Chapter 29 The Electron
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What are the properties of the electron
Orbits the nucleus
Very small mass
Negatively charged
Charge on electron is the smallest amount of charge found in nature
Who first measured the charge on the electron?
Robert Millikan
Who coined the term ‘electron’?
George Stoney (GJ Stoney)

What is Thermionic Emission?
The giving off of electrons from the surface of a hot metal
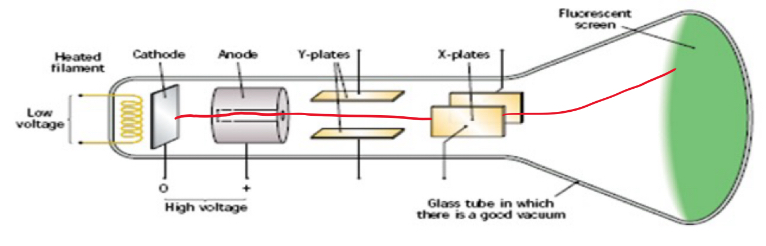
Draw a labelled diagram of the Cathode Ray Tube and describe how it works
Current is passed through the heating coil which heats the cathode and causes electrons to be emitted by Thermionic Emission
High voltage difference between the cathode and anode causes the electrons to accelerate across the tube. As a result current flows
Electrons pass through the hole in the anode and hit the fluorescent screen
Kinetic Energy from the electron is converted to Light Energy
What is the function of the filament?
It heats the Cathode
What happens at the Cathode?
Thermionic Emission occurs and electrons are emitted
What happens when the electrons hit the screen?
Their kinetic energy is converted to light energy. Light can be seen on the screen
What causes the electrons to be accelerated across the tube?
High voltage between Anode and Cathode
Why is there a vacuum in the tube?
So that the motion of the electrons is not opposed as they move across the tube
What are Cathode Rays?
The beams of electrons (streams of high-speed electrons) produced in a cathode ray tube are called Cathode Rays
Properties of Cathode Rays
They travel from the cathode in straight lines
They cause certain subtances to fluoresce
They can be deflected in electric and magnetic fields (x and y plates)
They can produce X-rays when they strike heavy metals
What is the Photoelectric Effect
The photoelectric effect is the emission of electrons from a metal due to electromagnetic radiation of a suitable frequency falling upon it
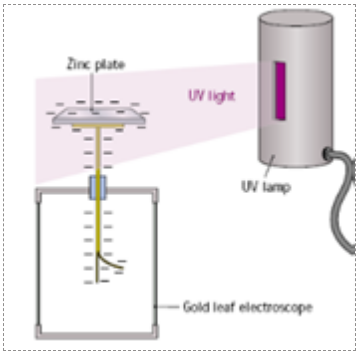
To demonstrate the Photoelectric Effect
Procedure
Charge a GLE negatively
Shine UltraViolet light on the zinc plate
Result
The leaf falls altogether
Observation
Shining UV light on the zinc plate liberates electrons from the zinc and therefore the leaves become neutralised and fall back together
Outline Einsteins explanation of the Photoelectric Effect
Light travels in packets of energy called Photons
Each Photon gives all of its energy to one electron
The energy needed to remove the loosest electron from the surface of a metal is the Work Function W of that metal
If the energy of each photon is less than the work function no electrons are emitted
If the energy of the photon is greater than the work function electrons are emitted
What is the Work Function?
The work function of a metal is the minimum energy needed to remove the loosest electron from the surface of that metal
What is the Threshold Frequency?
For a given metal the frequency below which photoemission will not occur is called the Threshold Frequency. Light of frequency above the threshold will cause photoemission
What is a Photon?
A photon is a packet of electromagnetic energy
The energy E of a photon is given by E=hf
Define the ElectronVolt
The electronvolt (eV) is the amount of energy gained or lost by an electron when it moves through a potential difference of 1V
Define Intensity
Intensity is defined as the incident light energy per unit area per unit time
As Frequency increases = electrons speed up
As Intensity increases = no. of electrons increases
What is a photocell?
A photocell is a device that conducts electric current when light of a suitable frequency shines on it.
The size of electric current it conducts is directly proportional to the intensity of light falling on it.
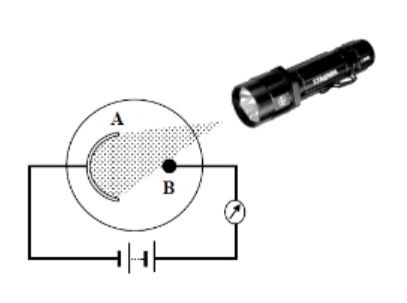
Draw a labelled diagram of a photocell
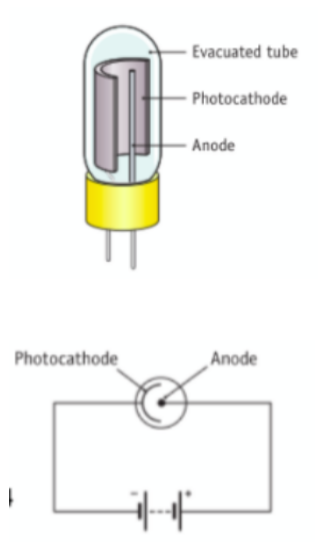
To demonstrate the action of a Photocell
Set up as shown
Light of a suitable frequency shines on A (photocathode)
This releases electrons by the photoelectric effect
Electrons are attracted to B (anode) and flow around the circuit
Note that if you bring the light source closer, there is a greater intensity of light (more photons) which will result in the release of more electrons and as observed, there is a greater deflection in the galvanometer
Applications of Photoelectric sensing devices
Automatic Doors
Fire Alarms
Photocopiers
Photocells
What are X-Rays?
X-rays are high frequency electromagnetic radiation produced when high speed electrons in a cathode ray tube strike a metal target that has a high melting point
Why can X-ray production be considered the inverse of the photoelectric effect?
In X-Ray production, electrons hit a target and electromagnetic radiation is given out
In the Photoelectric Effect, electromagnetic radiation strikes a target and electrons are given out
Draw a labelled diagram of a Cathode X-Ray Tube
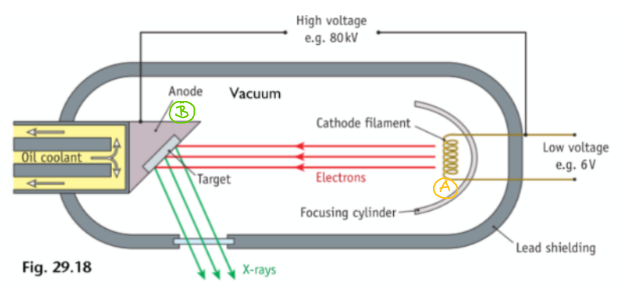
Decribe the Operation of an X-Ray
Low voltage supply heats the cathode
Electrons are emitted by thermionic emission
They get accelerated across the vacuum due to the very high voltage and smash into a high density anode (tungsten)
Most of kinetic energy gets converted to heat, which is removed with a coolant
X-rays are emitted all around and some exit through a small narrow window
Properties of X-Rays
They are electromagnetic waves
they cause ionisation of atoms
they have high penetration powers
What is ionisation?
Ionisation is the process where an atom or molecule loses or gains electrons to become an ion
Uses of X-rays in Real World
Medicine: To detect broken bones
Industry: To detect pipe breaks
What is the Hazard of X-Rays
They can ionise atoms in the body, causing them to become abnormal, which can lead to cancer