Alpha Carbons
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
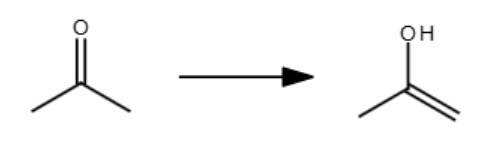
H3O+ or OH-
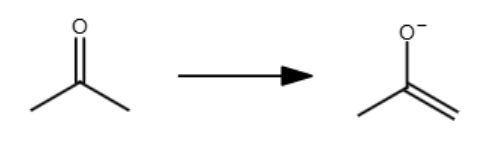
The double bond reforms at the _ substituted carbon.
NH2-
most
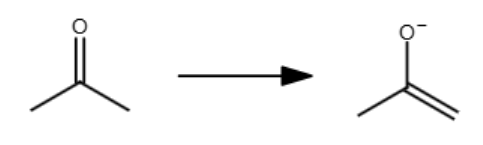
The double bond reforms at the _ substituted carbon.
LDA
least
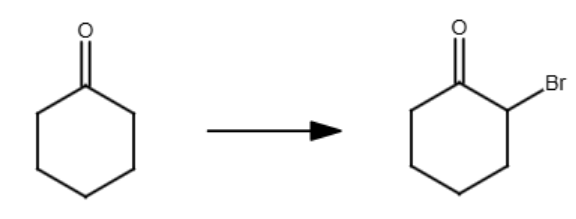
Br2, H3O+
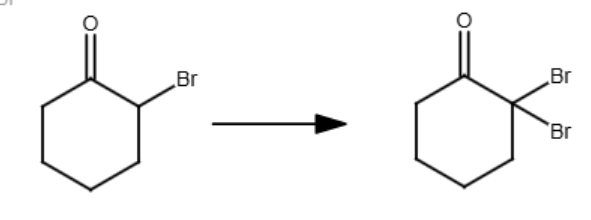
Br2, OH-
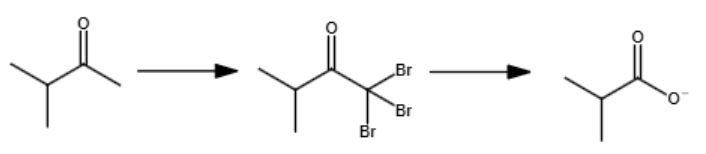
Excess Br2 and OH-
OH-
Only works for methyl ketones
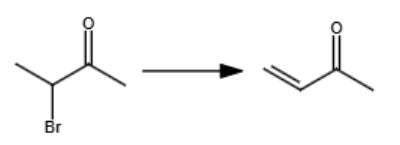
OH-
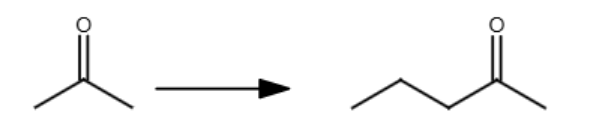
LDA
Br-ethyl
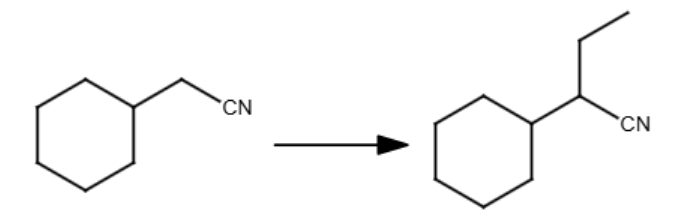
LDA
Br-ethyl
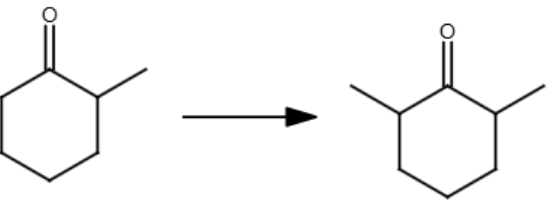
(109)
LDA
Br-CH3
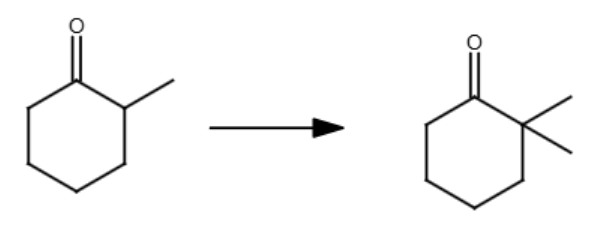
NH2-
Br-CH3
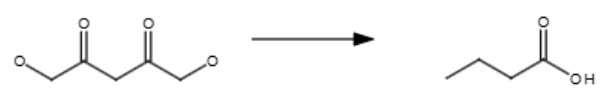
1/3
NaOEt (mild base)
C2-Br
H3O+
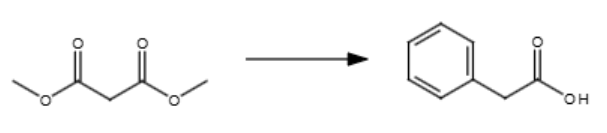
2/3
NaOEt (mild base)
methylbenzene
H3O+
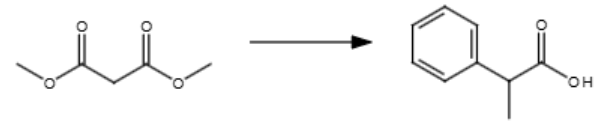
3/3
NaOEt (mild base)
Br-C
NaOEt (mild base)
Br-methylbenzene
H3O+

NaOEt (mild base)
methylbenzene
H3O+
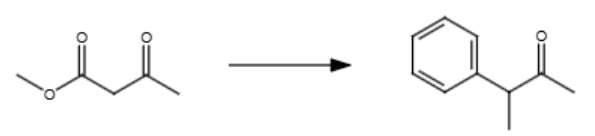
NaOEt (mild base)
Br-C
NaOEt (mild base)
Br-methylbenzene
H3O+
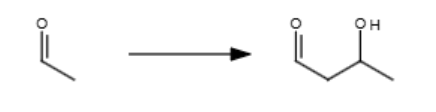
(111) 1/2
NaOH
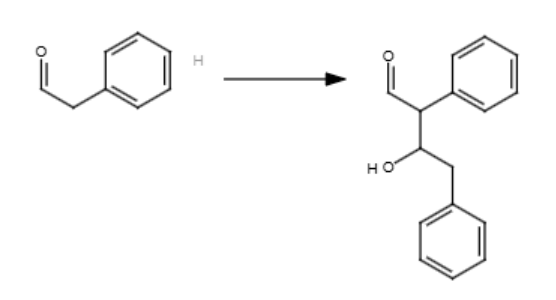
2/2
NaOH
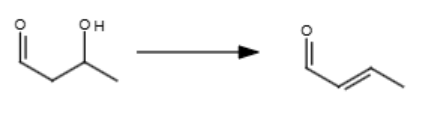
1/3
NaOH, heat
If you want to do this mechanism between two different reactants, make sure one of them doesn’t have an alpha carbon.
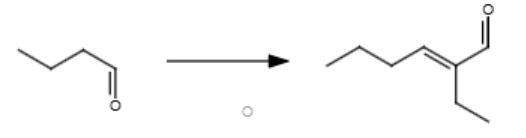
2/3
NaOH, heat
If you want to do this mechanism between two different reactants, make sure one of them doesn’t have an alpha carbon.

3/3
NaOH, heat

LDA (prevents molecules from interacting with each other)
picture
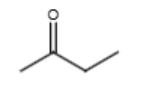
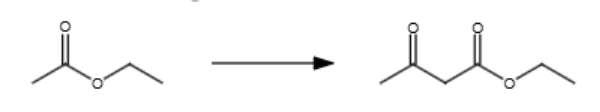
NaOEt
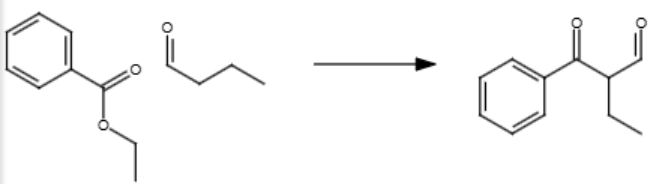
NaOEt, heat
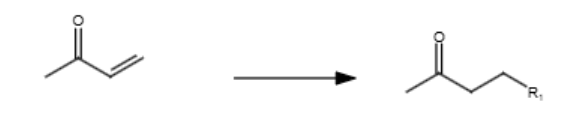
R = nucleophile
nucleophile
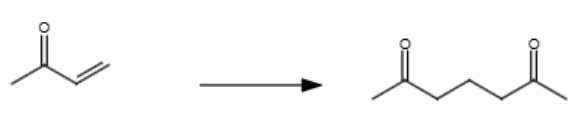
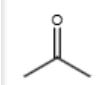
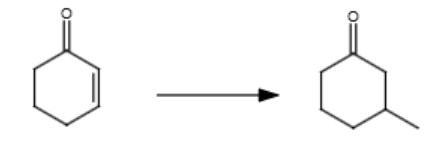
1/2 (116)
(CH3)2CuLi, (aqueous workup)
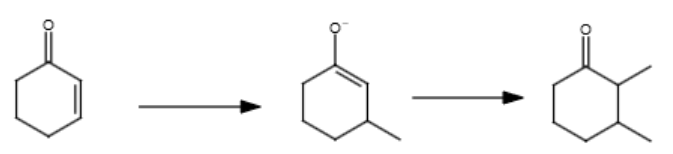
2/2
(CH3)2CuLi
Br-CH3 before aqueous workup

1/2 118 practice 119
NaOH, heat
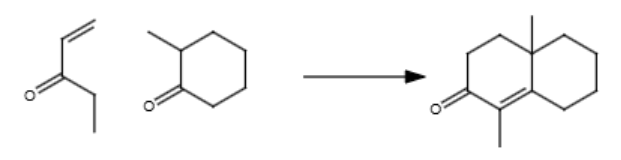
2/2 188 practice 119
NaOH, heat