Radt 142 Exam 2
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
Atom
tiny basic building block of all matter
Most common way to picture an atom
solar system model (Bohr’s Model)
What are atoms made of?
protons, electrons, neutrons
Protons
positive electrical charge
Neutrons
no electrical charge
Electrons
negative electric charge
Nucleus
protons, neutrons
What determines the mass of an atom and why
mass of nucleus
protons and neutrons are larger than electrons
Element
any material or substance that is composed of only one type of atom
Compound
different elements combined during chemical change
Molecule
smallest particle of a compound
Mixture
two or more substances that are mixed but not chemically joined
air
Difference between compound and molecule
Molecule: formed when 2 or more atoms join chemically (O2)
Compound: molecule that contains at least 2 different elements (CO2)
All __________ are ____________ but not all _________ are ______________.
All compounds are molecules but not all molecules are compounds
__________ can’t be broken down
element
______ possess the same characteristics of the element
Atoms
________ have the same characteristics and is the smallest particle of a compound
Molecules
Maximum number in each shell
2n²
Maximum amount of electrons N shell can hold
32
2(4²)
2(16)
32
Outermost shell (_______ ______) of an atom is referred as a _________ ______.
electron orbit
valence shell
Electron Binding Energy
energy level where electron orbits around nucleus of atom
orbital levels closer to nucleus have greater biding energy than orbital levels further from nucleus
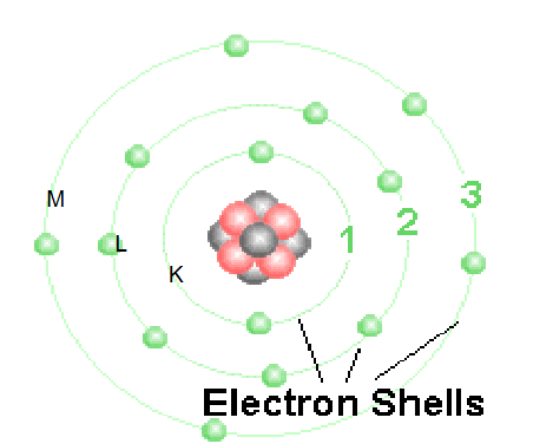
Which shell has the lowest binding energy
M
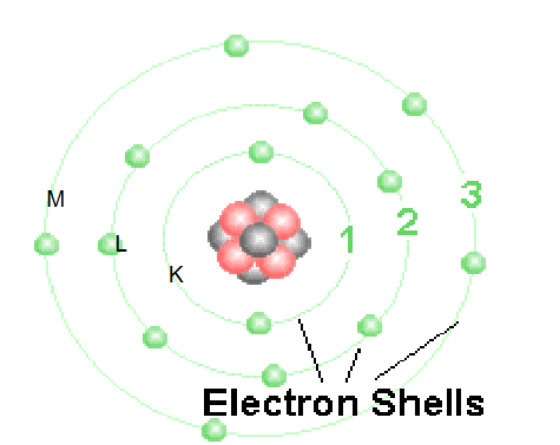
Which shell has the highest binding energy
K
What determines what the element will be
number of protons in nucleus
Atomic Mass
total number of protons and neutrons (nucleons/nucleus)
Atomic Number (z)
number of protons in nucleus determines what the element will be
Atomic mass formula
A = Z + N
atomic mass = atomic# + neutrons

X
atom’s elemental symbol

A
mass number

Z
atomic number symbol
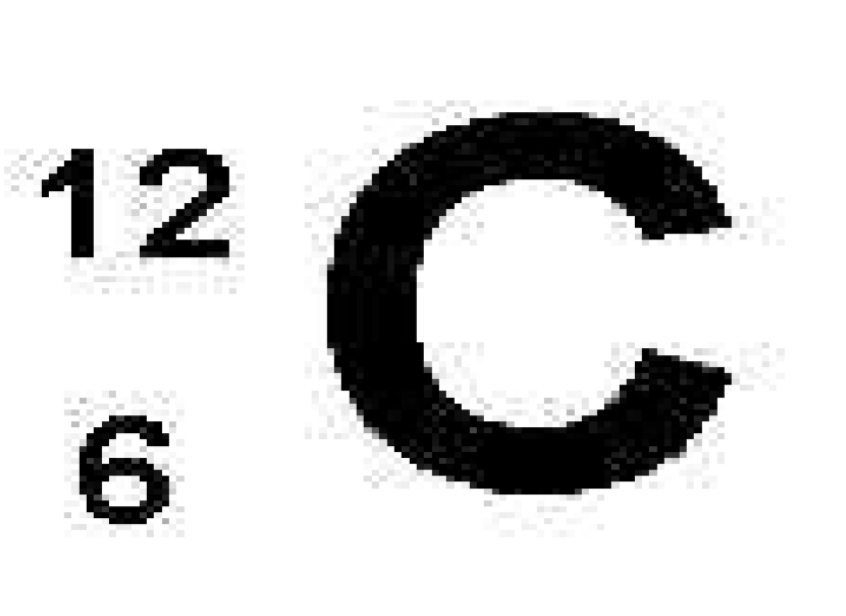
What is 12
mass number
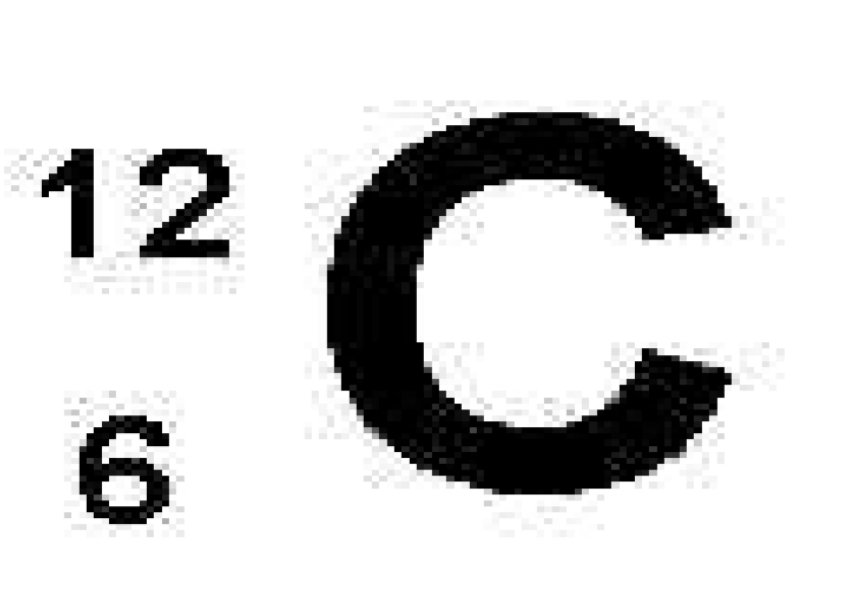
What is C
Atom’s elemental symbol
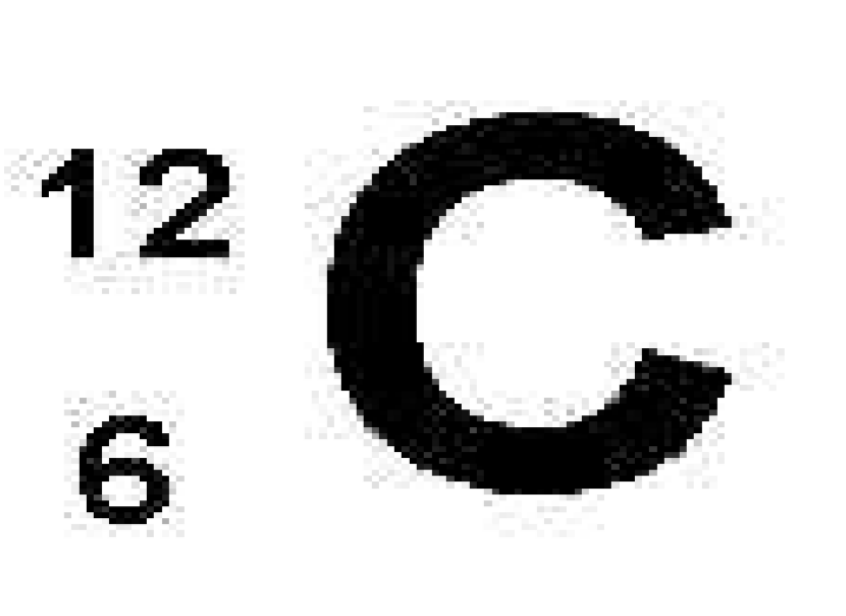
What is 6
atomic number symbol
Neutral Atom
stable
balanced protons and electrons
Ionized Atom
unbalanced protons and electrons
Ion
atom that gains or loses one or more electrons
Positive Charge Ion
electron is lost
Negative Charge Ion
electron is gained
2 Chemical bonds
Ionic
Covalent
Ionic
transfer of electron (gain or loss) between atoms
Covalent
sharing of electrons between atoms
Isotope
same number of protons
has different numbers of neutrons
Isotone
same number of neutron
diff number of protons
Isobar
same number of mass
diff atomic number (Z)
Nuclear Structure: Isotope
same z, diff A and N
Nuclear Structure: Isobar
same A, diff Z and N
Nuclear Structure: Isotone
same N, diff Z and A
Radioactivity
release of energy and matter
How does radioactivity occur
changes in nucleus of an atom
radioactive disintegration/decay
Half Life
period of time required for a quantity of radioactivity to be reduced ½ its original value
20 21
Radioactive decay results in the emission of what 3 things.
alpha particles
beta particles
gamma rays
Alpha particles
release of 2 neutrons and 2 protons
Beta particles
originate in nucleus
when emitted a neutron converts to proton
Gamma Rays
associated with alpha and beta emission
very high penetration power, health risk
Stopped by sheet of paper
Stopped by several feet of concrete or few inches of lead
Stopped by later of clothing or few millimeters of aluminum
alpha particles
gamma rays
beta particles
Smallest subdivision of a compounds that maintains the same characteristics of that compound
molecules
Smallest portion of element
atom
Two or more different atoms (elements)
compound and molecule
Two of more same atoms
molecules
What is a combination of different substances
mixture
7 electron shells
K = 2
L = 8
M = 18
N = 32
O = 50
P = 72
Q = 98