Chap 21B - Hydroxy
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
Describe reaction of alcohol with Na + general reaction
Alcohols react rapidly with sodium to form alkoxides (RO–) and H2 gas
Reagents and conditions: Na
ROH + Na -> RO-Na+ + 1/2H2
Describe distinguishing test for organic compounds with –OH group (alcohol, phenol or carboxylic acid) + observation
Add Na metal to the unknown organic compound
Observation:
If the compound contains an –OH group, effervescence is observed, H2 gas evolved extinguishes lighted splint with a ‘pop’ sound
If –OH group is absent from the compound, no effervescence is observed
Describe oxidation of primary alcohol to obtain aldehyde
Use primary alcohol, K2Cr2O7(aq), H2SO4(aq) and heat with distillation
NOTE: Using acidified KMnO4, heat under reflux will oxidise the primary alcohol to a carboxylic acid as it is a much stronger oxidising agent (no aldehyde)
NOTE: NOT heat under reflux
Milder reaction condition is required
Under the conditions of reflux -> further oxidation of the aldehyde to carboxylic acid
Aldehydes have lower bp than corresponding alcohol -> the distillation process would allow the removal of the product once it is formed, shifting POE right
Describe oxidation of primary alcohol to obtain carboxylic acid + general reaction
To obtain carboxylic acid: Use primary alcohol, K2Cr2O7(aq), H2SO4(aq) and heat under reflux
RCH2OH + [O] → RCHO + H2O
RCHO + [O] → RCO2H
Overall: RCH2OH + 2[O] → RCO2H + H2O
![<ul><li><p><span style="background-color: transparent;"><span>To obtain carboxylic acid: Use primary alcohol, K2Cr2O7(aq), H2SO4(aq) and heat under reflux</span></span></p></li></ul><p></p><ul><li><p><span style="background-color: transparent;"><span>RCH2OH + [O] → RCHO + H2O</span></span></p></li><li><p><span style="background-color: transparent;"><span>RCHO + [O] → RCO2H</span></span></p></li><li><p><span style="background-color: transparent;"><span>Overall: RCH2OH + 2[O] → RCO2H + H2O</span></span></p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/03f630da-e49f-4645-9758-06d2a21f2233.png)
Describe reflux
Process of continued heating with condensation of the vapour back into the reaction mixture
Most organic reactants are of high volatility and most organic reactions are slow and require heating
The reflux will prevent volatile components of the reaction mixture from boiling off during the prolonged heating -> maximum yield of the desired product
Draw distillation setup
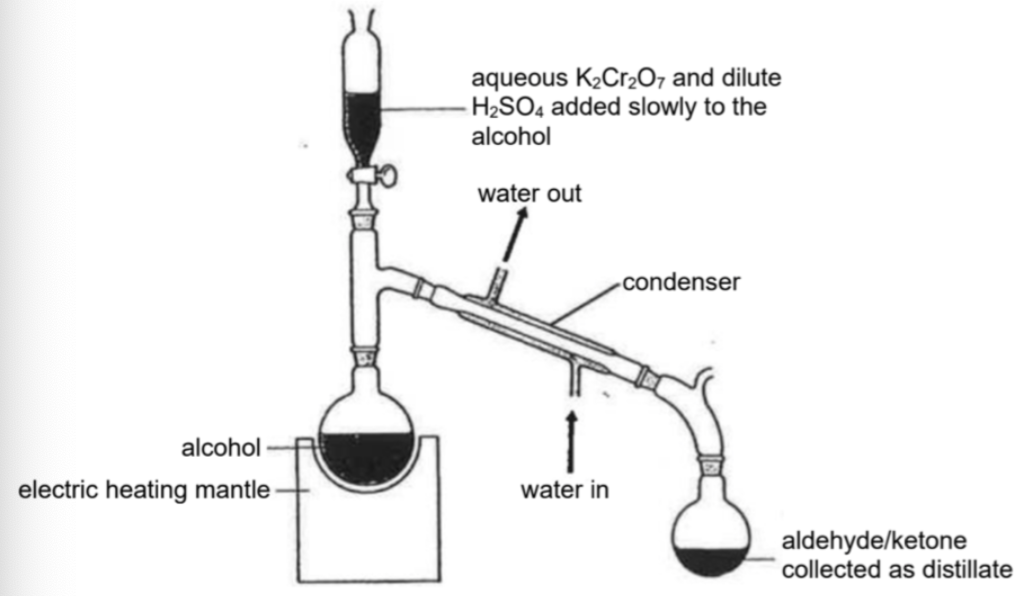
Describe oxidation of secondary alcohol
Reagents and conditions: K2Cr2O7(aq), H2SO4(aq), heat OR acidified KMnO4 and heat
RCH(OH)R’ + [O] → RCOR’ + H2O
Betanone (ketone)
![<ul><li><p><span style="background-color: transparent;"><span>Reagents and conditions: K2Cr2O7(aq), H2SO4(aq), heat OR acidified KMnO4 and heat</span></span></p></li><li><p><span style="background-color: transparent;"><span>RCH(OH)R’ + [O] → RCOR’ + H2O</span></span></p></li><li><p><span style="background-color: transparent;"><span>Betanone (ketone) </span></span></p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/34267c21-72f7-430a-9ec8-3f8fc48f3b5f.png)
Describe oxidation of tertiary alcohol and phenols
Compounds where an –OH group is directly bonded to a benzene ring have no hydrogen atoms on the carbon with –OH group (alpha carbon)
CANNOT be oxidised
Describe test for primary, secondary and tertiary alcohol + observation
Heat the alcohol with K2Cr2O7(aq) and H2SO4(aq)
Observation:
If acidified K2Cr2O7 changes from orange to green, primary or secondary alcohol is present
If acidified K2Cr2O7 remains orange, tertiary alcohol is present
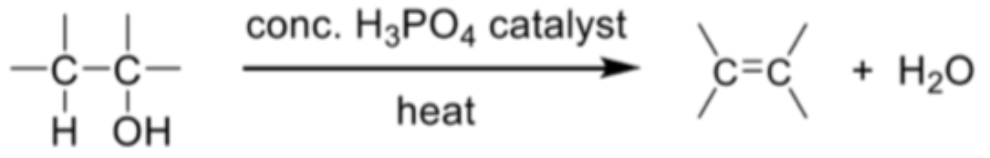
Describe dehydration of alcohol + general reaction
Elimination of water from alcohol gives the corresponding alkene
Reagents and conditions: concentrated H3PO4 catalyst, heat
Describe Saytzeff’s rule
Dehydration of secondary and tertiary alcohols containing 4 or more C atoms yields a mixture of 2 structural alkenes as products
More substituted alkene will be predominantly formed (more alkyl groups)