
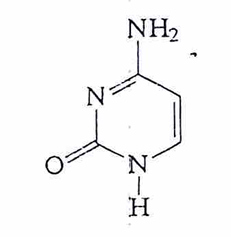
thymine
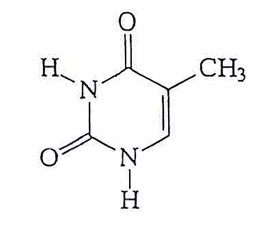
morphine
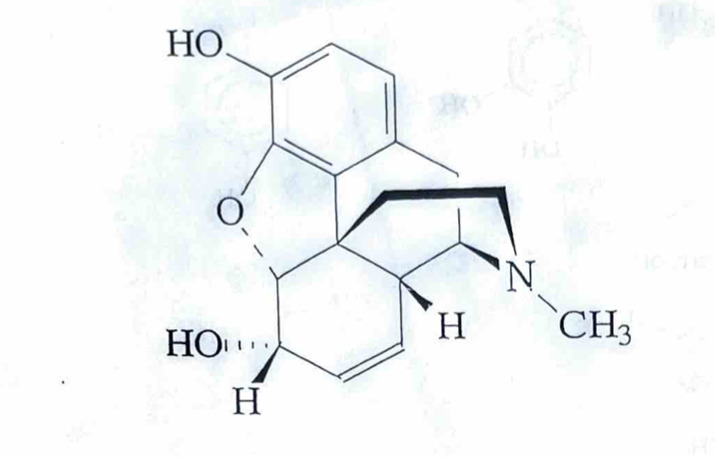
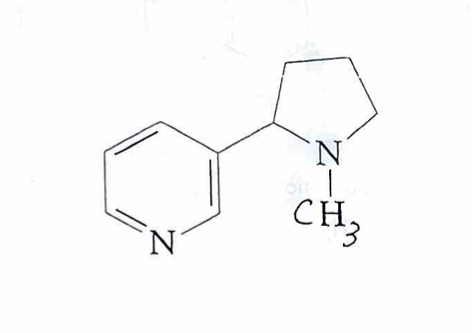
nicotine
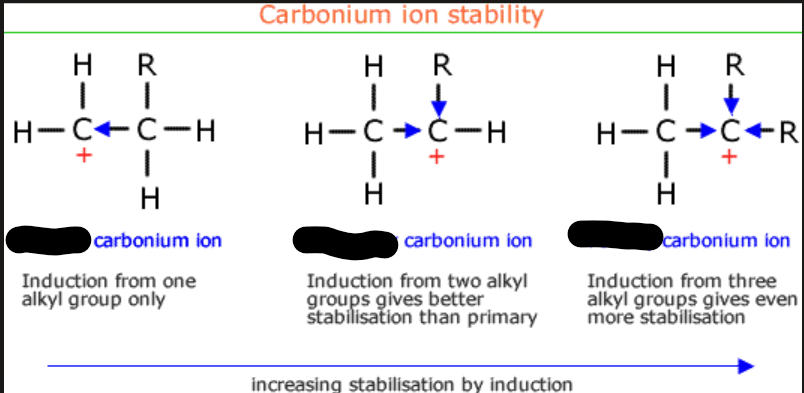
the first one is a ___: because it has one c bonded
the second one is __ because it has two c bonded
the third one is __: because it has three c bonded
primary
secondary
tertiary
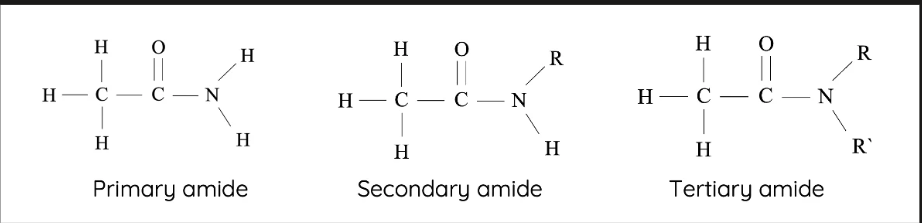
the first one is a ___ ammine because it has one c bonded
the second one is __ ammine because it has two c bonded
the third one is __ammine because it has three c bonded
primary
secondary
tertiary
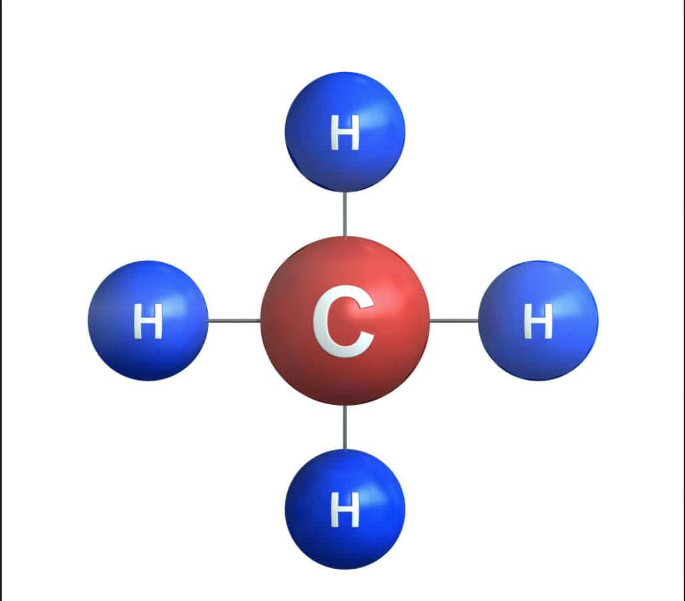
CH4
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
bp __
methan
1
liquid
methyl
-161.5
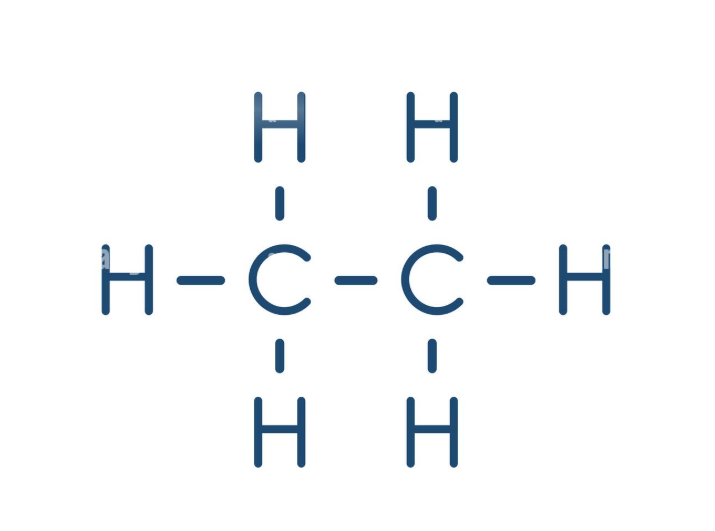
CH3CH3
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
ethane
1
gas
ethyl
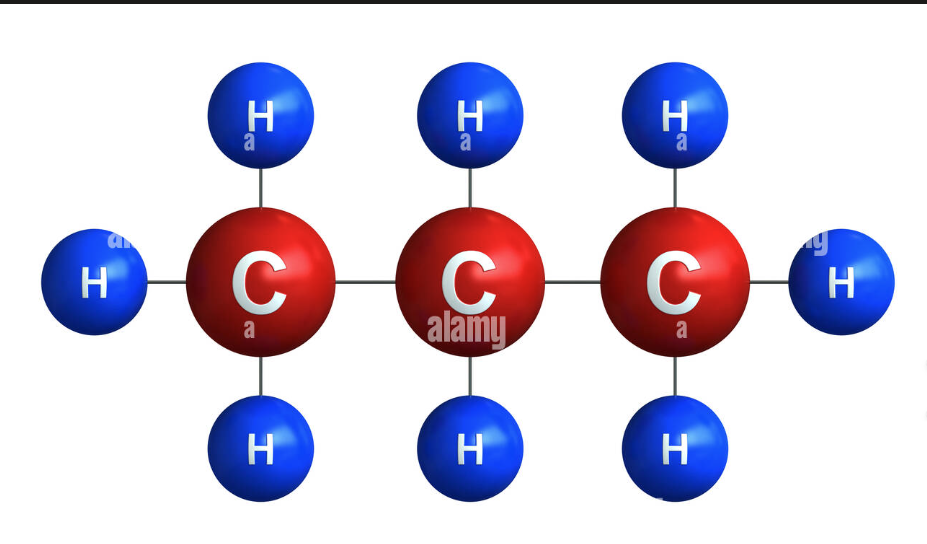
CH3CH2CH3
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
propane
1
gas
propyl
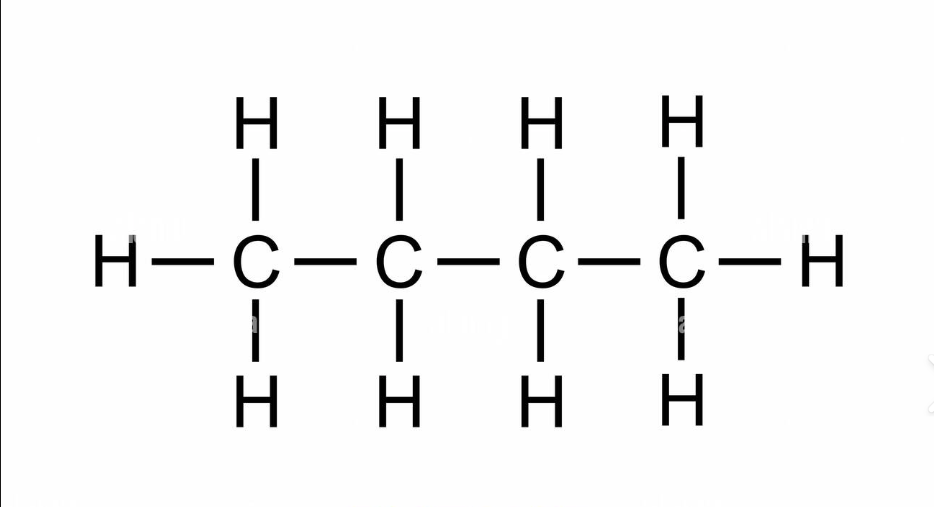
C4H10
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
butane
2
gas
butyl
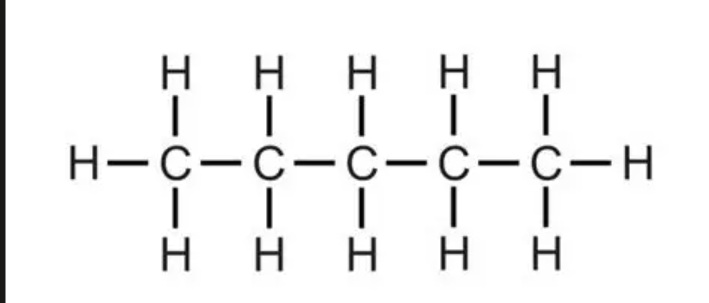
C5H12
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
pentane
3
liquid
pentyl
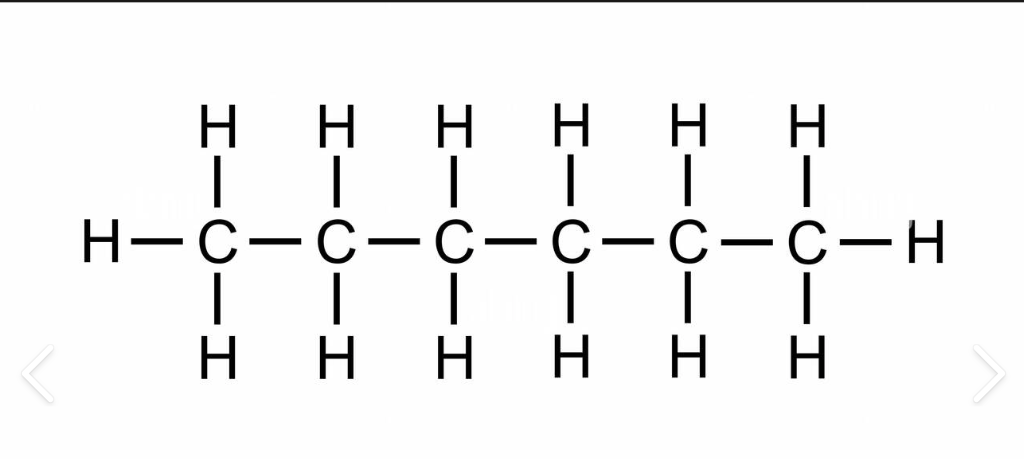
C6H14
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
hexane
5
liquid
hexyl
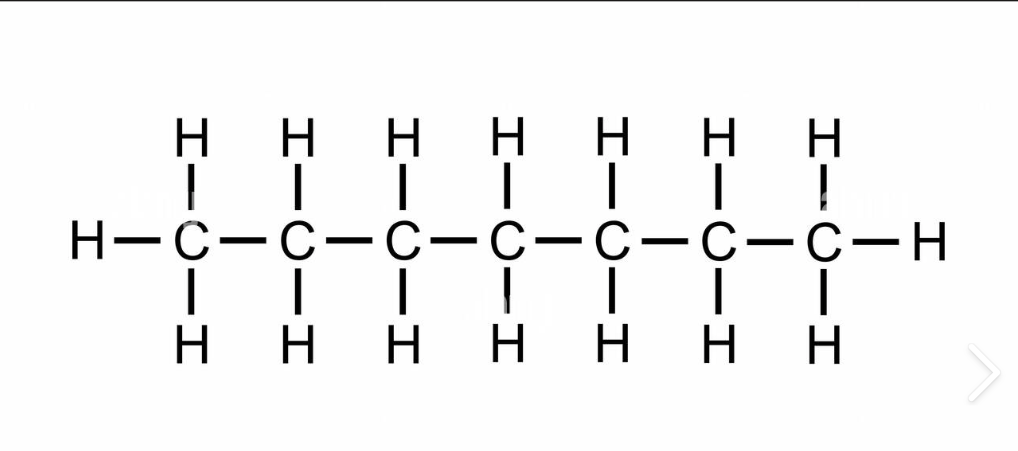
C7H16
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
heptane
9
liquid
heptyl
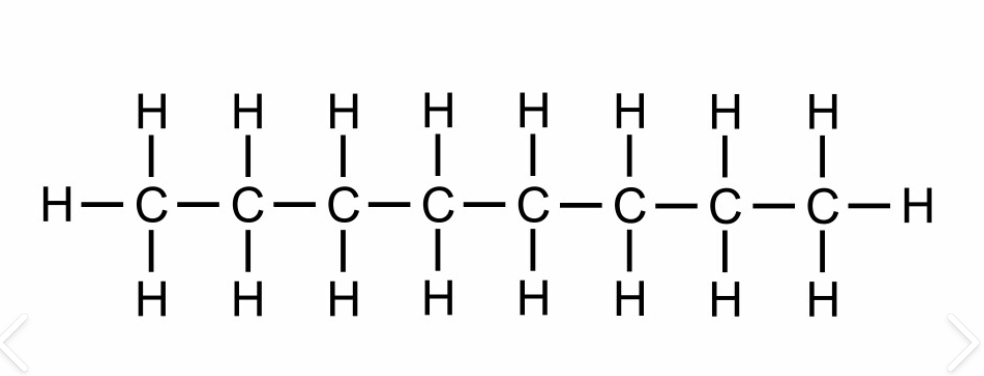
C8H18
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
octane
18
liquid
octyl
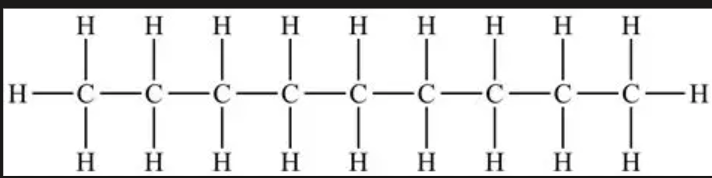
C9H20
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
nonane
35
liquid
nonyl
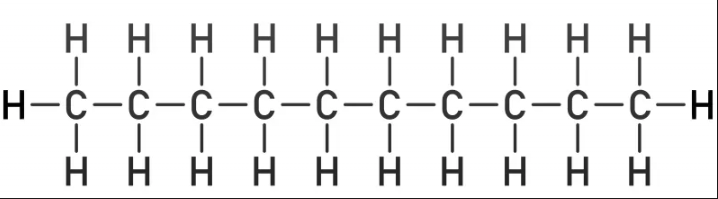
C10H22
straight chain: __
# of isomers ___
liquid or gas
substituent name ___
decan
75
liquid
deyl
__: compounds that have the same # of molecules but differ in the way the atoms are bonded together
if an compound has a ring shape you must place __ before it
if a compound has a cl it is ___
Rules for naming
locate the __ chain and name it. use the chain with the most ——
name the —-
give substituents the lowest # possible
still alphabetized disregarding di and tri etc
alkanes:
Cn +H2n+2
structural isomer
cyclo
chloro
longest substituents
substituents
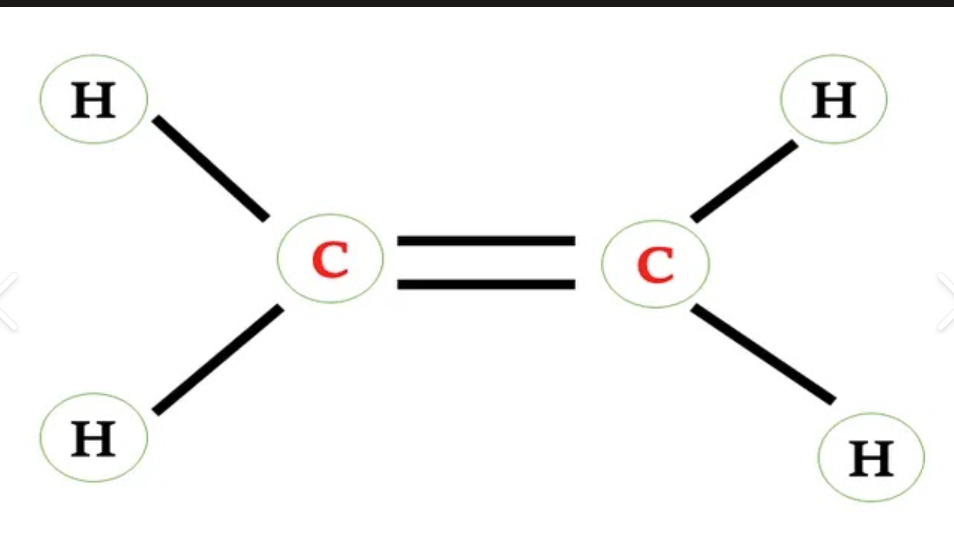
also called olcfine
hydrocarbon that has a c to c double bond
changes ethene to ethane
__ is two double bonds of c
alkane
dienes

has a c to c triple bond
ethane changes to ethyne
must add #-name to denote where the bond is
alkynes
aromatic hydrocarbons