3.3.16 chromatography
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
what is chromatography used to do?
to separate mixtures of substances into their individual components
all forms of chromatography work on what?
on the same principle
what needs to happen before chromatography can be performed?
the mixture must be dissolved in a solvent (or carried by a gas in gas-liquid chromatography) → this is called the mobile phase
what are the two parts a chromatography run is made up of?
a stationary phase
a mobile phase
outline the stationary phase
a solid (TLC + column chromatography) or liquid (gas-liquid chromatography) that the mixture can travel across
the stationary phase stays still whilst the mobile phase travels across it
what is the movement of the mixture in the stationary phase related to?
the more attracted a portion of a mixture is to the stationary phase, then the slower it moves
(retention can also be used to describe attraction)
outline what the mobile phase is + what happens
a liquid (TLC + column chromatography) or gas (gas-liquid chromatography)
with liquid, the mixture is dissolved in a solvent → this is called an eluent
with gas, the flow of gas carries the mixture
the mobile phase flows across the stationary phase + carries the components of the mixture with it
what is the movement of the mixture affected by?
the more soluble a component of the mixture is in the solvent, then the faster it moves with the solvent
different components of the mixture travel at ______ rates across the ______ phase, what does this allow?
different
stationary
this allows the mixture to separate out into its different parts
outline the two factors the separation of the mixture depends on
the attraction of each mixture component to the mobile phase
the more soluble components will travel more quickly with the solvent as it moves across the stationary phase
the less soluble components will travel but not as quickly
the attraction of each mixture component to the stationary phase (retention)
if a component has a very strong attraction to the stationary phase, it will move slowly across the phase with the solvent
if there is a weaker/no attraction to the stationary phase, the component will travel faster with the solvent
so why will the components separate?
because they have a different balance between their solubility in the mobile phase and their attraction to the stationary phase
what will the attraction to the stationary phase + solubility in the solvent depend on?
it depends on polarity
describe how polarity affects attraction to the stationary phase + solubility in mobile phase
polar substances dissolve well in polar solvents/mobile phases
non-polar substances dissolve well in non-polar solvents/mobile phases
polar substances attract well to polar stationary phases
non-polar substances attract well to non-polar stationary phases
*remember ‘like attracts like’
therefore one phase will be ____ and one phase will be ____
polar
non-polar
what are the 3 types of chromatography?
thin layer chromatography (TLC)
column chromatography
gas-liquid chromatography
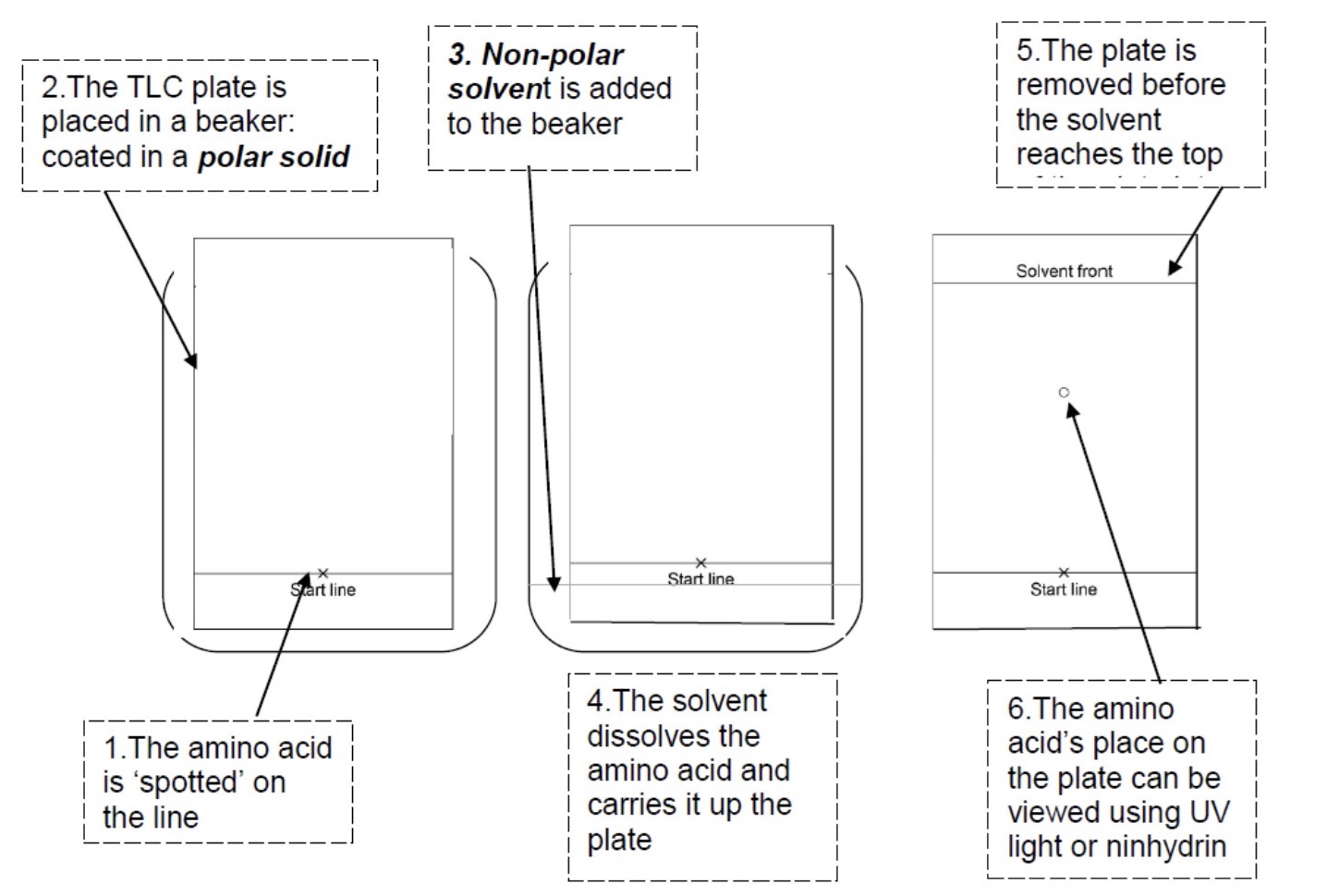
what does thin layer chromatography involve?
it is done using a thin, uniform layer of silica gel or alumina coated onto a piece of rigid plastic
give the stationary and mobile phases of TLC + their polarity
stationary phase: usually silica gel (polar)
mobile phase: is a suitable liquid solvent eg cyclohexane (non-polar)
describe the step by step method of TLC
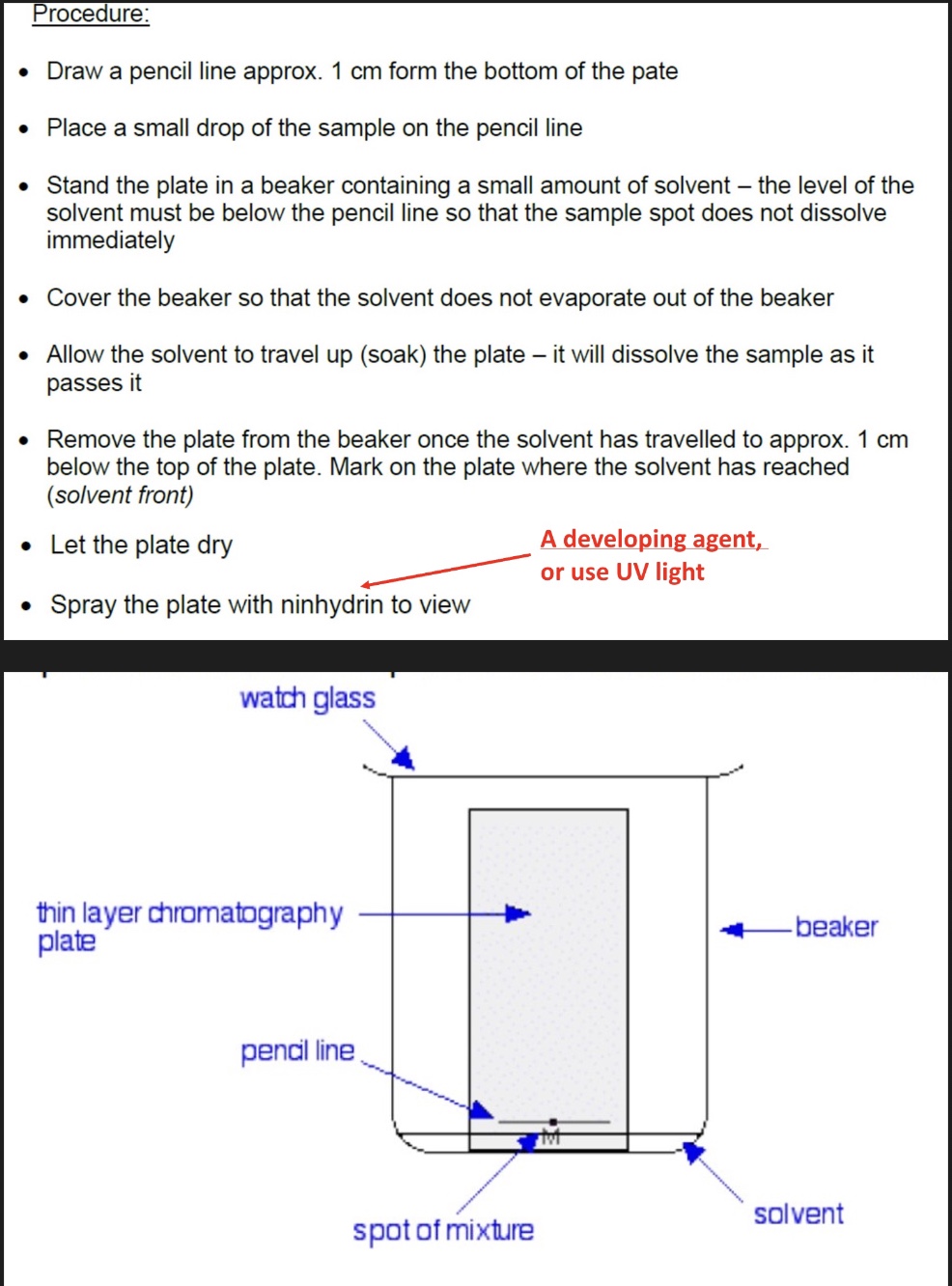
why is pencil used?
because pen would dissolve in the solvent
why is a watch glass used to cover the beaker in TLC?
to stop solvent from evaporating
also used to create a saturated atmosphere of the solvent in the beaker
how can you identify a component of the mixture?
a retention factor or Rf can be calculated for each spot
it can then be compared to known Rf values from a database
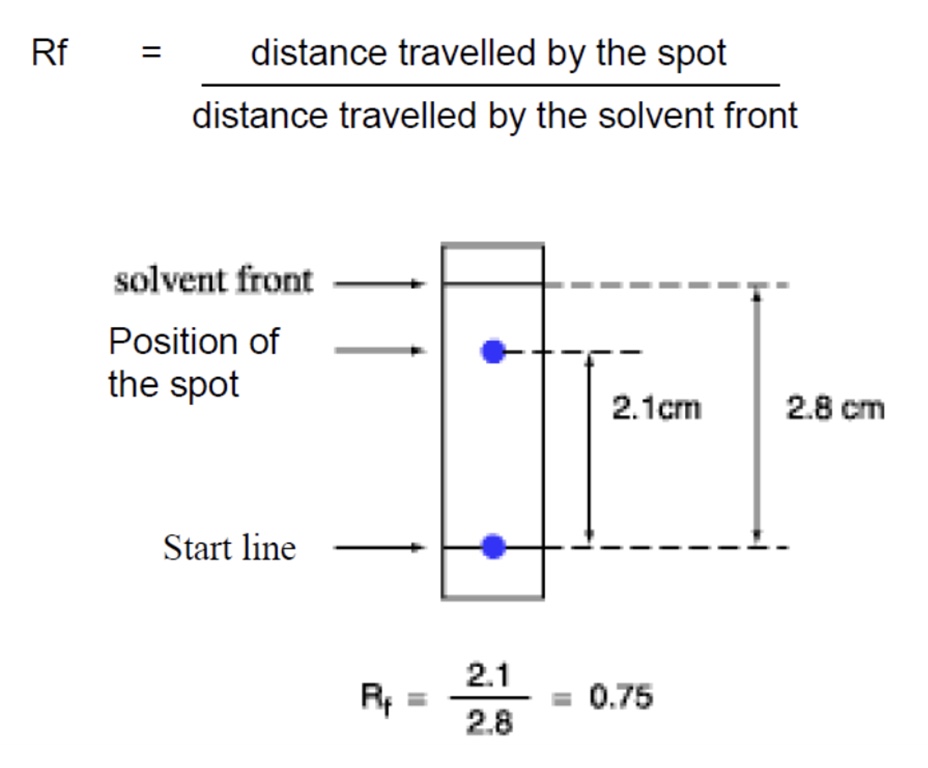
how are Rf values calculated?
value will always be between 0-1
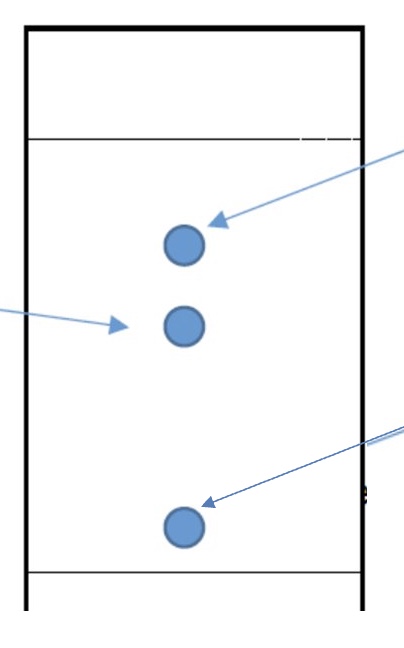
which component is the least polar + why?
which component is the most polar + why?
the top component is the least polar as it has better solubility in the non-polar solvent + less interactions with the polar silica
the bottom component is the most polar as it interacts with the silica more
what is TLC most commonly used for?
to separate a mixture of amino acids
describe how the primary structure of a protein can be determined using TLC

why is two way paper chromatography used?
to get around the problem of separating out substances which have very similar Rf values
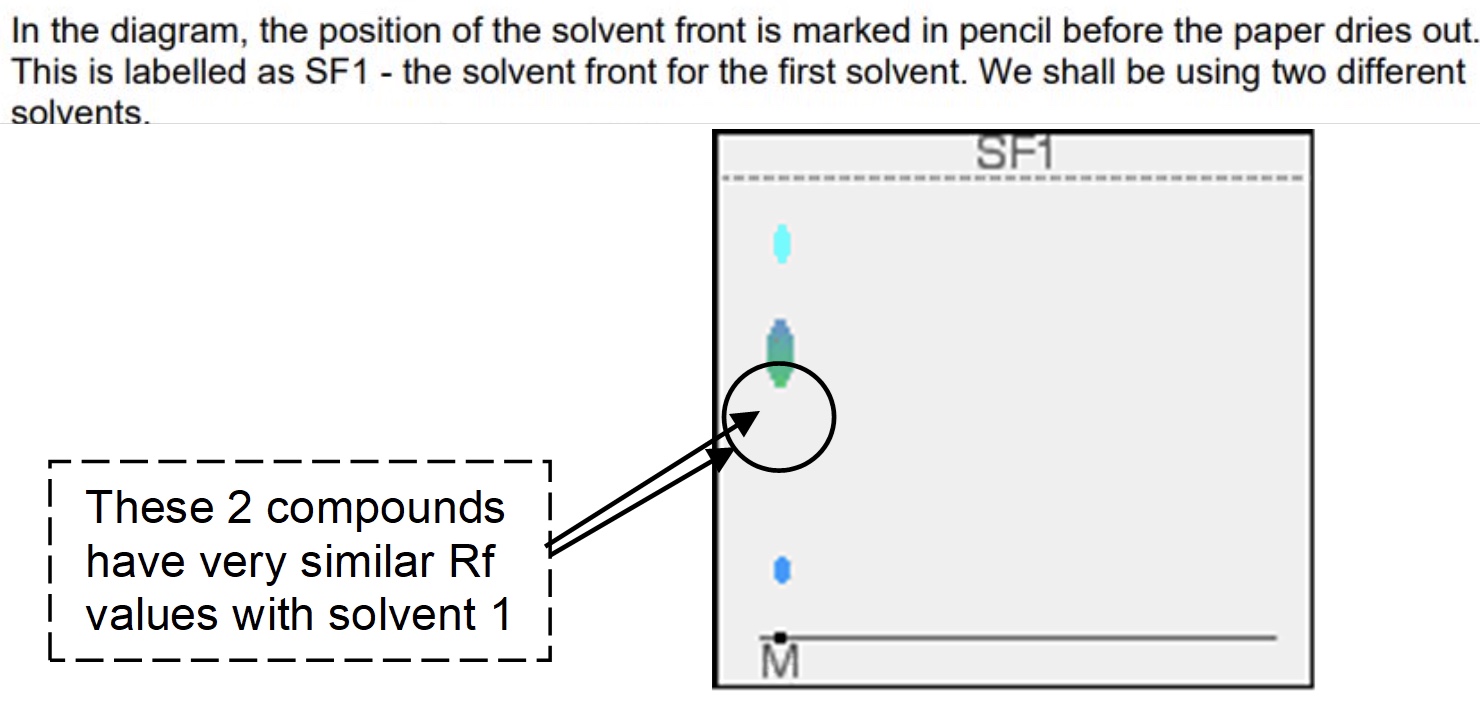
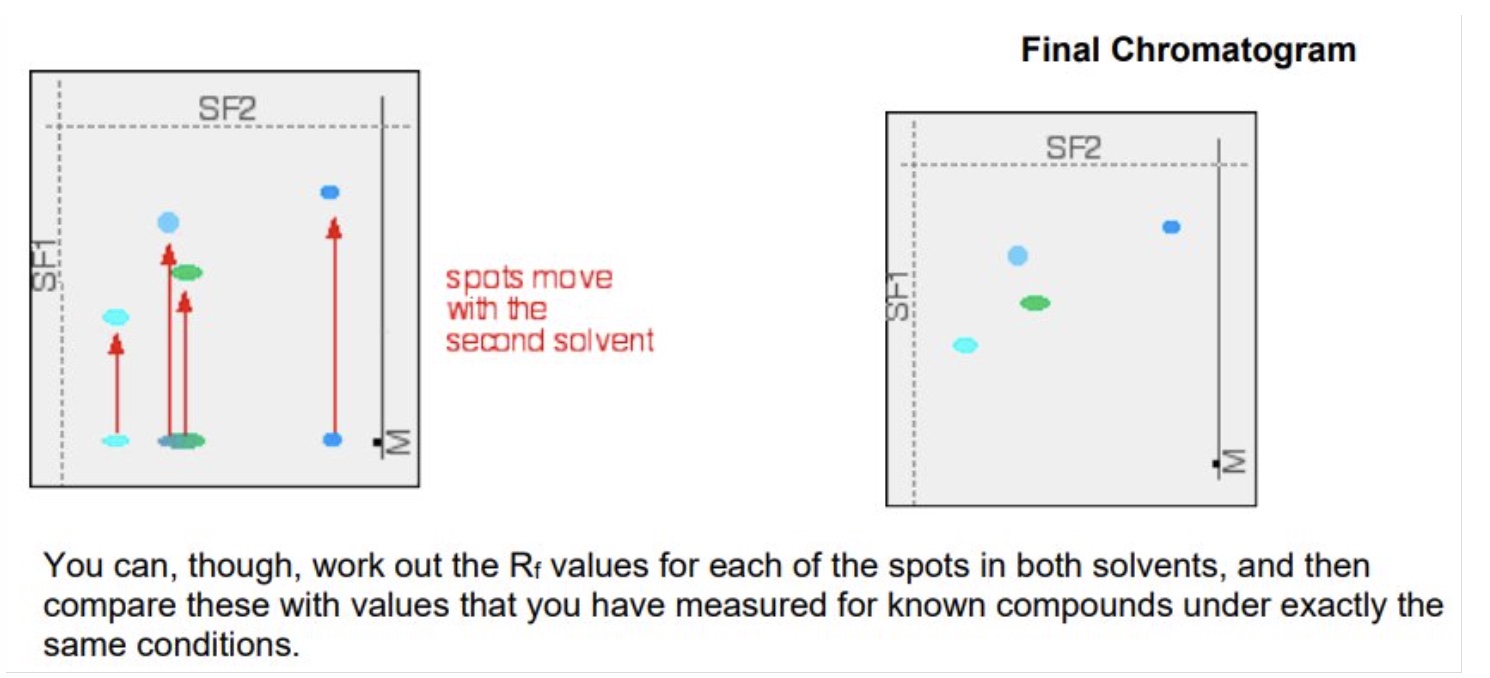
describe the method of two way paper chromatography
a single spot of mixture is placed towards one end of the base line
it is stood in a solvent as before + left until the solvent front gets close to the top of the paper
wait for the paper to dry out completely + then rotate it through 90° + develop the chromatogram again in a different solvent
it is very unlikely that the two similar spots will have the same Rf values in the second solvent as well as the first so the spots will move by a different amount
outline what column chromatography involves
a thin, vertical column (similar to a burette) packed with silica + the sample solution is poured through it
the solvent is added periodically from the top + is known as the eluent (mobile phase)
give the stationary and mobile phases + their polarities of column chromatography
stationary phase: silica gel (polar) within the column
mobile phase: is a suitable liquid solvent eg cyclohexane (non-polar)
describe the method of column chromatography
the mixture moves down the column + the different components travel at different speeds according to the balance between their solubility in the eluent + their affinity/interaction with the silica powder
this means with a beaker at the bottom we can collect each portion separately at the bottom
if one component of the mixture does not move down the column then a different solvent can be used that may better match the polarity of the molecule
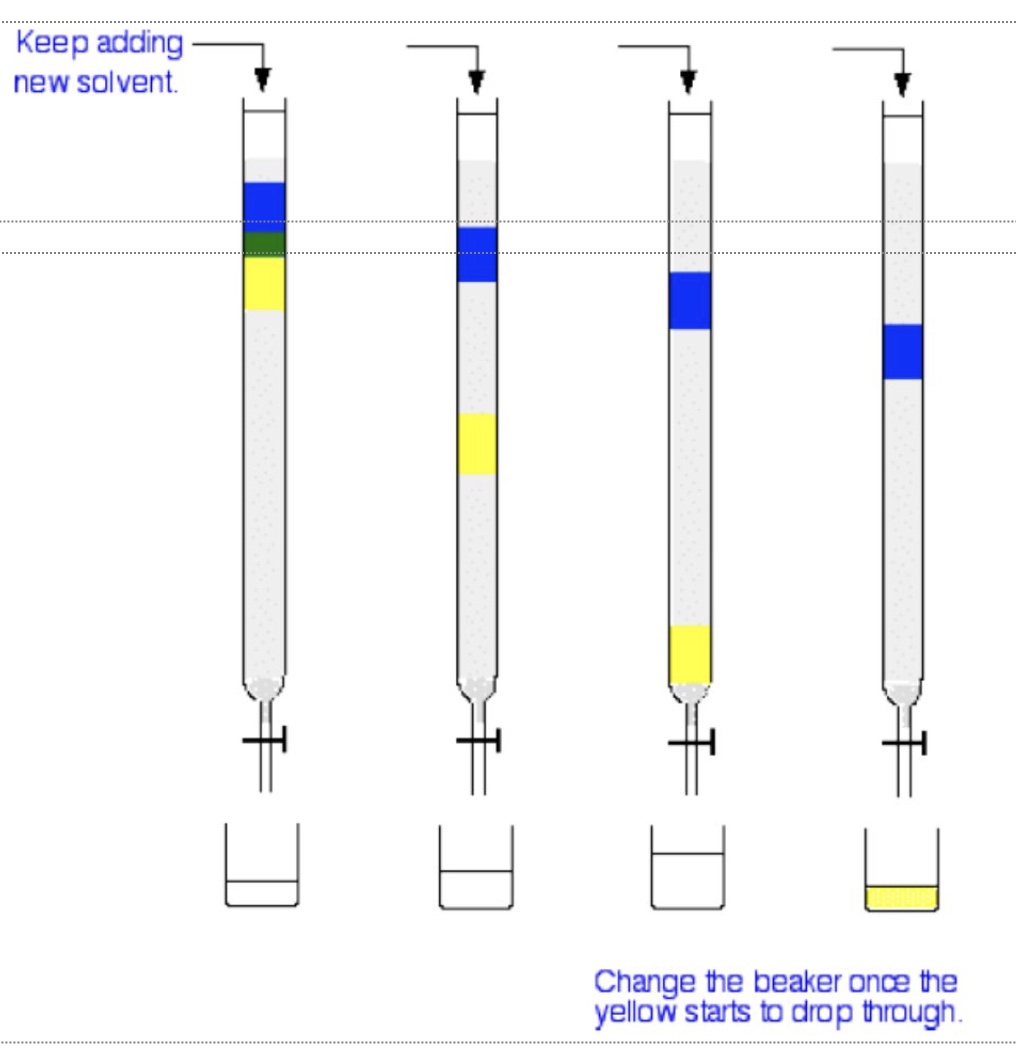
which compound is most polar + why?
the blue compound is the most polar as it interacts with the silica better
give the stationary + mobile phase of gas-liquid chromatography + their polarities
stationary phase: a thick viscous liquid, such as an oil (non-polar) or silica gel (polar) coated on the lining of the column
mobile phase: an inert gas eg nitrogen or helium (it carries the components through the column)
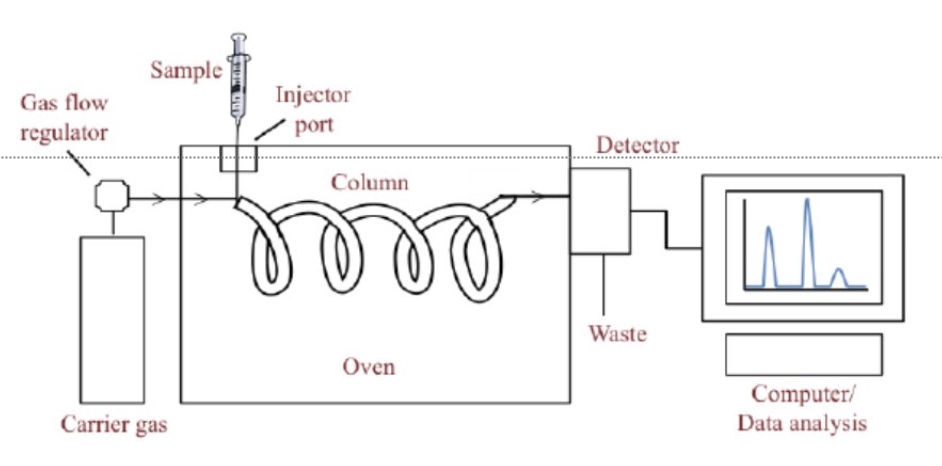
describe the method of gas-liquid chromatography

what is produced from gas-liquid chromatography?
a spectrum
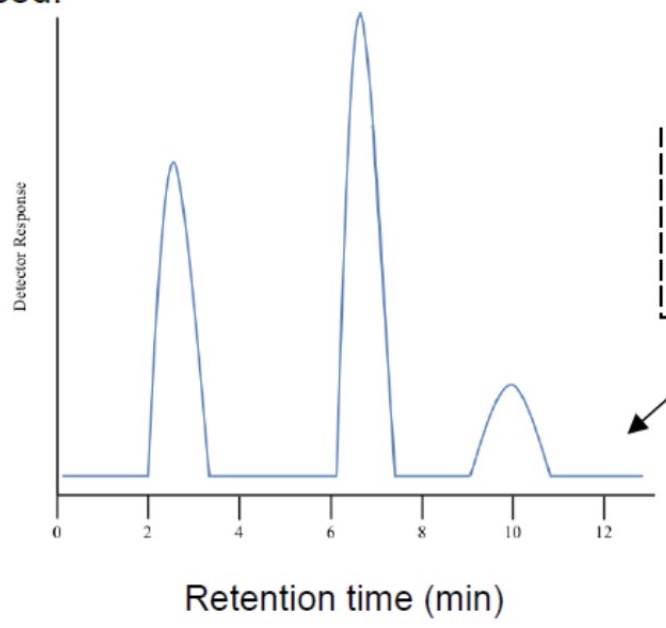
which compound is most polar + why?
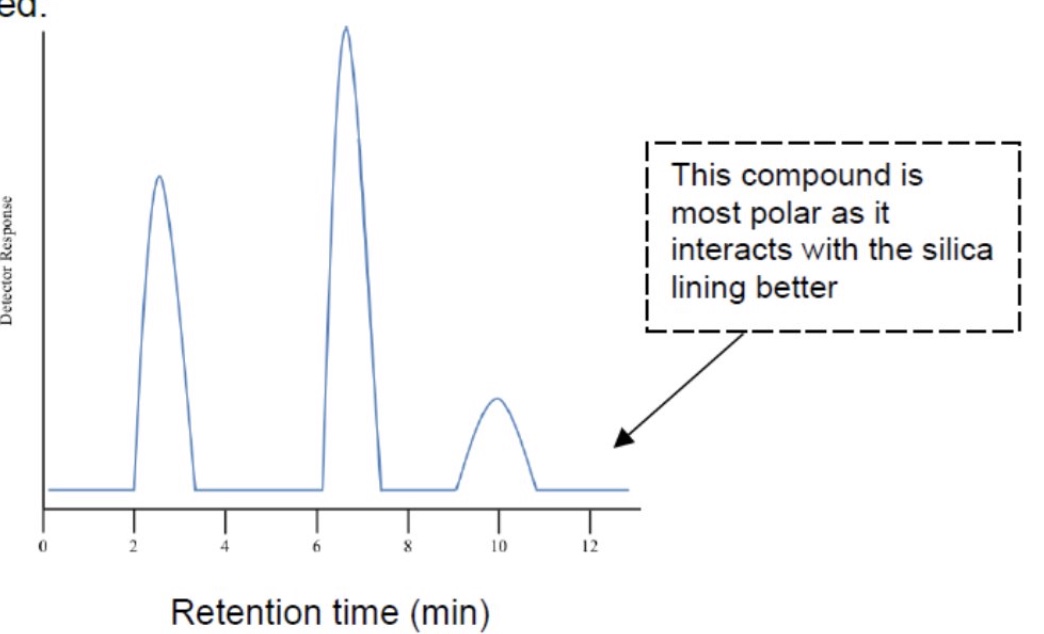
outline how you can interpret the spectrum produced from gas-liquid chromatography - ie what information can you gain from the spectrums

GLC is very ______ + is used to test for what?
very accurate
its used to test the urine of athletes for doping