O Chem 2 Exam 4
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Tautomerization
process that converts ketones to enols and reverse. Can be acid or base catalyzed.
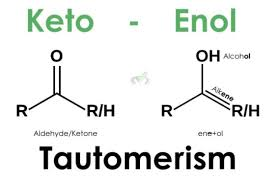
Conditions for when enol is present in larger quantities (ketone is usually more stable except when..)
Enol creates aromaticity
Creates new conjugation
Hydrogen bonds make 5 or 6 membered ring
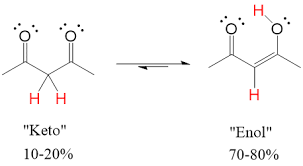
Enolate
when hydrogen from alpha carbon is removed by strong base
highly reactive
resonance stabilized
nucleophiles
kinetic enolate
strong non-nuclephilic base (LDA at low temp)
easiest/fastest hydrogen is removed
forms on most accessible side
faster than thermodynamic enolate
short reaction time
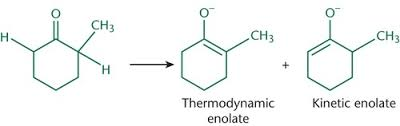
Thermodynamic Enolate
weaker base (alkoxides)
Long reaction time NEEDED, forms slower than kinetic
requires reversible conditions
most stable enolate
hydrogen removed on more substituted side
Carbonyls with Br2, Cl2, or I2
ACIDIC conditions
One halogen is added to More substituted side
Carbonyls with Br2, Cl2, or I2
BASIC conditions
Substitutes ALL hydrogens for halogen
Cannot stop at 1 addition will make two products if not in excess
Adds to LEAST substituted side
Haloform Reaction
ketone with NaOH, Br2/I2
forms carboxylic acid and hcX3
LDA
X2
ONE halogen added to least substituted side
LDA
R—X
Adds carbon group to least substituted side through kinetic enolate, also works on nitriles \
Must be strong base (LDA)
1-3 dicarbonyl compounds
middle proton is most acidic
always remove middle hydrogen for enol/ate formation
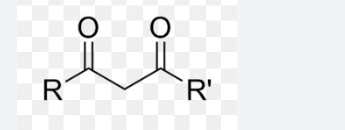
br2, can be H+ or NaOH, do opposite sides
followed by pyridine
does E2 reaction
Adds Br depending on acid or base conditions
pyridine does e2 makes double bond with carbon next to where Br was.