Phy Sci Test
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Chemical Change
A process that results in the transformation of one substance into another.
Physical Change
A change that affects one or more physical properties of a substance without altering its chemical composition.
Chemical Property
A characteristic of a substance that can only be observed during a chemical reaction.
Physical Property
A characteristic of a substance that can be observed or measured without changing the substance's identity.
Pure Substance
A material made of only one type of particle.
Homogeneous Mixture
A mixture that has a uniform composition throughout.
Heterogeneous Mixture
A mixture in which the composition is not uniform throughout.
Density Formula
Density (D) is defined as mass (m) divided by volume (V): D = m/V.
Standard Temperature and Pressure (STP)
Conditions under which the volume of a gas is commonly measured: 0°C (273 K) and 1 atm (101.3 kPa).
Specific Heat Formula
The amount of heat required to change the temperature of a unit mass of a substance by one degree Celsius: Q = m * c * ΔT.
Endothermic Process
A process that absorbs heat from the surroundings.
Exothermic Process
A process that releases heat to the surroundings.
Kinetic Energy
The energy an object possesses due to its motion, related to temperature.
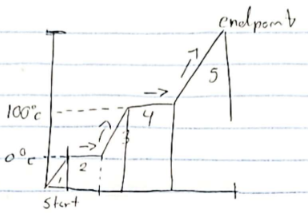
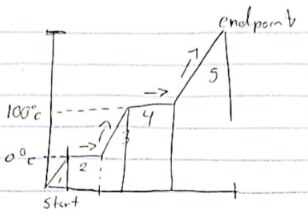
Hvap
stage 4
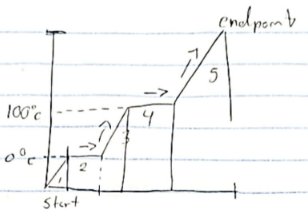
Hfus
stage 2
Temperature Conversion to Kelvin
To convert Celsius to Kelvin, add 273.
Units of Pressure
760 mmHg = 1 atm = 101.3 kpa
Calculating Total Heat
The sum of all heat changes during phase changes and temperature changes.
Heat Transfer Equation
Q = m * C * ΔT, where m is mass, C is specific heat, and ΔT is the change in temperature.
Q = m * C * ΔT
stage 1, 3, 5
Endothermic
absorbs heat Ex. Melting, vapor
Exothermic
release heat Ex. Freezing, condenation
Temp
the measure of average kinetic energy
CH2O
4.18 “g/”c
Cgas
2.02
Csolid
2.06
Hfus
334 J/g
Hvap
2260 J/g
Examples of Chemical Properties
Reactivity with acid, flammability, oxidation states, and pH
EX. Physical Property
Color, smell, freezing point, boiling point, and density
chemical change
rusting of iron, where iron reacts with oxygen to form iron oxide.
What is a physical change?
Melting, freezing, boiling, dissolving, and chopping.

what is the first picture?
Compound

2nd picture is
mixture

3rd picture is
element

4 is
mixture

5 is
element
what is the gas law equation
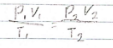
Solution
A homogeneous mixture composed of two or more substances, where one substance (the solute) is dissolved in another (the solvent), such as saltwater.
gas law is it Kelvin or Celsius
kelvin
specific Heat is it kelvin or Celsius
Celsius
compound
H2O
element
Cu
pure substance
Homogeneous
mixture
Homo and hetero