chem final mid term
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Ligand
ions/molecules with lone pairs that can act as lewis bases
Lewis base
an atom that has a lone pair that shares it
Lewis acid
accepts electron pair
ksp
ksp is a salt dissolving so the ksp will only be the molarity of salts ions multiplied
insoluble salt
less than 0.01 mol in 1 L
how is solubility determined
comparing ksps of salts with the same cation and anion ratio, if the mole ratios are different you can calculate the M of the salt at eq
when to use net k
net k=ksp*kf
AB(s) + C(aq)→. AC(aq) +B(aq)
←
low solubility salts
dissove better in solutions w relevant ligands
Q and ksp
Q=ksp its at equilbrium/saturatedd
Q<Ksp too few products
Q>ksp too many products, unsaturated some must precipitate so depending on how different molarity you can find how much precipitate
Delta s>0 when
S to liquid L to gas or s to gas
Dissolving solute in solid
Increasing volume of gas
Increasing temp
Increasing number of moles of gas (delta n gas>0)
∆G
the amount of energy that can be used to do work aka energy that is not lost because it is too dispersed
if it is negative it will be product favored
when is a system spontaneous
when gibbs is negative, entropy being positive or negative does not necessarily determine spontaneity
when a reaction is irreversible
How to manipulate gibbs free energy
to double or triple multiple by 2 or 3
add them together to combine
negative to turn around
Whats soluble
all compounds containing alkali metals (Li+, Na+, K+, Rb+, Cs+)
Nitrates (No3-)
Acetates (CH3COO−)
Sulfates (SO42
Microstates
-possible forms of energy dispersal, gives probability of energy being in atoms
-states that disperse energy over a larger amount of atoms are more probable
-entropy increases with number of microstates
Dispersal of atoms
-increase of volume causes increase of number of micro states in gases
-so mixing gases, mixing miscable liquids, dissolving soluble solid in liquid are spontaneous and irreversible
Trends in entropy
-larger molecules have larger entropy
-structures that are more complex have a larger entropy
-entropy of solid is lower than an entropy of liquid which is lower than gas
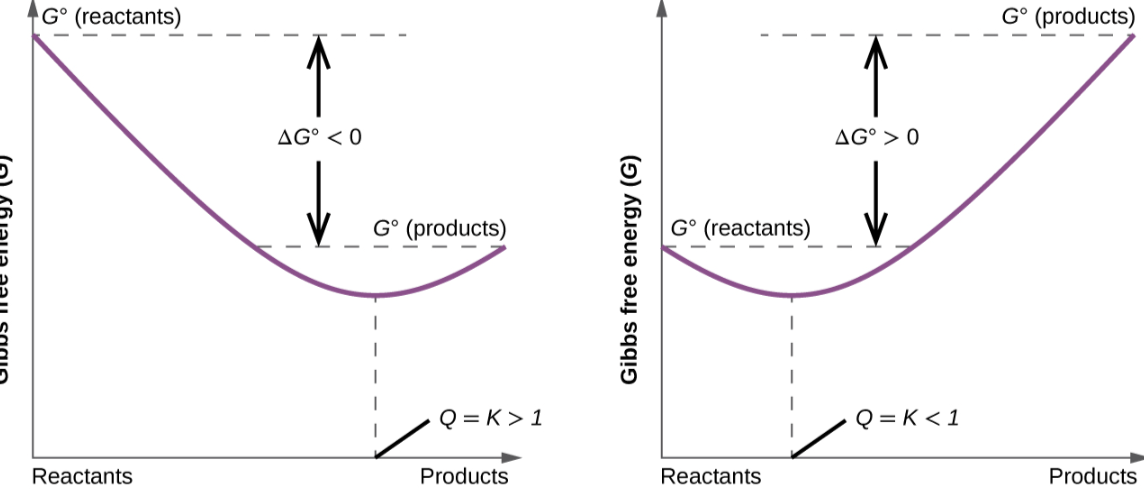
Gibbs change as reactants go to products
initially gibbs energy decreases, reaches 0 at equilibrium then increases
For product forward equilibrium is closer to products free energy and vice versa