Chemistry Lab 3 Review: Enzyme Inhibitors & Amylase Terms
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
Catalyst
A chemical agent that selectively increases the rate of a reaction without being consumed by the reaction
Most biological catalysts are...
Enzymes
Enzymes
A macromolecule serving as a catalyst; they're typically proteins
How do enzymes work?
They destabilize reactants so that they are more likely to react with each other to form product(s)
How do enzymes help speed up reaction rates?
They bind to one or more reactants (substrates) and hold them in such a way as to make them unstable and more likely to create product, which the enzymes release
We say that the enzyme lowers the activation energy needed for reactants to go to product
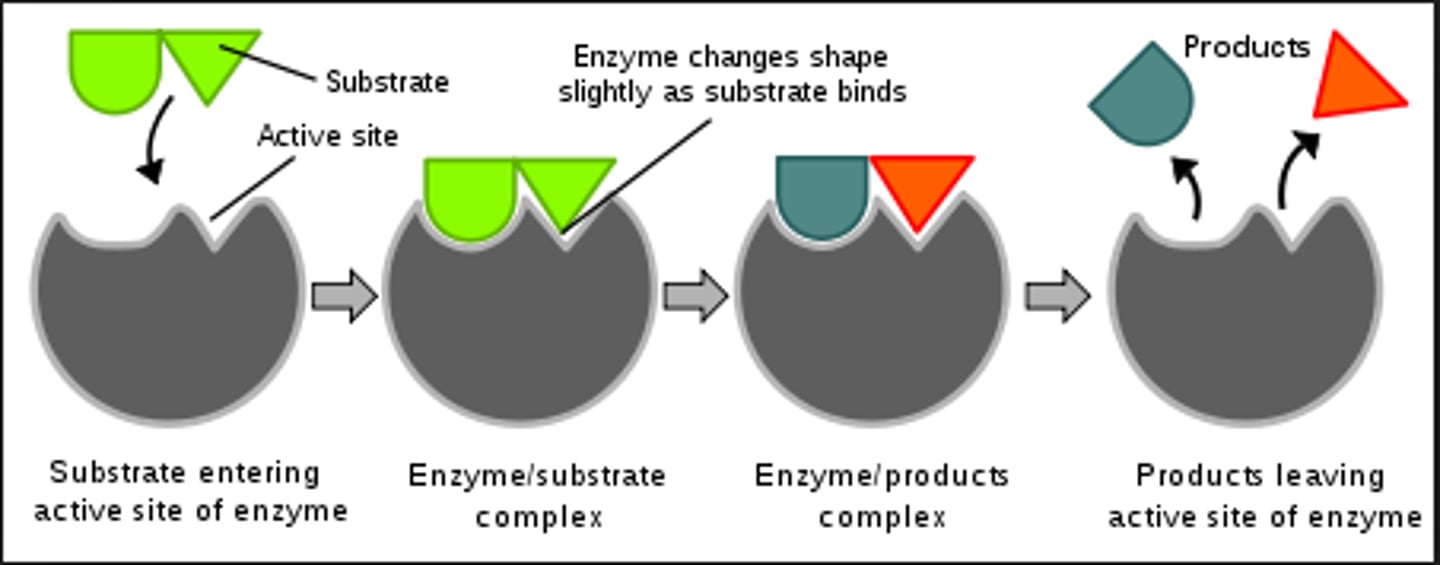
Active site
The location on the enzyme where the substrate binds
Enzyme-substrate complex
Functional groups found on amino acids in the active site interact with the substrate (since, remember, most enzymes are proteins)
The enzyme destabilizes the substrate(s) and allows it to form product(s) much more rapidly than it would on its own
What is the role of an enzyme?
It must lower the activation energy of BOTH the forward and reverse reactions, and so that which reaction is favored, or when the reaction reaches equilibrium, is not determined by the enzyme but by other conditions of the cell (such as concentration of substrate vs. product)
What affects a protein being denatured?
Changes in pH, temperature, or salt concentration. These changes can dramatically affect how an enzyme folds up and can alter its function
Cofactors
Inorganic or organic molecules that help enzymes carry out their catalysis
Coenzymes
Organic cofactors
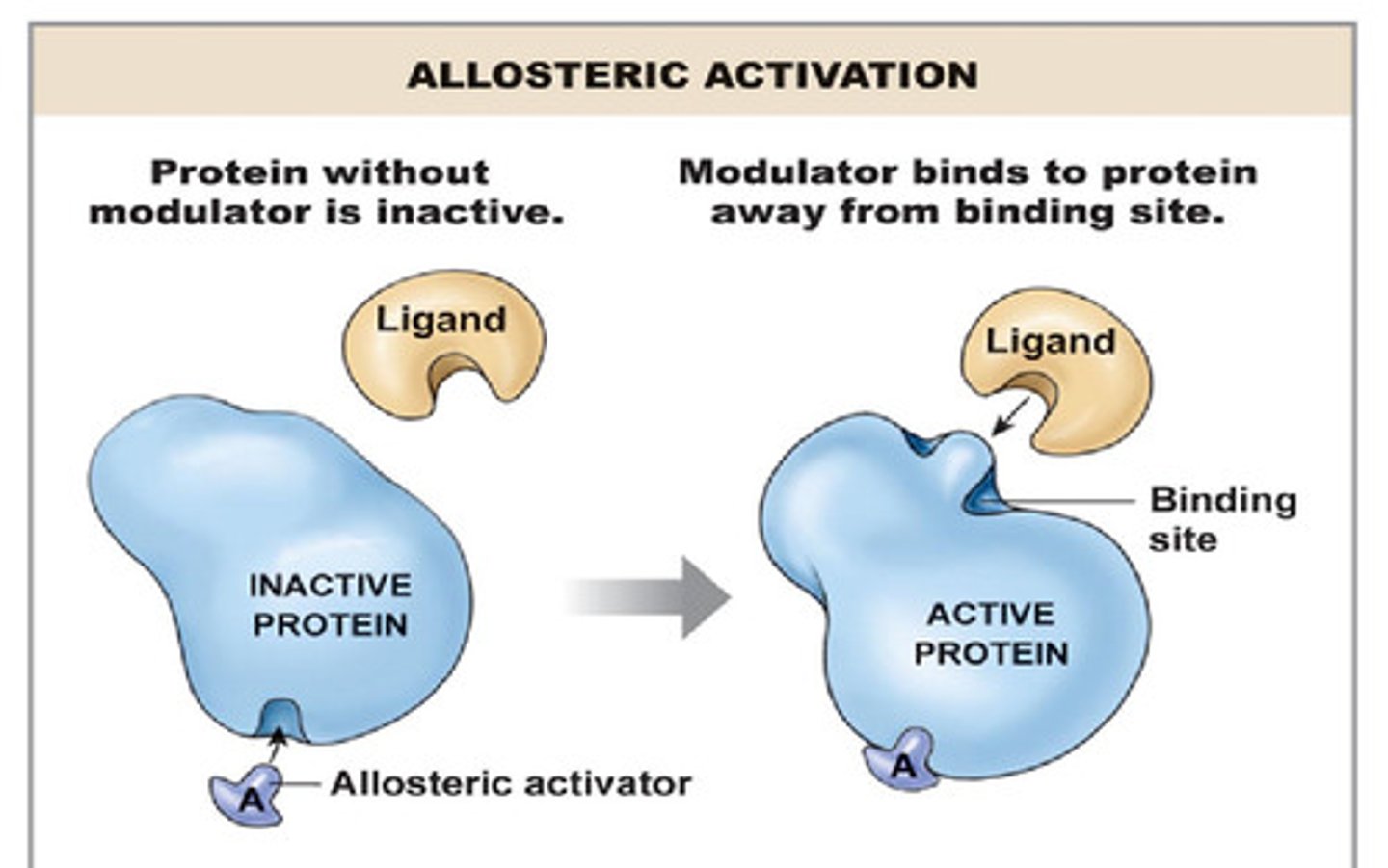
Activators and Inhibitors
Other proteins or chemical compounds that interact with the enzyme to affect its rate of activity
Activators
Bind to the enzyme and increase its reaction rate
Inhibitors
Bind to the enzyme to reduce or block its reaction rate
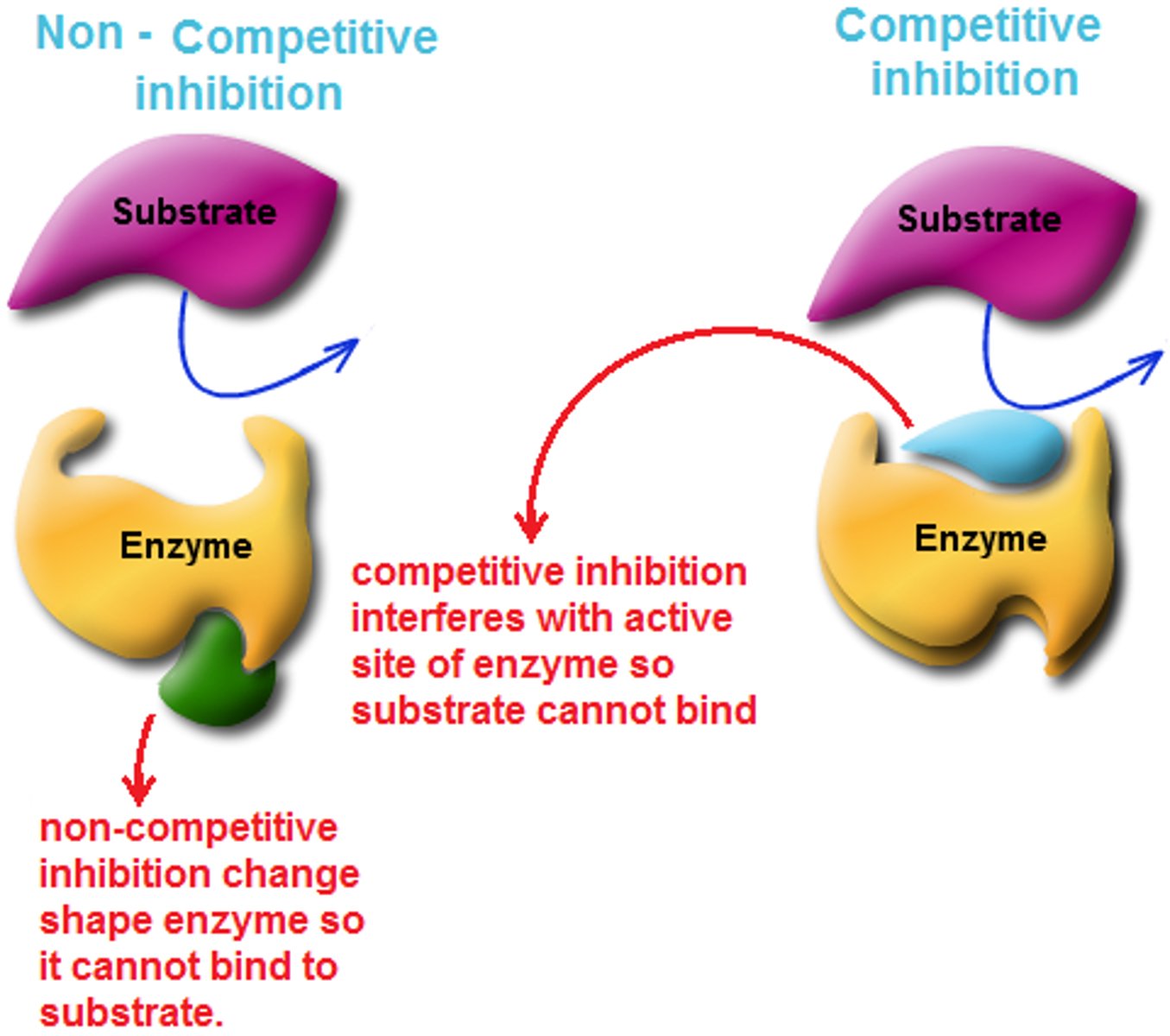
Competitive Inhibitors
Inhibitors that function by binding to the active site to prevent binding of substrate and compete with the substrate
They are often structurally similar to the real enzyme's substrate, which is why they fit into the active site.
The amount of substrate affects inhibition (more substrates = lower chance of inhibition)

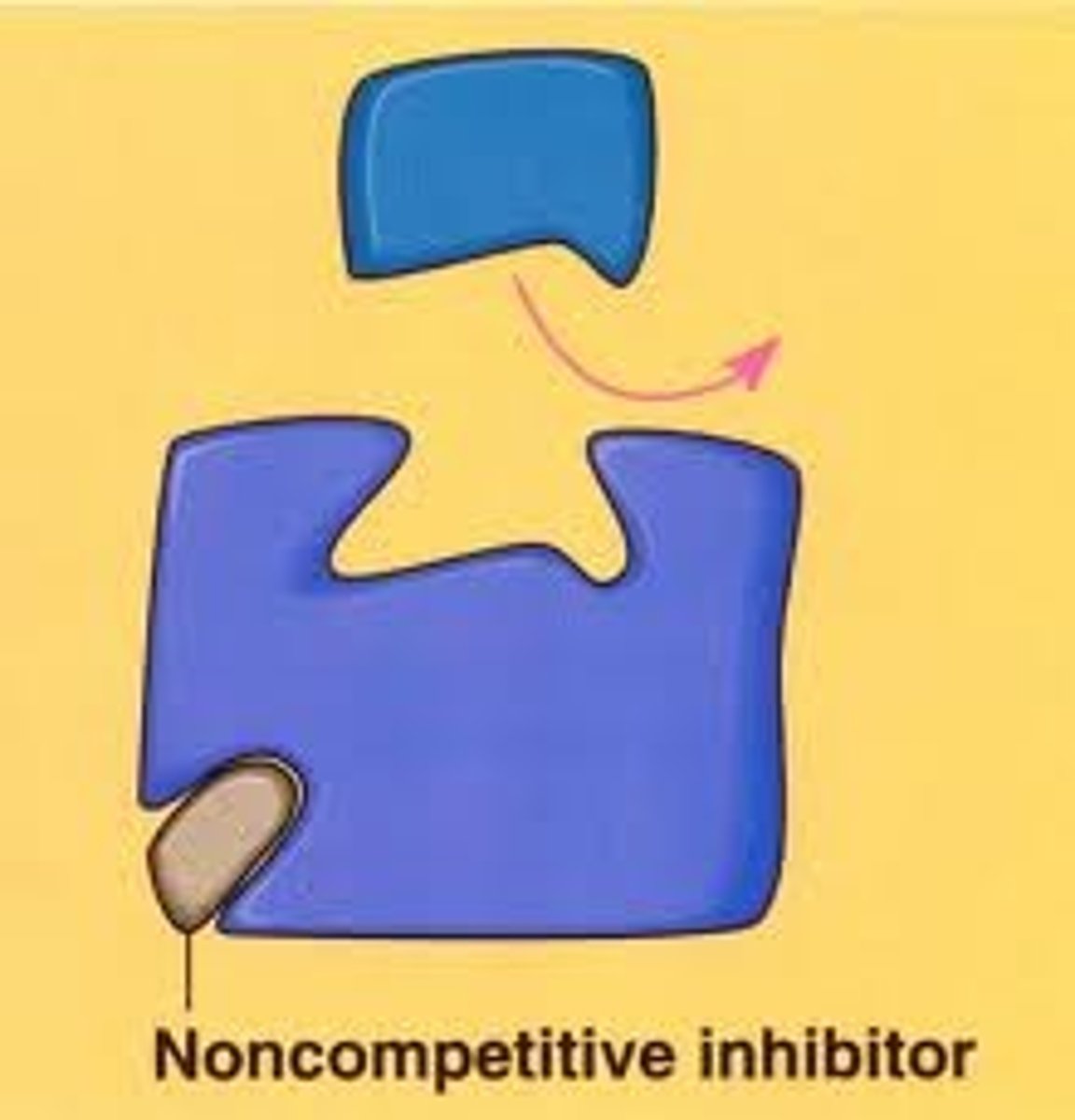
Non-Competitive Inhibitors
Also known as allosteric inhibitors, they function by binding to regulatory sites (distinct from the active site) on the enzyme and changes the shape of the enzyme so that it can no longer work properly. It may also bind near the active site to block it.
These compounds typically do not share structural similarities with the enzyme's normal substrate
The amount of substrate is irrelevant to inhibition
What are the two ways to measure enzyme activity?
1) Determine the rate of disappearance of the substrate
2) Determine the rate of appearance of the product
Catechol Oxidase
An colorless enzyme that catalyzes the removal of electrons and hydrogens from catechol
Catechol
A phenolic compound found in plant cells that is clear/light yellow (mostly clear)
Benzoquinone
A dark purplish-brown product that is the result of catechol being catalyzed by catechol oxidase. It is responsible for the darkening of fruits and vegetables, such as apples and potatoes, after exposure to air
In our lab, what did we consider potato extract to contain only?
Catechol oxidase
In reality, potato has both!
What are the products of a reaction catalyzed by catechol oxidase in the presence of oxygen?
Benzoquinone and water (hydrogens combine with oxygen)
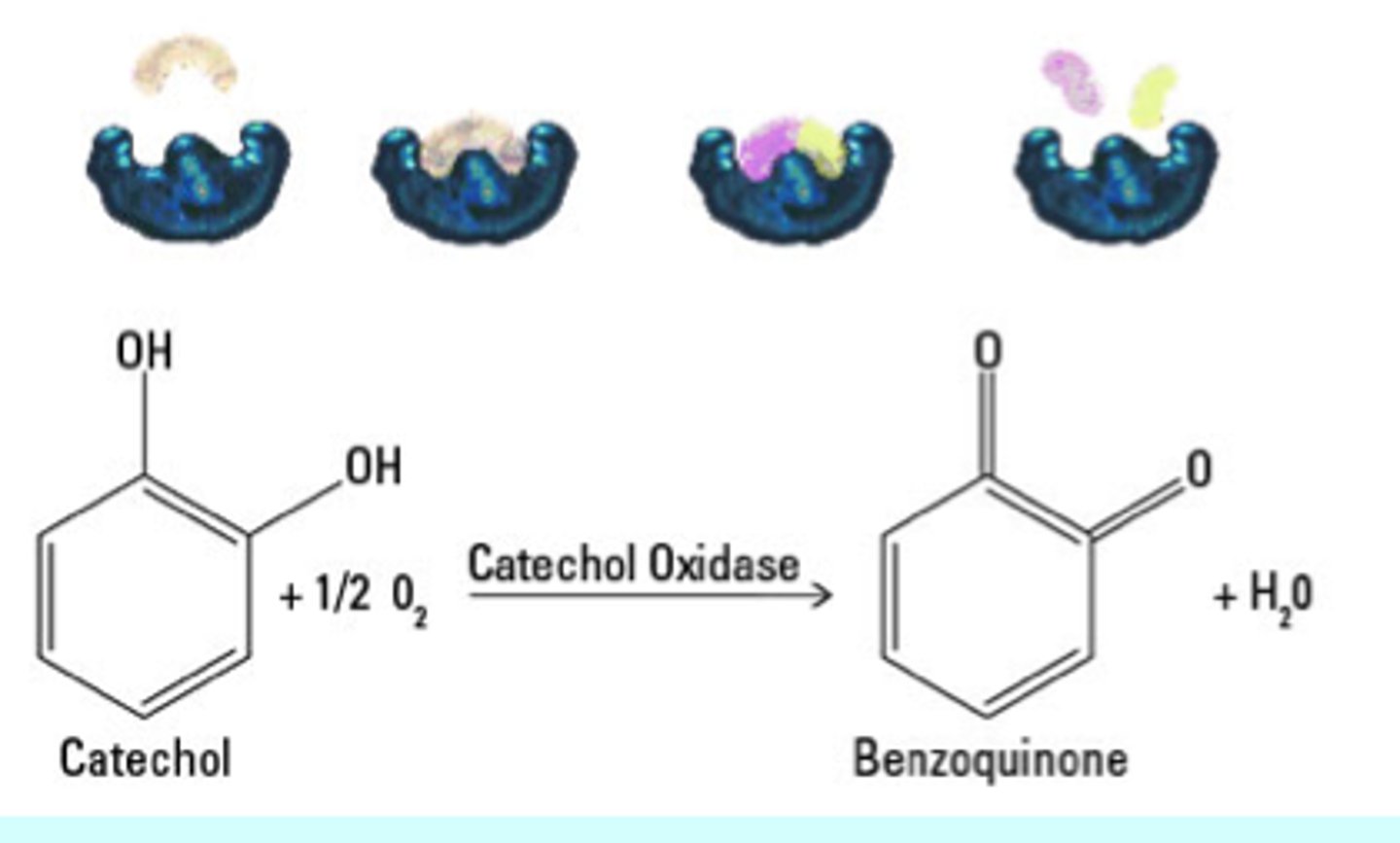
T/F Catechol is poisonous
True - Avoid contact with all solutions!
Exercise 1. Examining the Activity of Catechol Oxidase
Your goal is to use an extract of potato tuber to test for the presence of catechol oxidase and observe the appearance of benzoquinone product when the reaction takes place.
1) Form a hypothesis about varying concentration effects
2) Prepare 4 experimental tubes. Note that all tubes should contain the same total amount of solution. Use the 5mL pipette with the green pump to measure the DI water.
Tube 1
5mL Distilled water
8 drops Catechol
8 drops Distilled water
- Potato extract
Tube 2
5mL Distilled water
8 drops Catechol
- Distilled water
8 drops Potato extract
Tube 3
5mL Distilled water
- Catechol
8 drops Distilled water
8 drops Potato extract
Tube 4
5mL Distilled water
4 drops Catechol
4 drops Distilled Water
8 drops Potato extract
3) Use the rubber stopper to plug the tube then rotate the tube to mix the contents thoroughly
4) Observe the reactions in the tubes
5) Empty the test tube contents in the Catechol Waste container near the sink. Rinse tubes in sink and return to station tray
Result:
Tube 1 had no color change (-)
Tube 2 had strongest color change (+)
Tube 3 had no color change (-)
Tube 4 had color change (+)
The color change of the reactions indicate the presence of catechol oxidase. Tube 1 had no color change because catechol oxidase is contained in potato extract, and no extract was added to it. Tube 3 also had no color change because there was no catechol, so what would the oxidase react with?
Tube 2 had the strongest color change because benzoquinone concentration was not saturated by distilled water, and also the probability of catechol molecules interacting with catechol oxidase molecules was higher (due to more being added). Tube 4 had color change due to catechol oxidase and catechol presence, but the concentration is diluted due to less catechol added + distilled water added.
Phenylthiourea (PTU)
A catechol oxidase inhibitor
What is the cofactor used by catechol oxidase?
Copper
How does PTU work?
PTU is known to react with copper in catechol oxidase, resulting in the enzyme's catalytic ability
In our lab, was PTU competitive or noncompetitive?
Noncompetitive
Exercise 2. Inhibition of Catechol Oxidase
Your goal is to figure out if PTU is noncompetitive or competitive
1) Form a hypothesis
2) Prepare three experimental tubes. Be sure to add solutions in the sequence given (water first, potato extract next, PTU next, distilled water next, and catechol last)
Tube 1
5mL Distilled water
8 drops Potato extract
8 drops PTU
8 drops Distilled water
8 drops Catechol
Tube 2
5mL Distilled water
8 drops Potato extract
8 drops PTU
- Distilled water
16 drops Catechol
Tube 3
5mL Distilled water
8 drops Potato extract
- PTU
16 drops Distilled water
8 drops Catechol
3) Record observations
4) Empty test tube contents into Catechol Waste container. Rinse tubes in sink and return to station tray
Results:
Tube 1 color was slight pink (-)
Tube 2 color was slight pink (-)
Tube 3 color was dark purplish-brown [benzoquinone] (+)
Tube 1 and 2 had the same color, despite having different concentrations of catechol and potato extract. Tube 3 was the control, which showed what color ought to be if catechol was catalyzed. Because Tube 1 and 2 shared the same color, this indicated that concentration of the substrate did not affect the inhibitor from working. Therefore, our conclusion is that PTU is noncompetitive.
Theoretically, if PTU was competitive, the likeliness that Tube 2 would be + would be near always because there was so much substrate added compared to inhibitors. However, since Tube 2 is -, it means that no matter how many substrates are present, the noncompetitive inhibitor would prevent the substrates from binding by changing the overall shape of the enzyme,
How can the rate at which a chemical reaction takes place often be increased?
By increasing the temperature of the reaction, the molecules are moving faster, so reactants are more likely to bump into each other and react to form product.
For many enzymes, small increases in temperature can also help them bind their substrates and increase their rate of reaction.
However, be aware that every enzyme has optimal temperatures (and pH) that they work best in. If the temperature reaches a point too high, this can disrupt the bonds that help maintain a protein's secondary, tertiary or quaternary structure, and so the protein can denature and stop functioning!
What can changes in pH and/or salt concentrations do?
It can alter structure of an enzyme and cause it to denature
Amylase
An enzyme that breaks down starch polymers (which is abundant in plants we eat) into sugars
What are the two major types of amylase that mammals can have?
Salivary and pancreatic amylase
Salivary amylase
Secreted by the largest of the human salivary glands (the parotid gland), where it is one of the first digestive enzymes to work on breaking down the food you eat
Pancreatic amylase
Made by the pancreas, and is released into the digestive tract to further process starches into sugars
How do salivary and pancreatic amylase work?
Both enzymes catalyze the reaction in which water is used to break the alpha 1-4 bonds between glucose molecules that form starch polymers. They release maltose and glucose as products
As starch is being broken down by amylase, how would that affect I2KI being added to the solution?
There would be less starch to react with I2KI, changing the color of the solution.
A pure starch solution should be very dark when I2KI is added.
What could we use the change in color of the starch solution to measure?
We could measure the rate of reaction of amylase as it breaks down the starch (substrate) into sugars (product)
Exercise 3.1. Effects of Concentration on the Activity of Amylase
PART 1
Part 1: Purely making dilution solutions
TEST TUBE SET 1
1) Form a hypothesis
2) Prepare the amylase dilution (test-tube set 1)
3) Using the 5mL serological pipette, add 5mL distilled water to each test tube
4) Swirl the amylase flask in order to make sure that the enzyme has not settled to the bottom of the flask. Make serial dilutions as follows
Tube 1: Add 5mL of 0.5% amylase and mix by rolling the tube between your hands (Dilution 1:1, 0.25% amylase)
Tube 2: Add 5mL amylase solution from tube 1 and mix (Dilution 1:3, 0.125% amylase)
Tube 3: Add 5mL amylase solution from tube 2 and mix (Dilution 1:7, 0.063% amylase)
Tube 4: Add 5mL amylase solution from tube 3 and mix (Dilution 1:15, 0.031% amylase)
5) Rinse serological pipette
Exercise 3.1. Effects of Concentration on the Activity of Amylase
PART 2
Part 2: Analyzing 1mL of each dilution solution
TEST TUBE SET 2
1) Beginning with tube 4 of the first set, transfer 1 mL of this dilution into tube 4A of the second set. Use the 1-mL pipette with blue pump for the transfer.
2) Repeat for all the other tubes. Rinse pipette after each use. After these transfers, test tube set 1 will no longer be used
3) Add 20 drops pH 6.8 buffer solution to each tube in the second set. This stabilizes the pH to focus only on concentration. Mix by rolling tube between your hands and set aside
4) Add 1-2 drops of I2KI to each compartment of four rows of a test plate. You will use a separate row for each concentration of amylase
5) Test the starch solution by adding one drop of STARCH (a different solution) into the first compartment of your test plate. If the reaction does not turn blue immediately, it may mean your starch has been contaminated with amylase and will need to be replaced
6) Using the second set of tubes, proceed tests beginning with 4A
7) Using the clean 1-mL pipette, add 0.5mL of the 1% starch solution to tube 4A and mix by rolling the tube between your hands. This is time 0
8) Quickly remove 1 drop of the mixture with the plastic pipette and add it to a drop of I2KI in the first compartment of the test plate. When the enzyme and substrate are together, the reaction has begun!
9) Measure in 10-SECOND INTERVALS and continue until the blue color is no longer produced. If the solution remains yellow-amber, this means that the starch has been completely digested
10) Repeat steps for other concentrations
Result: As then amylase concentration increases, the time it took for all the starch to be digested decreases. More amylase = more digestion!
Exercise 3.2. Effect of pH on the Activity of Amylase
Warning: Buffers can burn skin! Avoid contact
1) Form a hypothesis
2) Use a 5mL graduated pipette with a green pump to add 5mL of appropriate buffer to each tube
It should be pH 5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0 (5-8 pH with 0.5 increments)
3) Using the 5mL graduated pipette with green pump to add 1.5mL amylase solution to each tube and mix by rolling tubes in your hand
4) Add 1-2 drops of I2KI to each compartment of four rows of a test plate.
5) Using only tube 5.0, add 2.5mL of the 1% starch solution with a clean 5mL pipette. Mix by rolling tube. This time is 0. Start testing immediately
6) Test the starch solution by adding one drop in the first compartment. It should turn blue immediately
7) Using a plastic pipette, remove a drop of the reaction mixture from tube 5.0. Add a drop of I2KI on the test plate
8) Sample the reaction mixture at 10-SECOND INTERVALS, each time using a new compartment of the test plate. Continue until the blue color is no longer produced (indicating digestion of all starch)
9) Record the time required to digest all starch. If after 7 minutes there is no color change, terminate the experiment with that reaction mixture. This is because the pH would've completely denatured and be ineffective. Continuing would just result in more blue solution because digestion has stopped.
10) Repeat steps
Result: The optimal pH is between 6.9-7.0.
Exercise 3.3. Effects of Temperature on the Activity of Amylase
1) Form a hypothesis
2) Prepare test tubes 1-4. Using the 5mL pipette and green pump, add 2mL of the 1% starch solution to each tube
3) Using a 5mL pipette, add 4mL of DI water to each tube
4) Using the 1mL pipette, add 1mL of pH 6.8 buffer to each tube
5) Prepare test tubes 1A-4A. Use the 1mL amylase to each tube
6) Place the test tubes as follows:
Tube 1 & 1A: 80*C water bath
Tube 2 & 2A: 37*C water bath
Tube 3 & 3A: Room temp (22*C) on tube racks
Tube 4 & 4A: 4*C ice bath
7) Let tubes sit for ~10 mins
8) Fill rows with I2KI
9) Test starch
10) Leaving the tubes in the above environments, mix 1 and 1A and record time (time 0), and use plastic pipette to immediately and drops to I2KI
11) Continue adding mixture drops at 30-SECOND INTERVALS until blue color is no longer produced. If within 10 minutes there is no color change, terminate the experiment with that particular reaction mixture. The temperature could've completely denatured the enzyme at that point, and would be ineffective, meaning starch would no longer be digested
12) Repeat steps for ALL tubes. For tube 4, after 10 minutes have passed OR the starch has been completely digested (unlikely), proceed to step 13
13) Transfer tube 4 (in ice, containing enzyme and substrate), into the 37*C water bath. After 2 mins, test the contents at 10-SECOND INTERVALS.
Result: In reality, the optimal temperature is 32C-37C (since this is our body temperature).
For the tube 4, the colder temperature makes the starch digestion slower, and the temperature itself denatures the enzyme. However, when you reheat to 37*C, it activates the enzyme again, and while heating the starch is digested, so when you try the 10-second interval drops, the starch is theoretically completely digested (if it isn't, then it would digest faster).
What color pump do you use for 1mL pipette?
Blue
What color pump do you use for 5mL pipette?
Green
What color pump do you use for 25mL pipette?
Red
Clean up Procedure
1) Empty contents of test plate into the Iodine Waste bucket. You need to shake the plate to get the solutions out of the wells. Rinse and place upside down on towel to dry
2) Test tubes with amylase and starch can be dumped in sink and rinsed. Place in test tube rack along with small test tubes so they can dry
3) Dump waste water beaker contents in sink
4) Organize station tray so it's neat