4.3 Particle model of matter
1/26
Earn XP
Description and Tags
Completed flashcard set
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Give the formula for density
density = mass / volume
ρ = m/ v
ρ - kilograms per metre cubed, kg/m3
m - kilograms, kg
v - metres cubes, m3
What can the particle model of matter be used to explain?
The different states of matter
Differences in density
Draw simple diagrams to model the differences between solids, liquids and gases
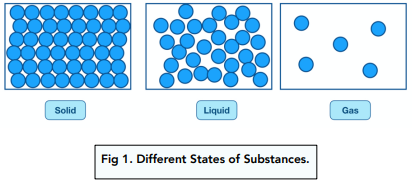
Solids
Solids have a very high density as strong forces of attraction hold the particles close together in a fixed, regular arrangement. The particles don’t have much energy so only vibrate about fixed positions.
Liquids
There are weaker forces of attraction between particles; they are close together but can move past each other and form irregular arrangements. They have more energy than particles in a solid - they move in random directions at low speeds. Liquids are less dense than solids but more dense than gases.
Gases
There are extremely weak forces of attraction between the particles; the particles have more energy than in liquids and solids. The particles are free to move and travel in random directions at high speeds. Gases have very low density.
Why are changes of state physical changes?
Changes of state are physical changes which differ from chemical changes because the material recovers its original properties if the change is reversed
No new substances are formed
It only alters the arrangement and motion of particles without changing the chemical composition of the substance
What is internal energy?
Internal energy is the total kinetic energy and potential energy of all of the particles that make up a system
Energy is stored inside a system by the particles that make up a system
What does the temperature of a substance represent?
The average kinetic energy of the particles that make up the substance
How does heating change the energy stored by a system?
Heating a system transfers energy to the particles that make up a system; they gain energy in their kinetic energy stores and move faster, increasing their internal energy.
As the kinetic energy of the particles increases, the temperature of the particles increases; this leads to an overall increase in the temperature of the system.
A temperature rise can lead to a change of state as as particles gain more kinetic energy, they can overcome the forces of attraction between particles, which can lead to a change of state.
This either:
Raises the temperature of the system
Produces a change of state
If the temperature of a system changes, what does the increase in temperature depend on?
The mass of the substance heated
The type of material (its specific heat capacity)
The energy input into the system
The following equation applies:
change in thermal energy = mass x specific heat capacity x temperature change
ΔE = m c Δθ
ΔE - joules, J
m - kilograms, kg
c - joules per kilogram per degree Celsius J/kg°C
Δθ - degrees Celsius, °C
What is the specific heat capacity of a substance?
The energy required to raise the temperature of one kilogram of a substance by one degree Celsius
What is the energy needed for a substance to change state called?
Latent heat
What happens when a change of state occurs?
The energy supplied changes the energy stored stored (internal energy) of the particles in the substance
But not the temperature of the substance
Define specific latent heat
The specific latent heat of a substance is the energy required to change the state of one kilogram of the substance without changing its temperature
State and define the two different types of specific latent heat
Specific latent heat of fusion: the energy required to change the state of one kilogram of the substance from solid to liquid (or liquid to solid) without changing its temperature
Change of state between solid and liquid
Specific latent heat of vaporisation: the energy required to change the state of one kilogram of the substance from liquid to vapour (or vapour to liquid) without changing its temperature
Change of state between liquid and vapour
Give the formula for specific latent heat
energy for a change of state = mass x specific latent heat
E = m L
E - joules, J
m - kilograms, kg
L - J/kg
Why does the temperature of a substance not change when it is changing state even when it is being heated/ cooled?
The heat is used to break or form intermolecular bonds, rather than being transferred to/from the kinetic energy of the particles of the substance
Describe the motion of particles in a gas
The particles of a gas are in constant random motion
They move at random speeds in straight lines in random directions
Particles within the same gas can move at a range of speeds (but total average speed is the same (dependent on temperature))
If you change the temperature of a gas at constant volume, what happens?
It changes the pressure exerted by the gas
If temperature is increased, the particles gain more kinetic energy so move at faster speeds
The faster moving particles will collide with the surface of their container more frequently
These collisions exert forces onto the surface
Since the volume of the container is constant and the number of collisions in a given time has increases, the force exerted onto the surface increases (and thus the pressure increases)
Describe how the motion of the particles of a gas is related to its temperature
If you increase the temperature of a gas, energy is transferred to the kinetic energy stores of its particles
The temperature of a gas is related to the average kinetic energy of its particles
The higher the temperature, the higher the average kinetic energy
As kinetic energy increases, the average speed of particles increases as kinetic energy = ½ mv² (so as kinetic energy increases, if mass remains constant, velocity will increase)
Therefore, the greater the temperature, the faster the motion of the particles
How can a gas be compressed or expanded?
By pressure changes (inside and outside of the gas container)
The pressure of a gas causes a net force at right angles to the surface of its container
There is also a force on the outside of the container due to the pressure of the gas around it
If the container can easily change size, if the pressure outside of the container increases, it will cause the gas inside of the container to be compressed due to the overall force. However if the pressure outside of the container increases, it will cause the gas inside of the container to expand due to the overall force
What does the pressure of a gas produce?
A net force at right angles to the wall of the gas container (or any surface)
Give the formula for the pressure and volume of a fixed mass of gas held at a constant temperature
For a fixed mass of a gas held at a constant temperature:
pressure x volume = constant
p - V = constant
p - pascaps, Pa
V, metres cubed, m3
What does doing work on a gas cause?
Work is the transfer of energy by a force
Doing work on a gas increases the internal energy of the gas as energy has been transferred to the kinetic energy stores of the particles
and therefore can cause an increase in temperature of the gas
Charlie xcx is using a bike pump
As she is inflating the bike tyre, the tyre gets warmer, why?
When inflating a bike tyre, more particles are being forced into the tire
Work is being done against a force (the pressure of the gas inside the bike tyre) to push more particles in (as gases exert a net outward force at right angles to the surface of its container)
By doing work, charlie xcx is transferring energy to the particles inside the tyre
This energy is transferred to the kinetic energy stores of the particles
Increased kinetic energy leads to increased temperature, which causes the temperature of the tire to increase
The temperature of the helium in the balloon was increased. The mass and volume of helium in the balloon remained constant. Explain why the pressure exerted by the helium inside the balloon would increase.
The particles would gain kinetic energy
So they would collide with the walls of the balloon more frequently
Meaning that collisions exert a greater overall force on the balloon
So there is a greater overall force on the balloon per unit area
Explain the changes in the arrangement and movement of the particles as the ice melted and the temperature increased to 5 °C.
Particles in a solid are in a regular pattern
Whereas patterns in a liquid are in a random arrangement
Particles in a solid vibrate in fixed positions
Whereas within a liquid, particles are free to flow over each other
As the water warms up, the particles move faster
Because energy is transferred to their kinetic energy stores
As ice changes to liquid, the temperature remains constant
as energy is transferred to the potential energy store of the particles, not the kinetic (temperature is a measure of the average kinetic energy of particles within a substance)
Internal energy is the total kinetic and potential energy of the particles
Explain why the pressure increases as more air is pumped into the tyre
more (air) particles (in the tyre)
greater number of collisions with tyre (walls) per second
The air pressure in a car tyre changes if the temperature of the air in the tyre increases. Explain why. [4 marks]
As temperature increases, the particles have greater (mean) kinetic energy
So more collisions with the tire walls per second
And greater force in each collision
Each particle has greater momentum as they have greater velocity
This means that when they collide with the walls, they will have a greater change of momentum
Force during a collision is directly proportional to the rate of change of momentum
As force = momentum x acceleration
So force = (momentum x change in velocity) / time
So there is a greater force in each collision
Greater mean force per square metre causes greater pressure on wall of the tyre