3.1.11.1 electrode potentials + cells
1/108
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
109 Terms
deifne an oxidising agent + reducing agent

outline what an half-cell is
when a piece of metal is dipped into a solution of its metal ions, an equilibrium is set up that represents a half equation
this is known as a half cell
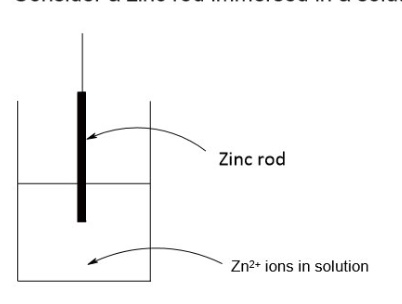
for example, describe what happens for a zinc half cell (a zinc rod immersed in a solution containing Zn²⁺ ions (eg a solution ZnSO₄)
give the half equations for this half cell
the Zn atoms on the rod can deposit two electrons on the rod + move into solution as Zn²⁺ ions
Zn → Zn²⁺ + 2e⁻
alternatively, the Zn²⁺ ions in solution could accept two electrons from the rod + move onto the rod to become Zn atoms
Zn²⁺ + 2e⁻ → Zn
this can be written as an equilibrium reaction
Zn²⁺ + 2e⁻ ⇌ Zn
if the equilibrium lies to the left hand side, what does this mean?
that there are more electrons on the surface of the rod
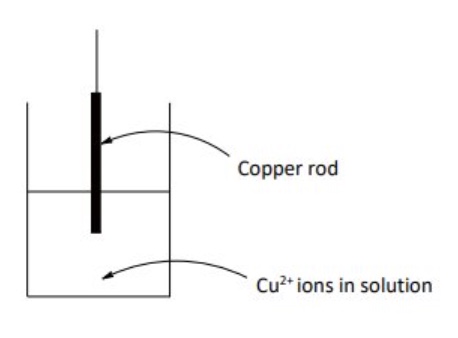
for example, describe what happens for a copper half cell (a copper rod immersed in a solution containing Cu²⁺ ions (eg a solution Zn(NO₃)₂)
give the half equation for this half cell
this can also be written as an equilibrium reaction
Cu²⁺ + 2e⁻ ⇌ Cu
in this half-cell the equilibrium could lie further to the right hand side so fewer electrons would be deposited on the rod
what is the electrode potential?
what the potential difference set up between the rod + solution is known as
what does measuring the electrode potential tell us?
how readily electrons are released by the metal so how good a reducing agent the metal is
outline what will happen if two different electrodes are connected
the potential difference between the two electrodes will cause a current to flow between them
an electromotive force (EMF) is established + the system can generate electrical energy
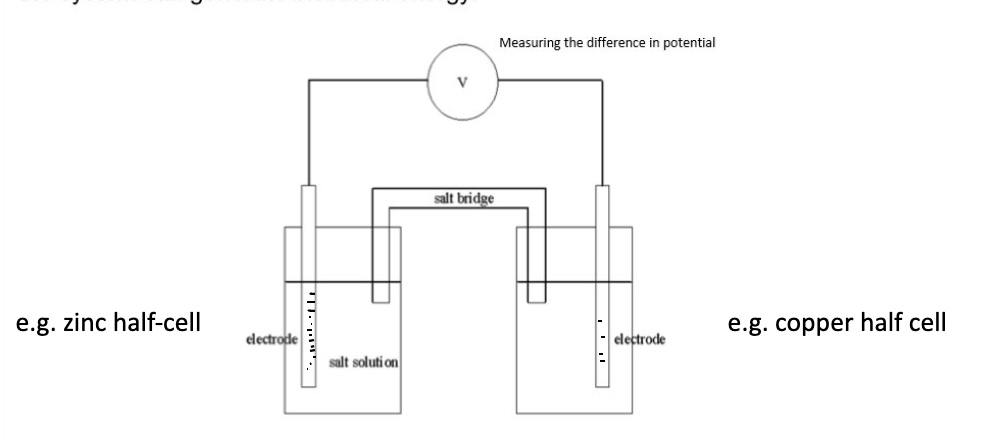
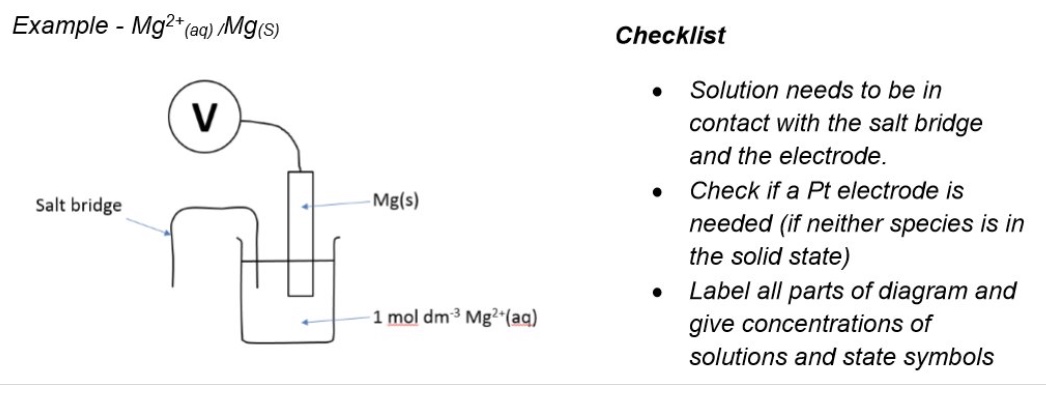
draw a representation of a cell + outline how one is created
each half-cell is placed in a separate beaker
the two beakers are then connected by a salt bridge + a voltmeter
(negative electrode is always on left side of drawing)
give 4 features of a cell
voltmeter
wire
electrodes
salt bridge
what is the purpose of the voltmeter?
measures the potential pushing power of electrons through the circuit but keeps the current at zero
what is the purpose of the wire
allows the movement of electrons
what are the electrodes?
these are where the half equations are taking place
and are so referred to as the half cells
what is the salt bridge?
its usually a filter paper soaked in a solution of KNO₃
what is the purpose of the salt bridge?
to complete the circuit + allows the movement of ions
why is KNO₃ a suitable solution?
it does not react with any of the ions in solution so it doesnt interfere with the redox reaction
and its ions can move
why is KCl not used as a salt bridge solution for a cell that contains Ag⁺ ions?
it contains Cl⁻ ions which would react with Ag⁺ to form a white ppt
there are other types of half cells, outline these half cells
there is no solid metal involved in the half-equation
for these half cells, a metal electrode is required + usually platinum is used as it is so unreactive (an inert electrode) + conducts electricity
outline the 3 general types of electrode

what is the effect of surface area of the electrode on the EMF?
the surface area of the electrode will not affect the EMF of the cell
how can a typical electrochemical cell be made?
by combining a Zn/Zn²⁺ electrode and a Cu/Cu²⁺ electrode

outline what the positive + negative electrodes are
the positive electrode is the one which most favours reduction (to the right)
the negative electrode is the one which most favours oxidation (to the left)
in terms of example of the copper, zinc electrochemical cell, state which is the positive + negative electrode, which way the electrons flow + give half equations
copper electrode is positive + zinc electrode is negative
electrons flow from the zinc electrode to the copper electrode
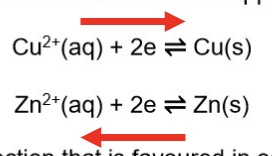
it is not possible to measure…. it is only possible to measure the…
the absolute potential of a half electrode on its own
the potential difference between two electrodes
how can we measure the potential difference between two electrodes?
the electrode has to be connected to another half-cell of known potential + the potential difference between the two half cells is measured
what will measuring the potential difference help us workout?
which electrode will be positive or negative
+ therefore its tendency to release electrons
by convention, what can we assign to each electrode + how?
can assign a relative potential to each electrode
by linking it to a standard electrode
what is the primary standard used?
the Standard Hydrogen Electrode (SHE)
what potential is the SHE assigned + why?
its assigned the potential of 0 volts, by definition, as a reference
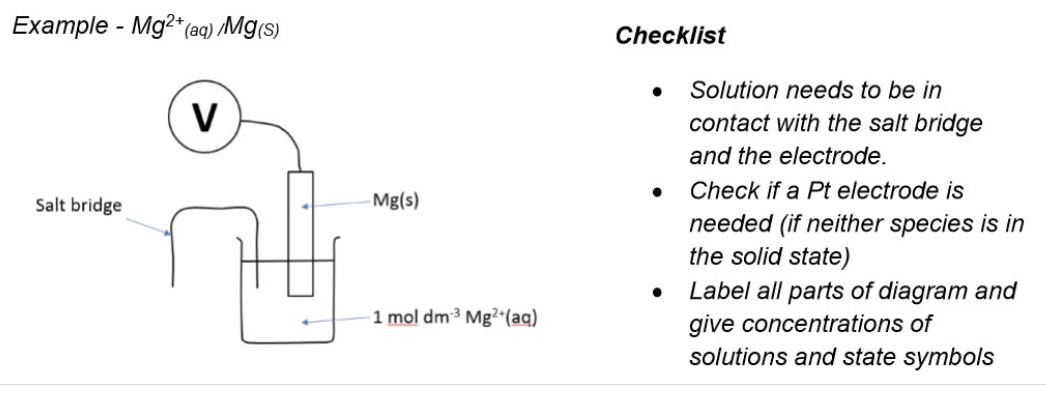
draw a labelled diagram of the SHE
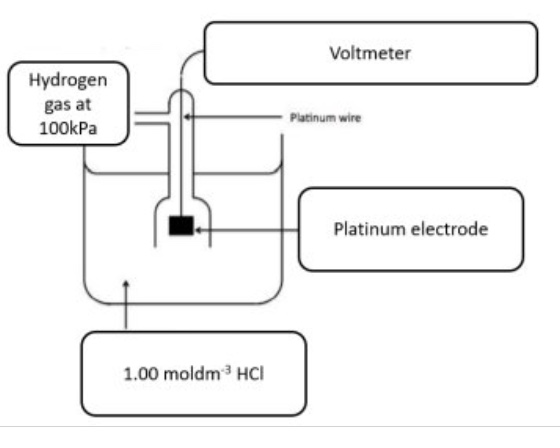
outline
a. how the SHE works
b. the half equation
c. standard conditions
d. components
a. hydrogen gas is bubbled into a solution of H+ ions and since hydrogen doesnt conduct, a platinum electrode is used


metal electrode
for the substances given, draw a labelled diagram of the electrode
redox electrode
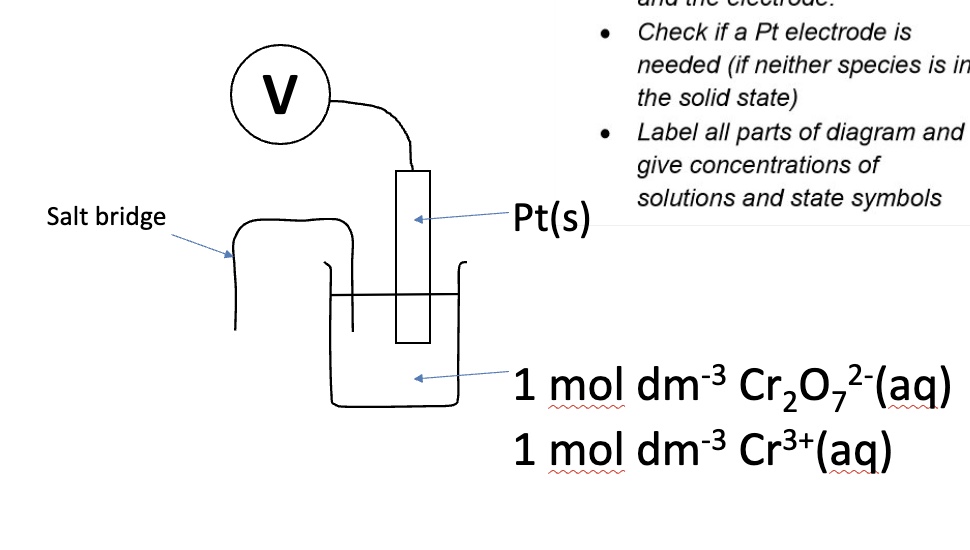
for the substances given, draw a labelled diagram of the electrode
gas electrode
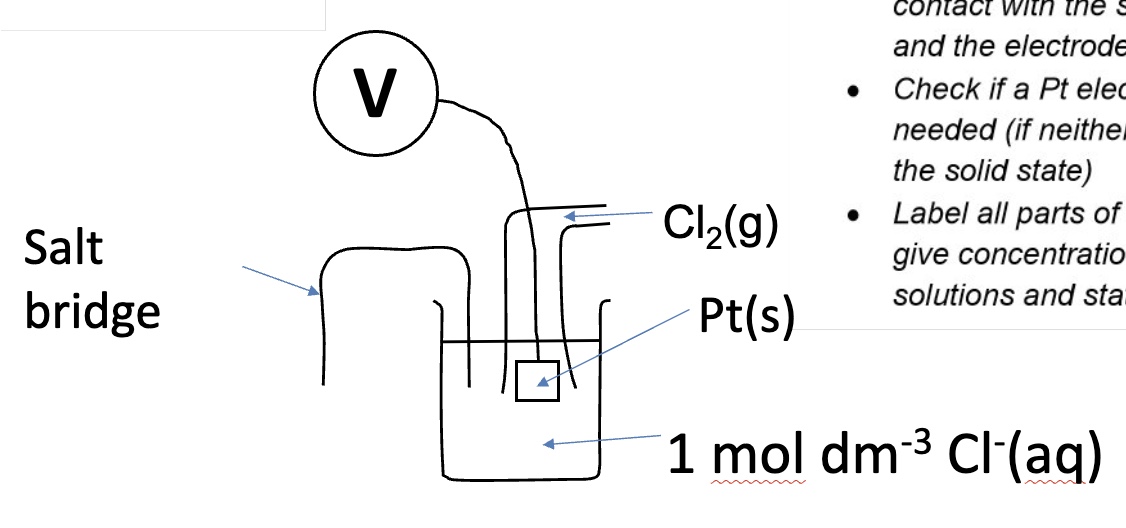
what happens once the electrode potentials have been measured using the SHE?
the electrode potentials are displayed in a table called the electrochemical series
what is the electrochemical series?
a list of electrode potentials for different half cells in order of decreasing (or increasing) potentials
outline how redox is demonstrated in the electrochemical series
all electrochemical series display reduction reactions only
but all reactions are in equilibrium so can be reversed to become oxidation equations
which ones are the best reducing agents on the electrochemical series? outline why
these have very negative potentials which mean that they are good at giving away electrons
it is the species on the right of these equations → are the best reducing agents
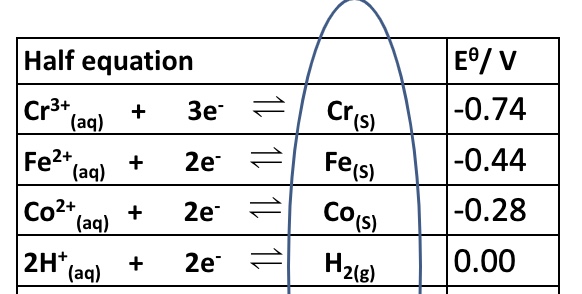
which ones are the best oxidisng agents on the electrochemical series? outline why
these have very positive potentials
which means that they are very good at attracting electrons
it is the specifies on the left of these equations - are the best oxidising agents
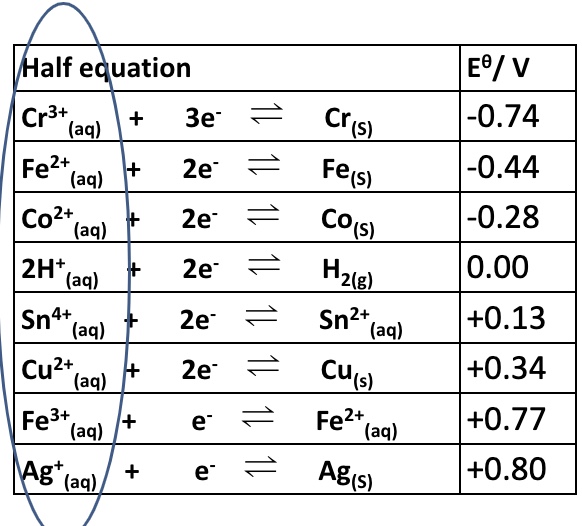
what can we use the values of electrode potentials to calculate?
to help calculate the overall electromotive force (EMF) of a cell + to predict whether reactions will take place
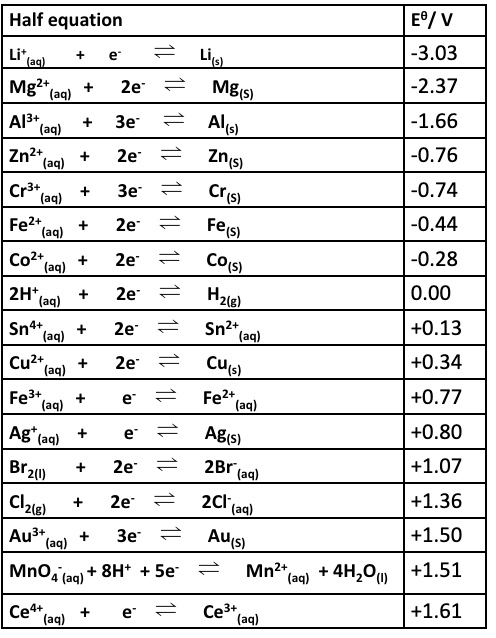
give the strongest reducing agent + explain why

what is it possible to use electrode potentials data from the electrochemical series to do?
to decide whether a redox reaction (and electron transfer) between two half-cells will work, as not all combinations will
those combinations that do react are called what?
feasible reactions
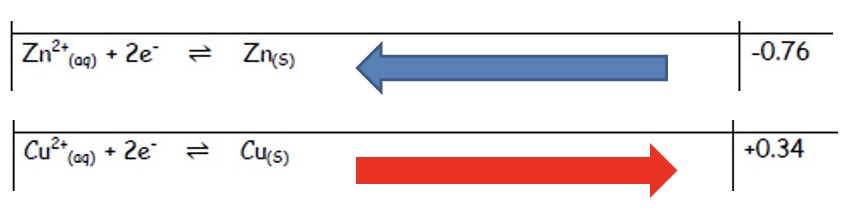
consider the reaction of Zn (s) and Cu²⁺ (aq)
explain which half equation is more likely to show oxidation
combine the half equations to give the overall equation
the Zn²⁺/Zn half equation has a lower E⍬ value (-0.76) than the Cu²⁺/Cu half equation (+0.34) so will become the oxidation half equation
this means the half equation must be reversed to show oxidation
the two new half equations + overall equation is shown in the image
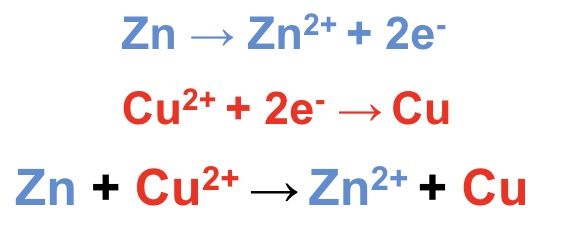
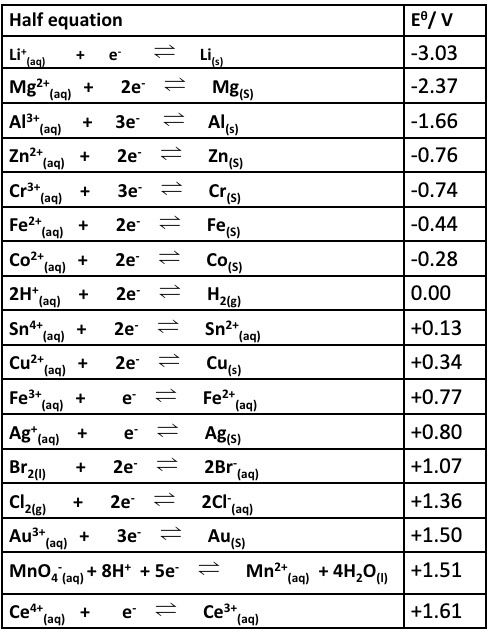
comment on the feasibility of the reaction between Zn(s) and Cu²⁺(aq)

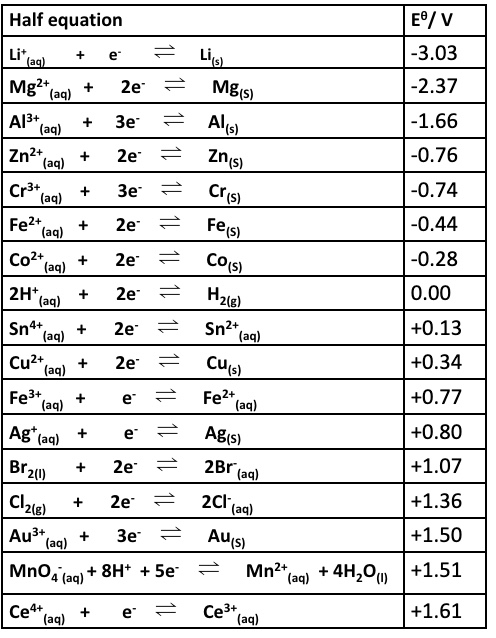
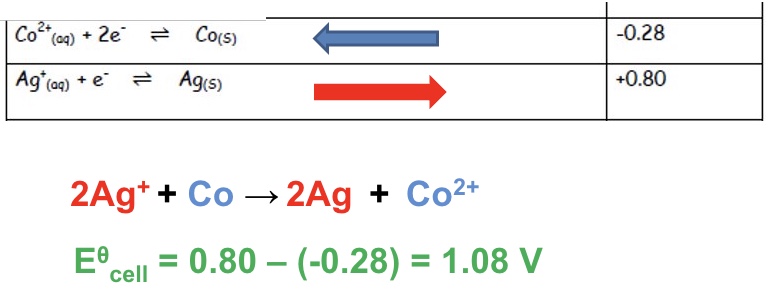
explain why the reaction below are feasible + give an overall equation:
Co and Ag⁺

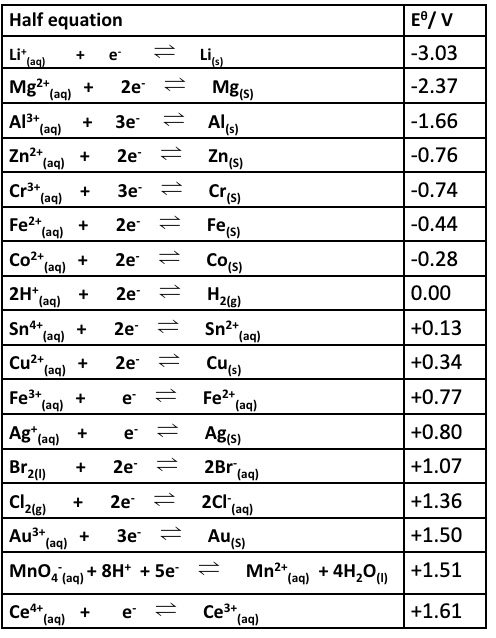
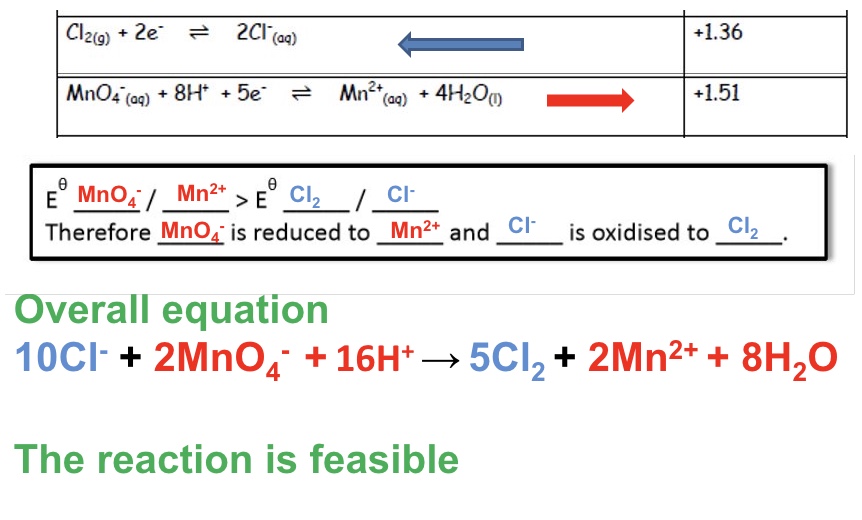
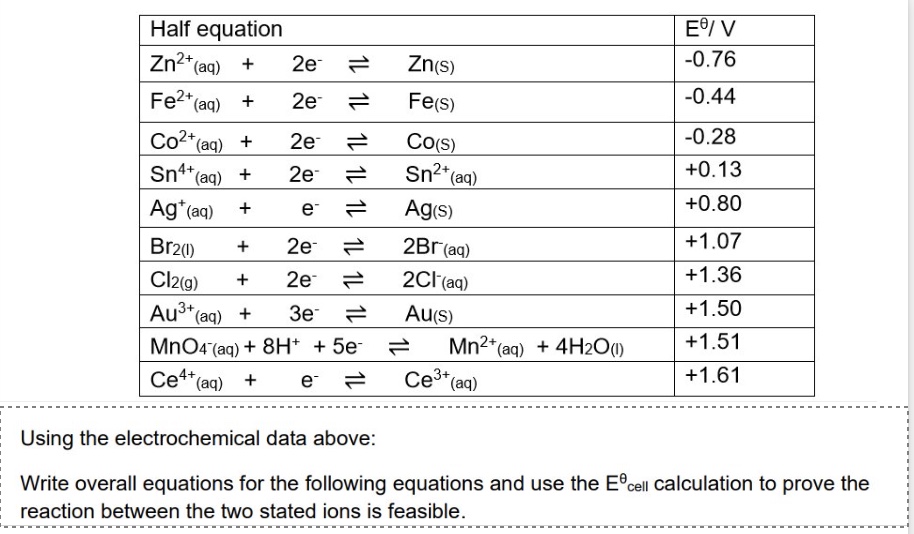
explain why the reaction below are feasible + give an overall equation:
Cl⁻ and MnO₄⁻
what is the final oxidation product of the reaction between Ag⁺ and Fe(s)?
in this question, check + see if the product of oxidation can be further oxidised by the oxidising agent

what is the final oxidation product of the reaction between Cu²⁺ and Fe(s)?

when will a reaction not be feasible?
when you construct the overall redox equation, if the suggested species are not reactants (ie on the same side of the equation)
eg Mn²⁺ and Zn²⁺, explain why they do not react together?

summary:
outline what happens at the negative and positive electrodes
negative electrode:
oxidation
equilibrium lies left
positive electrode:
reduction
equilibrium lies right
what is the E⍬ cell?
also known as the e.m.f (electromotive force)
its the potential difference across the two electrodes
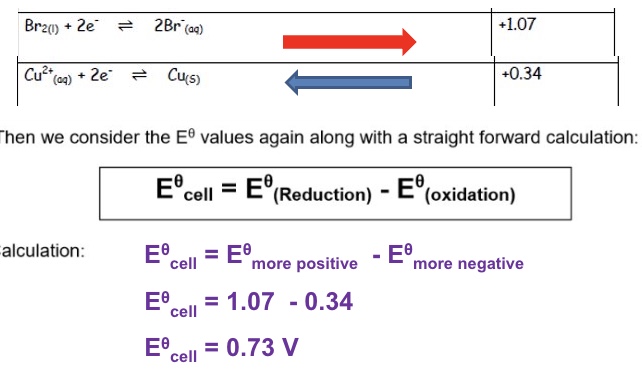
outline how to calculate emf (E⍬ cell)
first part is to choose two half equations + decide what the overall (feasible) redox reaction is when they combine
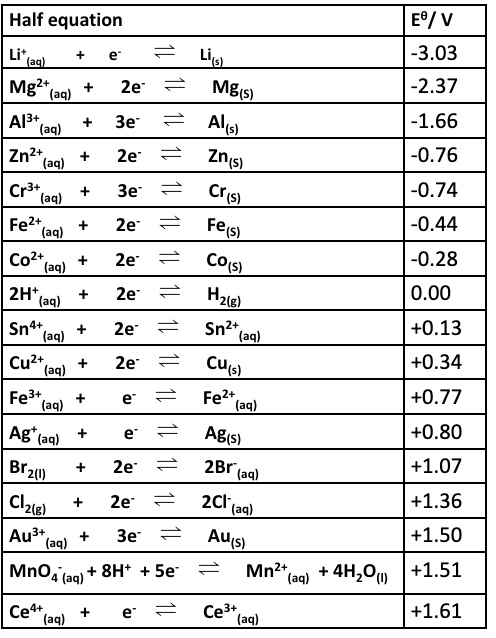
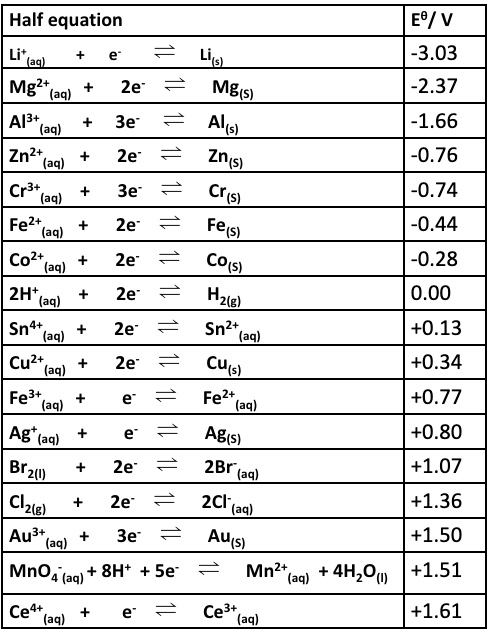
then we consider the E⍬ values long with a straight forward calculation:

eg give the overall equation for the reaction between Br₂ and Cu and calculate the emf (E⍬ cell)
Br₂ + Cu → 2Br⁻ + Cu²⁺
how can we use the value of emf to predict the feasibility of a reaction?
a positive value means the reaction is feasible
a negative value means the reaction is not feasible
the more positive a value, the more feasible a reaction
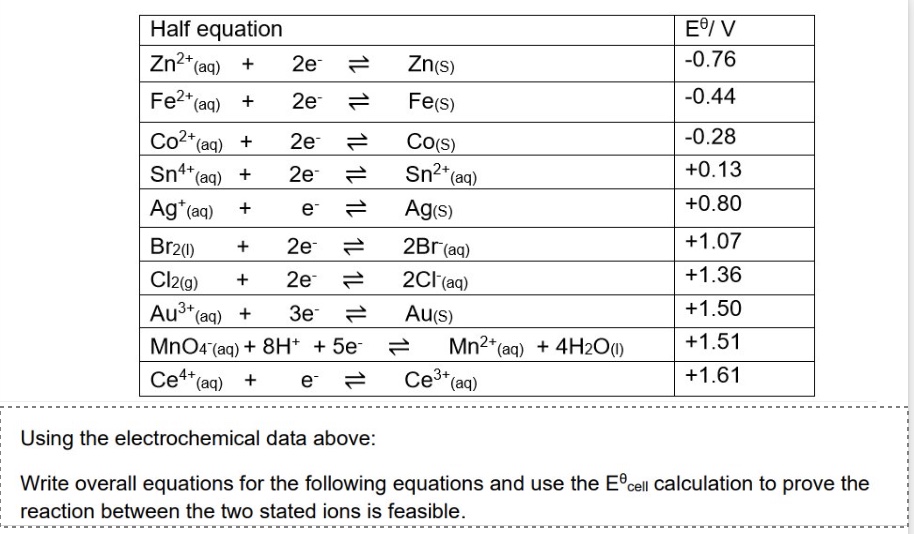
for reaction between Co and Ag⁺
for reaction between Cl- and MnO₄⁻

two different explanations (red and blue)

what is the conventional cell diagram?
a pictorial representation of the two electrodes that make up an electrochemical cell
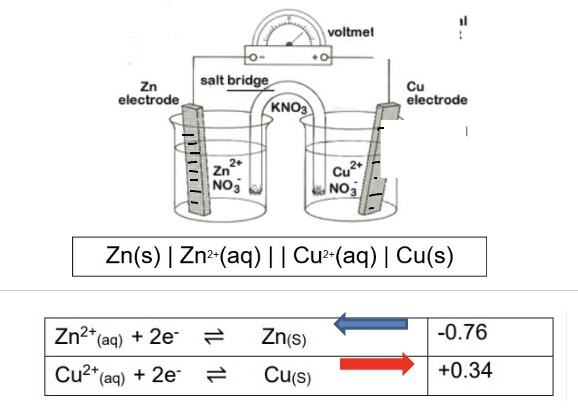
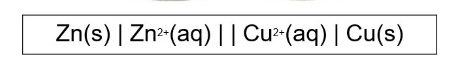
outline what the single lines represent

outline what we use commas for in conventional cell diagrams

what is the double line used to represent?
the salt bridge
explain how to write a conventional cell diagram
species on the LHS of diagram is species being oxidised (giving the electrons) → the negative electrode
species on the RHS of diagram is species being reduced (accepting the electrons) → the positive electrode
H⁺, H₂O are usually not included (unless theres a change in ox state) and e- from half equation are never included in the diagram
Pt electrode is represented as |Pt in cell diagram and they go on the outside of diagram
the order of elements is based on the direction of their half equation
what is used in a cell where there is no solid to act as the electrode? why?
a platinum electrode will be used
it provides a conducting surface for electron transfer
Pt is used as it us unreactive + can conduct electricity
by convention, on what side is the SHE always written?
the left-hand side
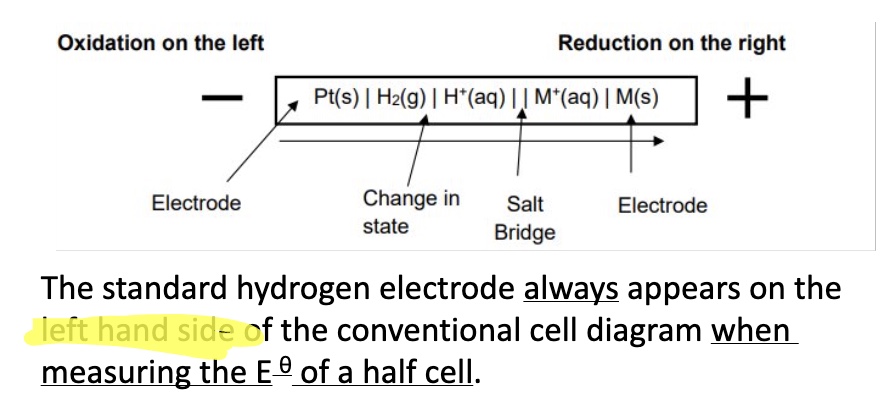
SHE always goes on the LHS when writing conventional cell diagrams but what else is different as well?
the oxidation is still written on the left and reduction on the right even if the oxidation is not actually happening - eg shown in image

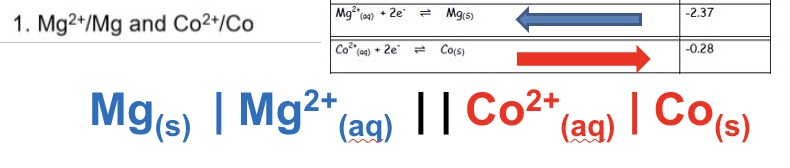
draw the conventional cell diagram for the following cell:
Mg²⁺/Mg and Co²⁺/Co

draw the conventional cell diagram for the following cell:
Fe²⁺/Fe and MnO₄⁻/Mn²⁺
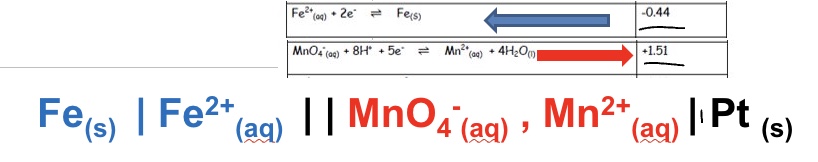

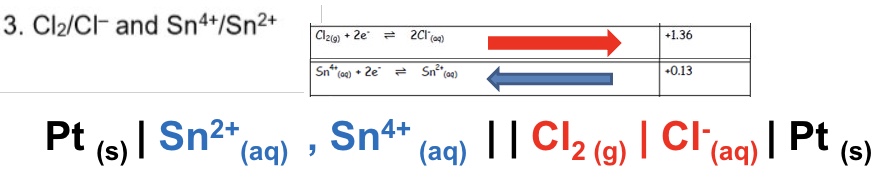
draw the conventional cell diagram for the following cell:
Cl₂/Cl⁻ and Sn⁴⁺/Sn²⁺
outline what changing the concentrations of a species in a redox half equation will do?
it will change the position of the equilibrium
changing the position of equilibrium will change the E⍬ value for the half-cell as the number of electrons are either increasing or decreasing
explain how changing the position of equilibrium both ways changes the value of E⍬

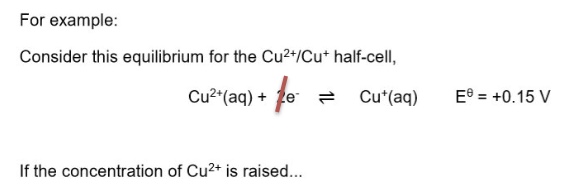




*Cu⁺ raised


*Cu⁺

outline how you would increase and decrease Ecell


what changes to concentration would make the Ecell more positive + therefore the reaction more feasible?

electrochemical cells are ____ and can be used as what?
batteries
can be used as a commercial source of electrical energy
cells can be ….. or …..
rechargeable or non-rechargeable
outline what happens in non-rechargeable cells
the chemicals are used up over time + the EMF drops
once one or more of the chemicals have been completely used up, the cell is flat + the EMF is 0 volts
what then happens to non-rechargeable cells?
these cells cannot be recharged + have to be disposed of after their single use
give an example of a non-rechargeable cell
the Zn/Cu cell
also known as ‘The Daniell Cell’
why is the Zn/Cu cell non-rechargeable?
because the Zn electrode over time starts to run out meaning that the equation cannot be reversed
also the solutions make it not practical for transport
another example of a non-rechargeable cell is Zinc/Carbon cells
outline what happens during this cell
zinc is negative electrode + acts as a case + has a paste electrolyte solution
carbon acts as positive electrode + is located as a rod down centre of canister — it behaves like the platinum in the hydrogen cell
what will happen to the emf of non-rechargeable cells over time?
it will decrease over time as the reactants are used up

the zinc is oxidised to Zn²⁺, causing the case to wear away
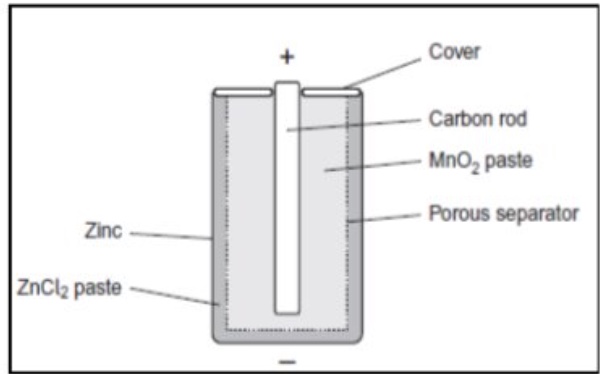
deduce one essential property of the non-reactive porous separator labelled in the diagram
to allow the movement of ions
alkaline batteries are a _____ cost cell but has a _____ life
higher cost cell
longer life
outline rechargeable cells
these are cells where the current (electrons) can be forced back the opposite direction by an applied external current (plugging them in) so that they can work again like new
what happens in the equation for rechargeable cells?
the half cell equations are reversed when it recharges
what is the main difference of rechargeable cells than non-rechargeable cells?
the electrodes are designed so that they do not decompose or wear down as quickly → these can be very small, light + portable
describe lithium ion batteries
lithium is a lightweight metal + gives for a light weight, small battery (cell)
the electrolyte phase (liquid) is a polymer which means the battery wont leak
this cell can generate about 4V so can be found in most laptops, tablets + phones
give the half equations, + overall equations for the lithium ion battery during discharge and recharge
symbol is forwards arrow
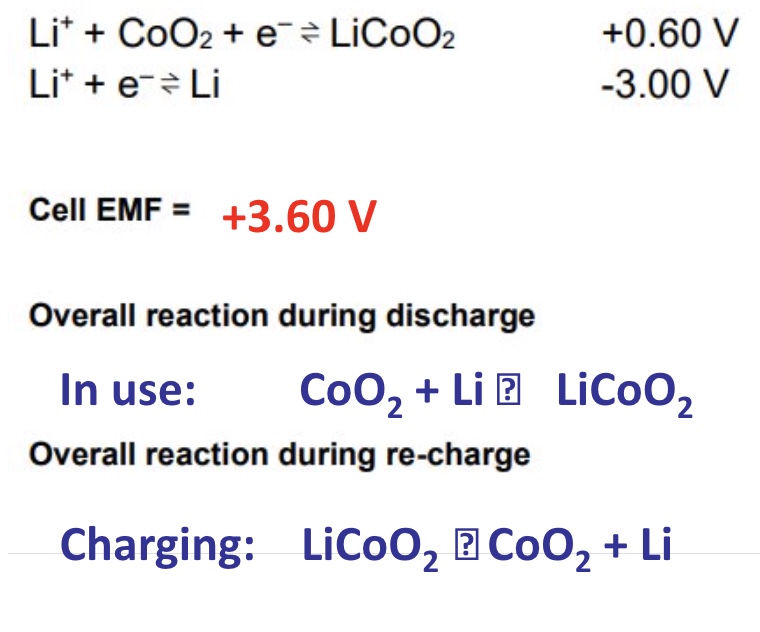
describe fuel cells
these are somewhat different to other cells
they have a continuous supply of the chemicals into the cell + so neither run out of chemicals nor need re-charging
but they do need to have a constant supply of the required chemicals
what is the most common fuel cell?
the hydrogen-oxygen fuel cell
outline the hydrogen oxygen fuel cell?
this is the alternative to actually burning hydrogen as a fuel
it consists of two platinum electrodes separated by a special polymer electrolyte which will allow ions to pass through it
in what conditions can the hydrogen-oxygen fuel cell be run in?
in alkaline or acidic conditions but the overall equation + overal emf is the same
give the two half equations + E⍬ for the hydrogen-oxygen fuel cell in acidic conditions

give the two half equations + E⍬ for the hydrogen-oxygen fuel cell in alkaline conditions
