3.2.4 - properties of period 3 elements and their oxides
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
order of elements in p3
Na, Mg, Al, Si, P and S
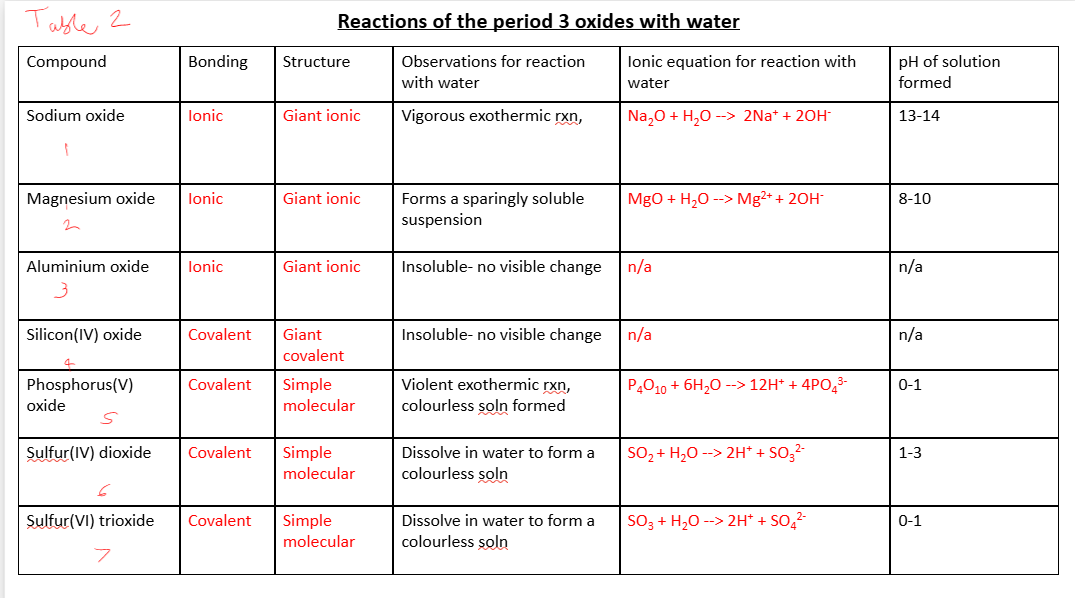
how does Na react with cold water
what does it produce
eq. for this
v. exothermic rxn
melts to form a ball
fizzes
moves on surface of water
sodium hydroxide and hydrogen
2 Na (s) + 2 H2O (l) —→ 2 NaOH (aq) + H2 (g)
observations when Mg reacts with cold water
what does this produce
observations when reacting with steam
what does this produce?
what color is this product solid
eqs. for rxns with steam + cold water
slow release of bubbles on surface of Mg
magnesium hydroxide
Mg glows bright white
magnesium oxide
white
steam - Mg (s) + H2O (g) —→ MgO (s) + H2 (g)
cold water - Mg(s) + 2H2O(l) —→ Mg(OH)2(aq) + H2(g)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
what do all the elements react with oxygen to form
oxides
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
what color flame does Na burn with
it produces a w_____solid
eq. for this (learn formula of product)
yellow
white
4 Na (s) + O2 (g)—→ 2 Na2O (s)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
observations when Mg reacts with water - what color light is there?
what color solid is produced
eq. for this
white light
white solid
2Mg (s) + O2 (g) —→ 2MgO (s)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
observation when Al reacts with water
what color solid is produced
eq. for this
white SPARKS
white
4Al + 3O2 (g) —→ 2Al2O3 (s)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
observations when Si reacts with water
what color solid is produced
eq. for this
orange sparks
white solid
Si + O2 (g) —→ SiO2 (s)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
what color flame does P burn with
this gives a w______ s______
eq. for this
white
white smoke
P4 + 5O2 (g) —→ P4O10 (s)
Trends in the reactions of the elements Na, Mg, Al, Si, P and S with oxygen
what color flame does S burn with
it forms an a_______ c__________ g__
eq. for producing SO2
eq. for producing SO3
blue
acidic choking gas
S + O2 (g) —→ SO2 (g)
S + 1.5O2 (g) —→ SO3 (g)
what are the ionic metal oxides of period 3
why are they ionic
high or low mp?
why
Na2O, MgO, Al2O3
large EN diff. between metal and O
high
giant ionic lattices
lots of ionic bonds —→ electrostatic forces of attraction between oppoisitely charged ions
these are very strong + take a lot of energy to break
why is there increasing mps from Na to Al
increased charge on cation (The strength of an ionic bond is directly proportional to the charges on the ions. Na2O contains Na+ ions, MgO contains Mg2+ ions, and Al2O3 contains Al3+ ions. The higher the charge, the stronger the electrostatic attraction to the ion.)
also, across the period, the metal ions become smaller (Na+ > Mg2+ > Al3+). Smaller ions can get closer together, resulting in stronger attractions (greater lattice enthalpy of dissociation), making it harder to separate the ions during melting.
so stronger ionic bonds
why is the melting point of Al2O3 is sometimes lower than that of MgO
the high-charge Al3+ ion distorts the O2- electron cloud, giving it significant covalent character.
what is the macromolecular metal oxide in period 3
is its mp low or high
why
SiO2
very high
has many strong covalent bonds between atoms.
High energy needed to break these
what are the simple molecular metal oxides in period 3
lower or higher mps
why
why are they covalent
why does P4O10 have a higher mp than SO2
P4O10 and SO2
lower
weak intermolecular forces between molecules (van der waals + permanent dipoles)
small electronegativity difference between the non-metal and O atoms
it is a bigger molecule, so more e-s, so more vdWs, so higher mp
how to prove experimentally all these compounds contain ions
melt the solids and show they conduct electricity
The reactions of the oxides of the elements Na —→ S with water (check sheet from booklet)
what do metal ionic oxides react with water to form
are these acidic or alklaine
reaction of Na2O
what pH is the solution formed
hydroxides
alkaline
Na2O (s) + H2O (l) —→ 2NaOH (aq)
13
are ionic oxides basic or acidic?
summary of rxns of period 3 oxides
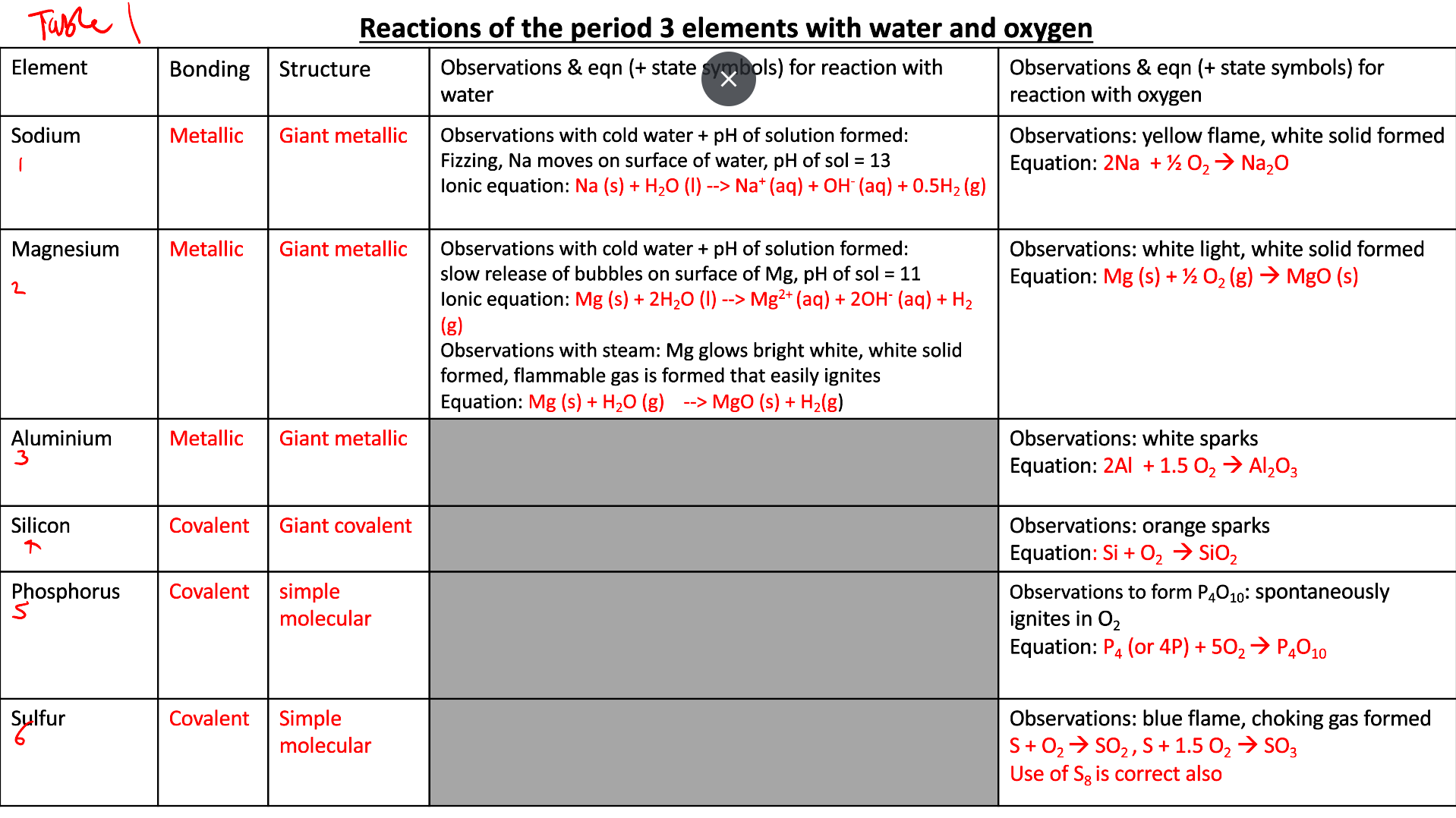
summary of reactions of period 3 oxides with water