The Halogens and Qualitative Analysis
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Trends in Group 7: Electronegativity
Electronegativity decreases:
The number of principle energy levels increase as there is a weaker attraction between the nucleus and the outer electron
Trends in Group 7: Boiling Point
Boiling Point increases:
The size of the atom increases so there are more electrons, this means there are more London forces between the molecules that need more energy to be broken
Fluorine at room temperature
Pale yellow gas
Chlorine at room temperature
Pale green gas
Bromine at room temperature
Dark red/orange liquid
Iodine at room temperature
Dark purple/black solid
Oxidising agent
Electron acceptor
Why does the ability of a halogen to act as an oxidising agent decrease as you go down the group?
Atomic radius increases
Shielding increases
Halogen atom gains an electron less easily as there is a weaker attraction from the nucleus
Halogen displacement
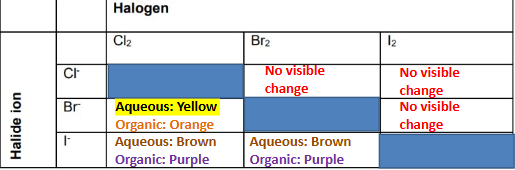
Simple ionic equations for halogen displacement
2Br- + Cl2 → 2Cl- + Br2
2I- + Cl2 → 2Cl- + I2
2I- + Br2 → 2Br- + I2
Reagents and Conditions to distinguish between halides
Reagents - Silver Nitrate
Conditions - Must be acidified
Reaction - precipitate
Aqueous ions reacted with silver halide
AgF - No Visible Change
AgCl - White precipitate
AgBr - Cream precipitate
AgI - Yellow precipitate
Why do we use nitric acid to to remove the hydroxide and carbonate ions?
They will interfere with the test
Overall and Ionic equation for nitric acid and carbonate
2HNO3 + Na2CO3 → 2NaNO3 + CO2 + H2O
2H+ + CO32- → H2O + CO2
How to distinguish between AgCl and AgBr
Add diluted Ammonia, AgCl dissolves and AgBr does not
How to distinguish between AgBr and AgI
Add concentrated ammonia, AgBr dissolves and AgI does not
Disproportionation
Oxidation and Reduction of the same element
What colour is present when chlorine is added to a universal indicator paper?
The paper first turns red, the red colour then disappears and the universal indicator paper turns white
Reaction of chlorine with cold dilute sodium hydroxide
Cl2 + 2NaOH → H20 + NaCl + NaClO
Importance: NaClO is an active ingredient in household bleach
Conditions: Dilute sodium hydroxide
Inorganic test for ions (CaSH)
Carbonate
Sulfate
Halide
Benefit of using chlorine in water treatment
Kills bacteria
Disadvantage of treating water with chlorine
Carcinogenic compounds are produced
Toxic
Carbonate Test
React Sodium Carbonate with Nitric Acid
Ionic Equations: 2H+ + CO32- → H2O + CO2
Observation: Effervescence
Sulfate Test
React Sodium Sulfate with Barium Nitrate or Barium Chloride
Ionic Equation: Ba2+(aq) + SO42-(aq) → BaSO4
Observation: White precipitate
Test for Cations
Ammonium is heated with aqueous sodium hydroxide to produce ammonia gas
Ionic equation: NH4+(aq) + OH-(aq) → NH3(g) + H20
Observation: Turns moist litmus paper blue
How can a student modify their experiment when testing for haloalkanes
Heat test tubes in a water bath
Why can chlorine extract bromide ions but iodine cannot?
Chlorine is more reactive than iodine