CHEM 3111 | Spectroscopy
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
wavelength symbol
λ
frequency symbol
v
energy equations
E = hc/λ (for one photon; 6.022 × 10²3 photons = 1 mol)
E = hv
in molecular absorption, energy ___ and is ____
increases, excited
in molecular emission, energy ____ and is _____
decreases, relaxed
visible region of light
400-700 nm
the highest energy molecular process that occurs when a molecular absorbs a microwave photon is ___
rotation
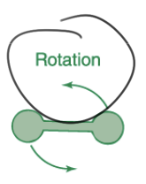
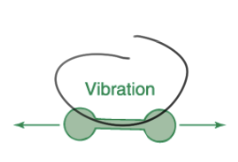
the highest energy molecular process that occurs when a molecular absorbs a infrared photon is ___
vibration
the highest energy molecular process that occurs when a molecular absorbs an ultraviolet photon is ___
electronic excitation

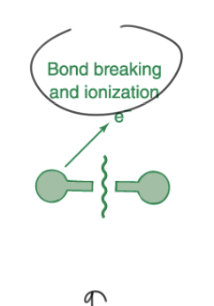
the highest energy molecular process that occurs when a molecular absorbs an x-ray photon is ___
bond breaking
cancer :(
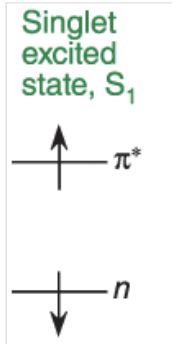
singlet state
no spin flip (faster)
triplet state
spin flip occurs (slower)

the first thing a molecule does when it absorbs is ____
relaxes
energy hierarchy
electronic excitation, vibration, rotation
any time a photon gains energy/becomes excited, it also changes the rotation and vibrational energy as a result
radiative process
photons are involved
nonradiative processes
no light emitted; energy lost as heat
rigid, sp2 hybridized molecules can’t relax as easily and need to ____ or _____
fluoresce or phosphoresce
does fluorescence involve spin flip?
no (shorter duration)
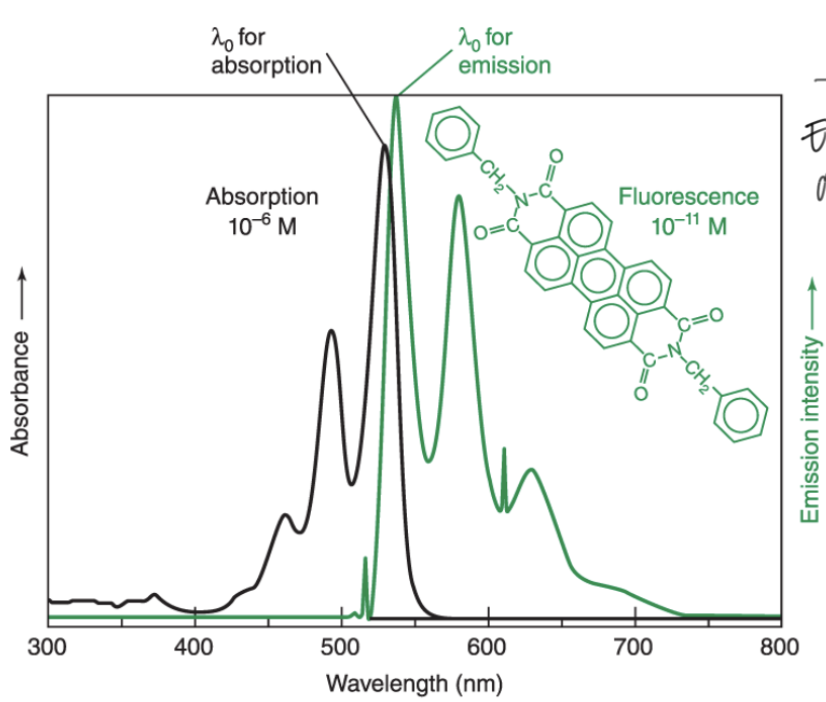
Stokes shift principle
Emission (energy released) always occurs at a LONGER wavelength than absorbance
Absorbance involves energy increase; shorter wavelength
Absorbance and transmittance have an inverse _____ relationship, where absorbance measures the light _____ by a sample and transmittance measures the light that ____ through
log, absorbed, passes
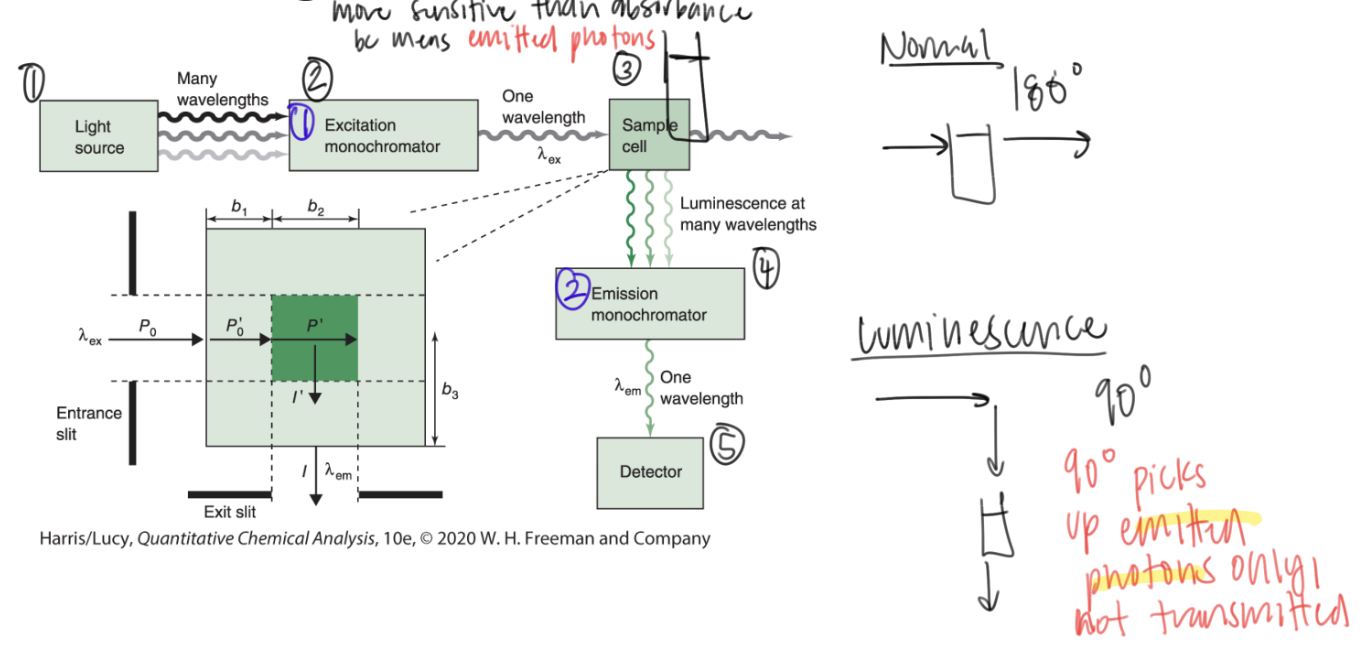
measuring luminescence differs from measuring regular light because it picks up ____ photons only, not transmitted photons
emitted
90 degrees
an ideal blackbody is an object that
absorbs all the energy that strikes it and emits light based off temperature (sun)
continuum sources
emits light at a large range of wavelengths
line sources
emits light only at specific wavelengths (laser)
monochromator
instrument that isolates white light into the visible spectrum and selectively isolates one wavelength (based on angle)
reflective or transmissive optical device w closely spaced lines
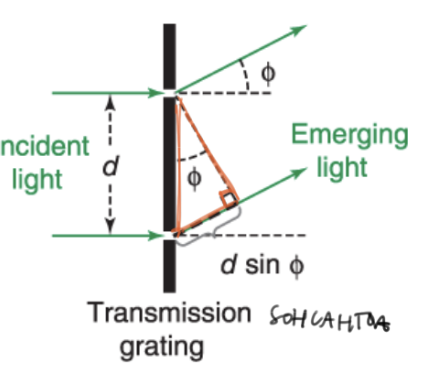
resolution
the ability of a spec to distinguish between two closely spaced wavelengths
minimum wavelength difference between 2 peaks that can be distinguished
resolving power (N) formula
λ/Δλ = nN
λ = avg of two wavelengths
Δλ = resolution (difference between two wavelenths)
n = defraction order
N = number of grooves (resolving power)
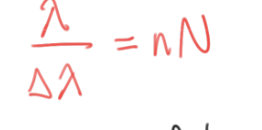
phototubes
phototubes produce electric currents when hit by transmitted light (light → electricity)
weak current
photomultiplier tubes
amplifies current in phototubes
electrons are accelerated when they hit a dynode (mario kart)