MII2 vaccination and immunotherapy
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
what is passive immunisation
immediate, temporary transfer of antibodies to an individual to provide rapid protection against infection
what are the two forms of passive immunisation
natural - infants acquire antibodies from their mother
artificial - transfer of antibodies from the blood of immune people
what is active immunity
the immunity that results from the production of antibodies by the immune system in response to the presence of an antigen.
what are anti-sera
a blood serum (often non human) containing polyclonal antibodies used to provide rapid, passive immunity against specific diseases, toxins, or venoms
how is the animal immunised
given non-lethal doses of an antigen, then the blood is collected and antigen purified
what are the applications of antibody transfer and give examples
rapid treatment during acute illness
- try to prevent full infection after exposure to a pathogen
- snake venom or toxins
- Ebola, measles, rabies etc
as a preventive measure
- cytomegalovirus after transplantation
what are two advantages of antibody transfers
fast acting (important for snake venom or rabies)
support for immunodeficiencies
what are 4 disadvantages of antibody transfers
antibody levels fall quickly
given intravenously
serum sickness
expensive and complex to produce and store
what is serum sickness
immune system reacting to the foreign antibodies from the transfer
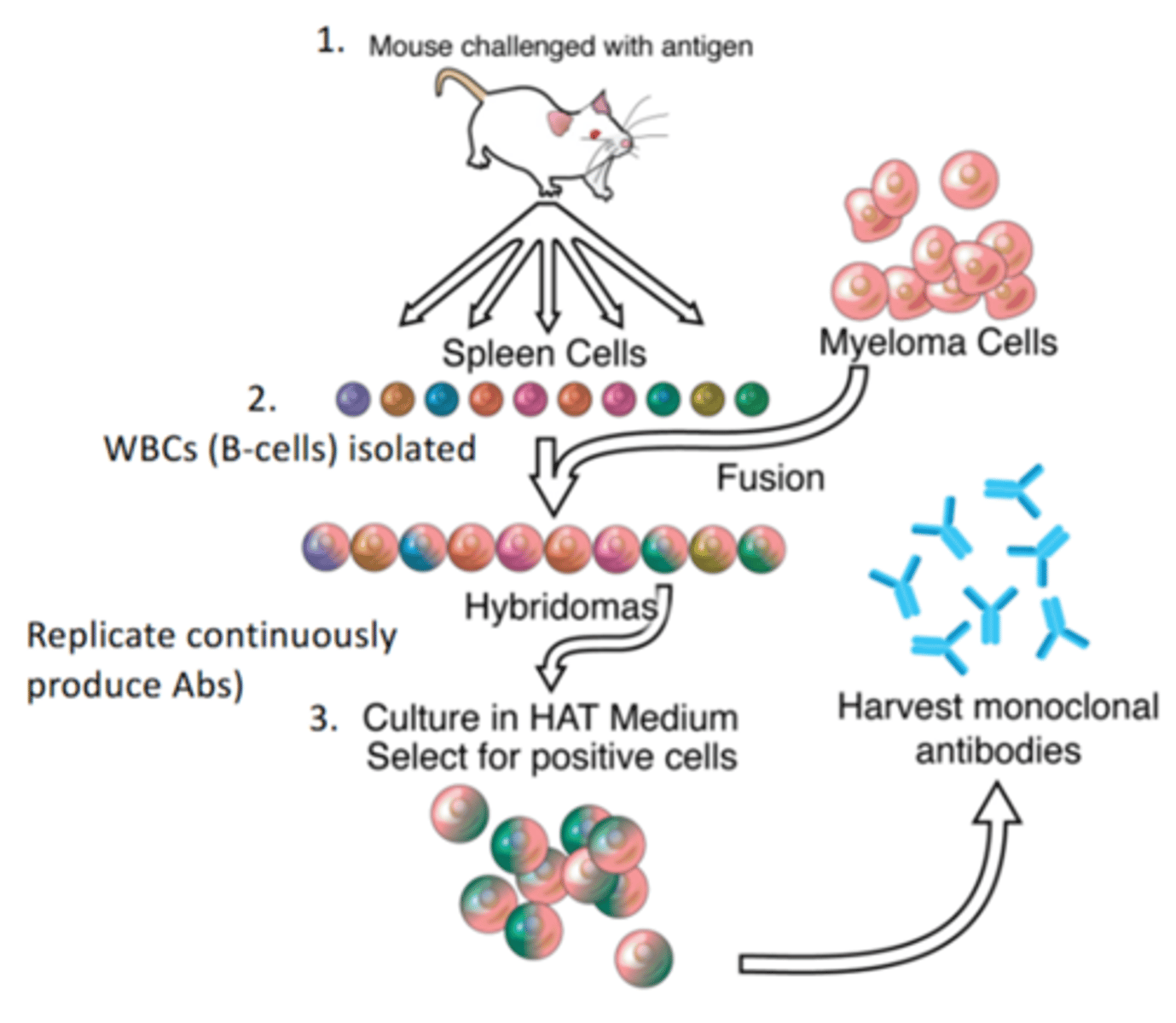
how are monoclonal antibodies made
Monoclonal antibodies are made by combining mouse B cells and a tumour cell to form a hybridoma
what are the advantages of monoclinal antibodies (2)
near unlimited supply
even rare antibodies can be isolated
can be manipulated
have a single specificity
why can't humans have mice antibodies
serum sickness
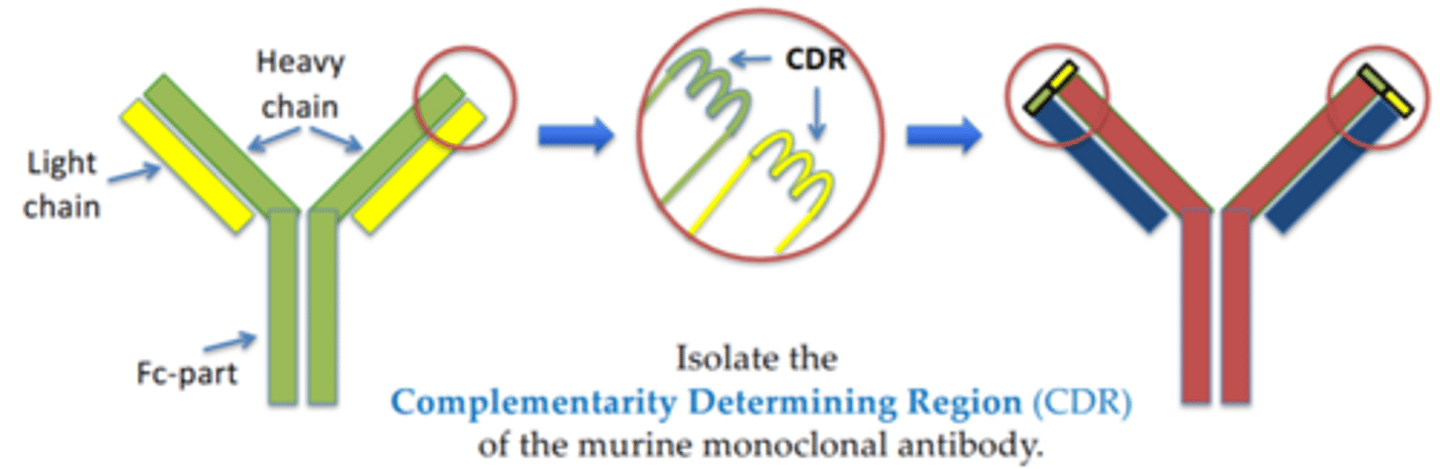
how are mice antibodies converted to human antibodies
Raise a monoclonal antibody with the right specificity from an immunised mouse.
Replace the CDR (hypervariable region) of a human monoclonal antibody with the CDR derived from the mouse monoclonal antibody.
what are the three modern ways of making monoclonal antibodies
1. phage display
2. EBV transformation
3. single cell expansion & selection
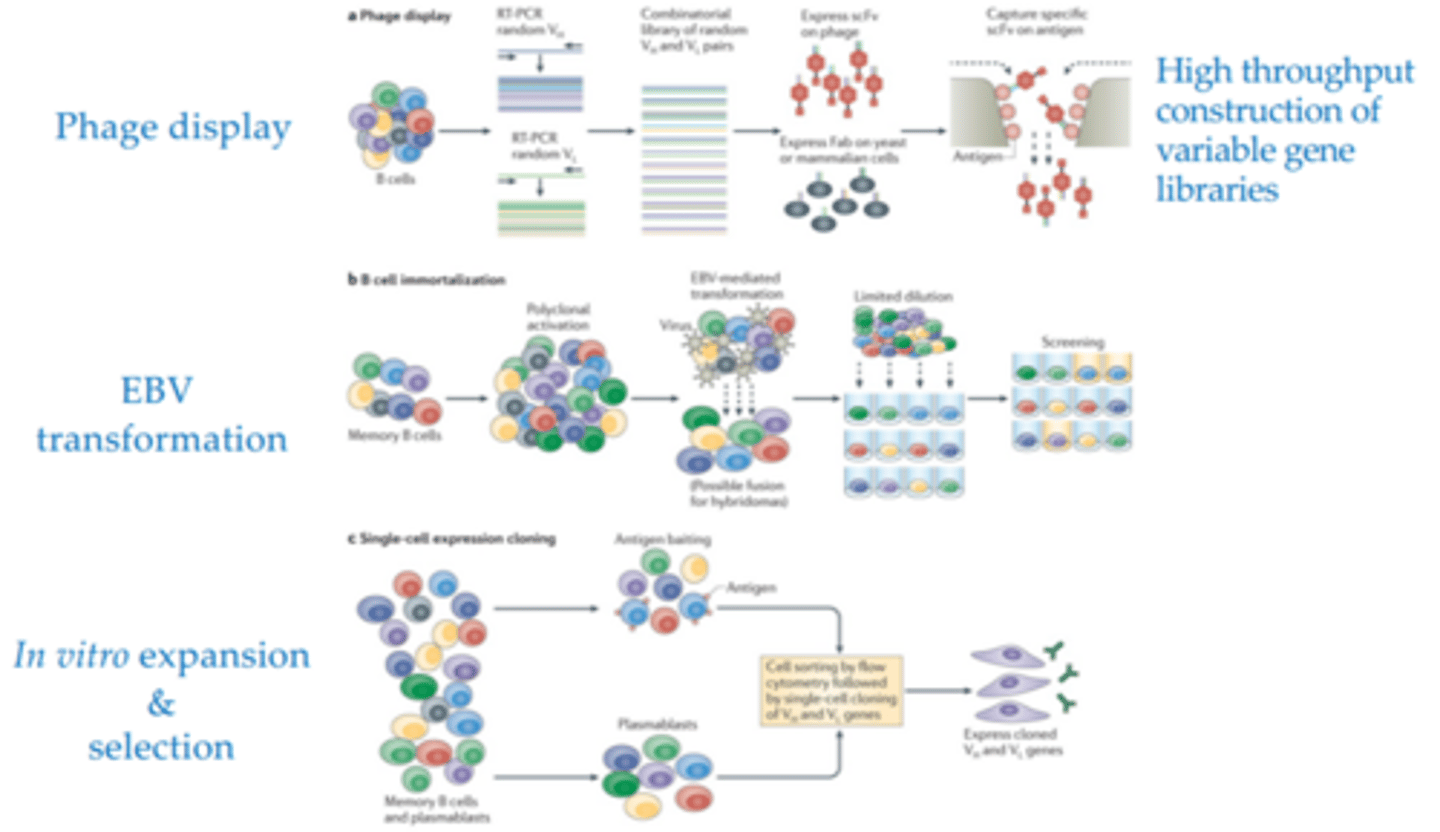
what is phage display
a bacteriophage is engineered to display a particular antigen on its surface
what is the principle of vaccination
• introduce the immune system to a pathogen in a "controlled" environment
• cause the immune system to remember the pathogen and respond to it
• enable the immune system to effectively clear the pathogen to prevent disease
what is the most common type of vaccine
live attenuated -- weakened/inactivated pathogen
how might a pathogen be weakened
virulent genes are deleted or modified
what chemical is often used for attenuating pathogens
formalin
give examples of diseases prevented with a live-attenuated vaccine
MMR, flu, yellow fever, polio
what is the mechanism of the live-attenuated polio vaccine
passage through non-human cells causes spontaneous mutation which cannot replicate in the nervous system
what are subunit vaccines
use components of pathogenic organisms, not the whole organism
what is the advantage of subunit vaccines
no extraneous pathogenic particles such as DNA
what is the principle of vector vaccines
Antigen genes are inserted into the vaccinia virus genome with virulence factors removed
which type of vaccine covered so far might require multiple doses
killed whole organism
how are chicken eggs used in vaccine production
Primarily for influenza—acts as a sterile environment to replicate large quantities of the virus.
- Virus strains are injected into fertilised chicken eggs to incubate and harvest for purification.
- These are used for the vaccine
what are some limitations to traditional vaccines
- not all organisms grow in culture
- expense
- insufficient attenuation?
- reversion to infectious state
what are virus-like particle vaccines
self-assembled homogenous nanoparticles derived from the coat proteins of viral capsids
give an example of a non enveloped virus like particle vaccine
HPV
do virus like particles contain genetic material
no
what are outer-membrane vesicle vaccines
Non-replicating, nano-sized particles secreted by gram-negative bacteria. These are highly immunogenic.
what uses outer membrane vesicle vaccines
N. meningitis
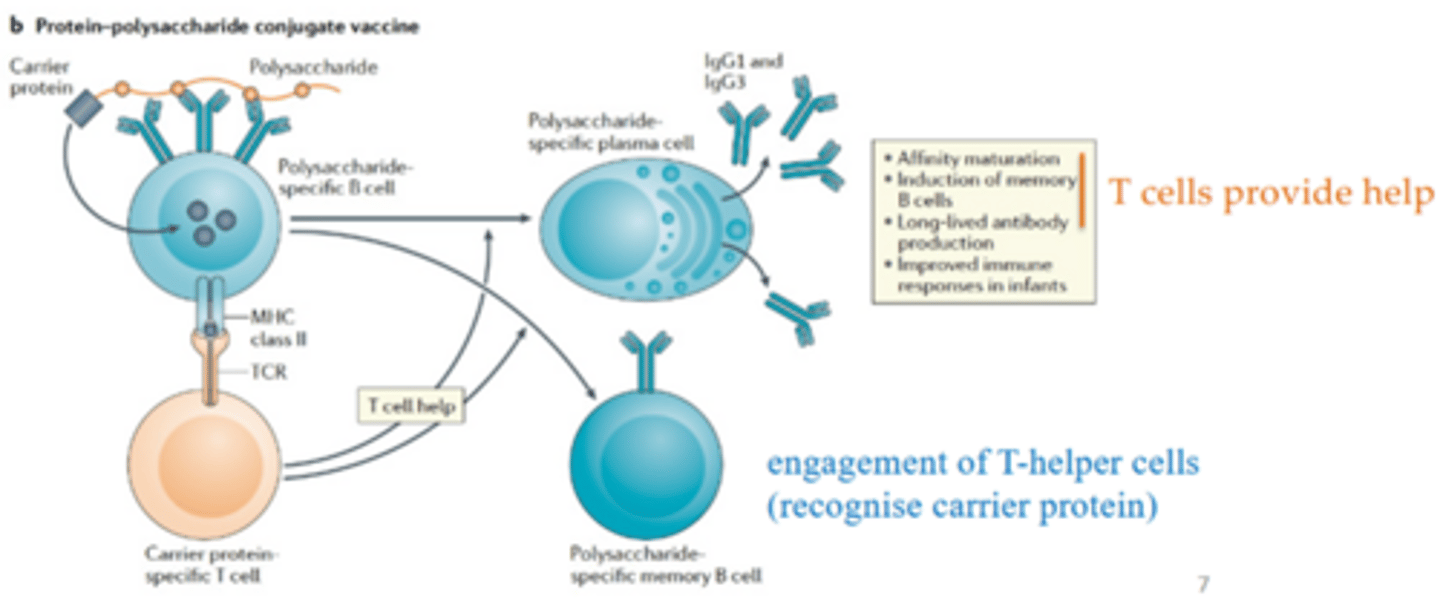
what is the principle of protein-polysaccharide conjugate vaccines
Combines a bacterial capsular polysaccharide with a carrier protein to induce strong T cell immunity.
what type of vaccine was the covid-19 vaccine originally
viral vector
why was a chimpanzee adenovirus used for the covid vaccine (2)
1. unlikely to be pre-immune to the vector
2. encoded the spike protein of covid
what type is the modern covid vaccine
mRNA vaccine
what is the mRNA for the covid vaccine enclosed in
lipid nanoparticles
do the covid vaccines require adjuvant?
no
how does the intranasal 'FluMist' flu vaccine work
it is live-attenuated and replicates in the nose (cold adapted), causing a small immune response. this modified virus strain cannot replicate in the temperature of the lungs
what is the necessity of adjuvants
enhance and direct the adaptive immune response to the vaccine antigens.
what might happen without adjuvants
immune tolerance/ignoring the antigen
how do adjuvants direct adaptive response
activate DCs by mimicking components of microbes that bind to PRRs
what is the most widely used adjuvant
alum or aluminium salts
what do adjuvants increase release of at injection site
chemokines and cytokines
what are two risks of vaccine adjuvants
1. increases reactogenicity locally or systemically
2. nonspecific immune activation (rare)
- immune mediated disease
- inflammatory disease

when can vaccine derived infection occur
in areas where sanitation is poor, pathogens (from the vaccine) can replicate and be passed on to non vaccinated people through faecal/oral routes
what can cause live-attenuated pathogens to become dangerous
mutations can revert them to their pathogenic state
also lowkey not good for immunocompromised people
what are 3 advantages of live-attenuated
activates all phases of the immune system
provides more durable immunity, less boosters needed
fast acting
what is original antigenic sin, and what it is also known as
Also known as immunological imprinting, it is the tendency of the immune system to preferentially utilise immunological memory based on the first strain of a pathogen it encountered when subsequently encountering a slightly different strain.
which process makes flu vaccines change each year
antigenic drift
what is a phase I human trial for
testing safety and dose response - 6-20 people
how many participants in a phase II human trail
200-300 people
what is a phase III human trial for
efficacy (reduces cases), identifying rare side effects (1000s of participants)
what is an experimental way of testing vaccine efficacy
check for the antibodies using radioimmunoassay or ELISA protocol
what is the synovium
The Synovium is a thin layer of tissue inside the synovial capsule that secretes synovial fluid.
what causes rheumatoid arthritis
autoimmune disorder
what causes immune cells to attack mucosal sites in RA and what does this result in
post translational modifications to joint cells leading immune cells to not recognise them as self, cyto/chemokine build-up and autoantibody formation.
hyperplasia of the lining layer, bone and cartilage destruction
what are the two types of antirheumatic drugs (DMARDs)
Synthetic DMARDs and Biological DMARDs
what does DMARD stand for
disease modifying anti-rheumatic drugs
what is the most common target of biological DMARDs
Tumor necrosis factor alpha - TNFa (TNFa inhibitors)
how is TNFa involved in rheumatoid arthritis
it is a cytokine involved in inflammation. When it is overproduced it causes inflammation and cell death which leads to arthritis
what are the two methods of blocking cytokine action
1. neutralise the cytokines before they reach the receptor
2. block the receptor with an antagonist or monoclonal antibody
which method of blocking cytokine action is used for TNFa
neutralise the cytokine
what are the three methods of preventing cytokine binding to tis receptor
soluble receptor, natural antagonist, monoclonal antibody
describe the immune systems control of cancer (4 steps)
1. dead cancer cells release antigens that activate the immune system
2. T cells are trafficked to the tumour
3. T cells infiltrate the tumour and kill the cells
4. T cells die and release more antigens
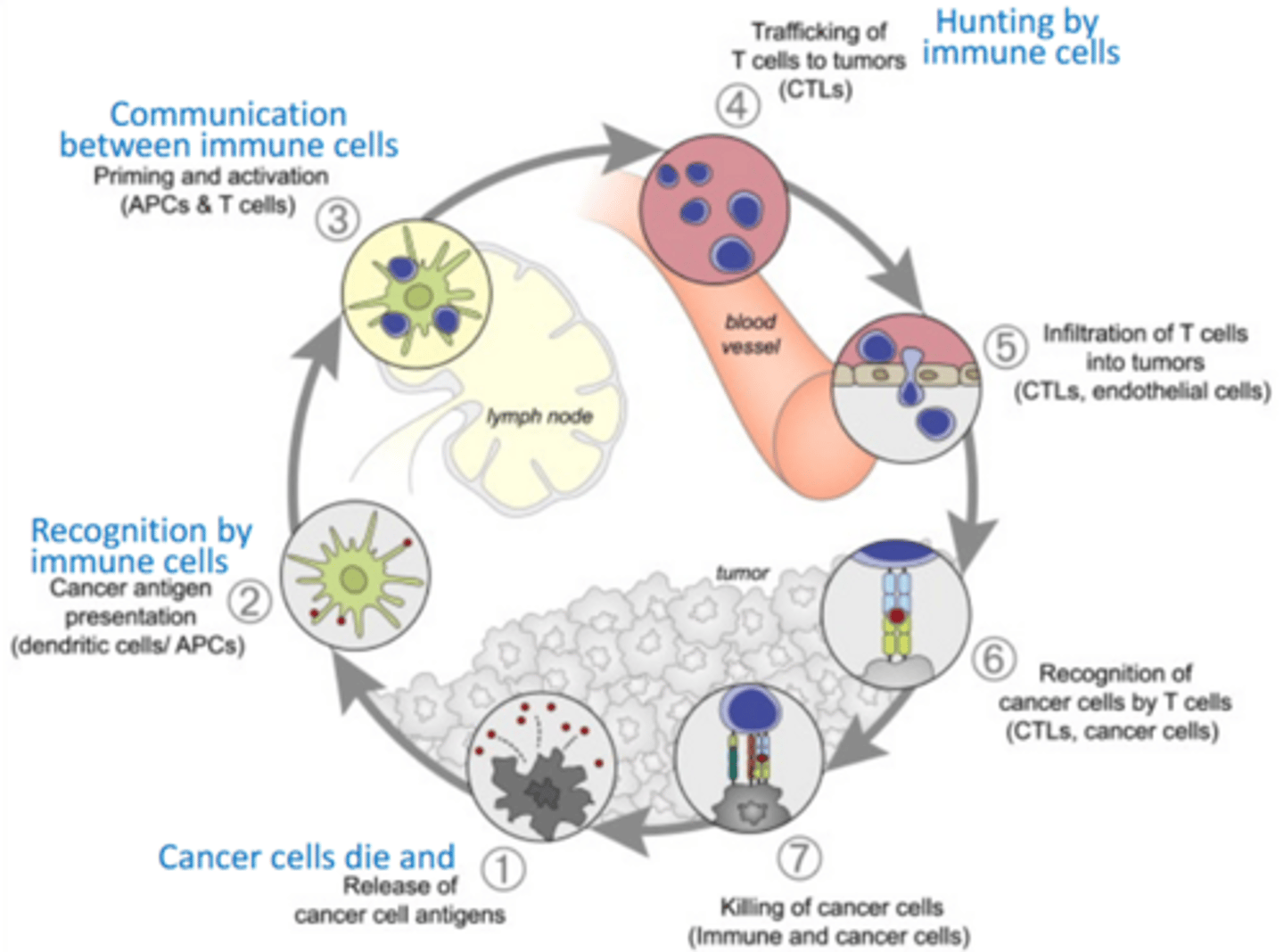
what is the 3 step process by which tumour cells can evade the immune system
1. immunoediting
2. immunosuppression
3. escape
describe the stages of tumour cells evading the immune system
1. immunoediting - The cancer cells change from highly immunogenic to poorly immunogenic
2. Immunosuppression - Use of immunosuppressive molecules such as PDL1
3. Escape - Large tumours induce stromal cells to surround the tumour and act as a physical barrier to the immune cells
how can monoclonal antibodies contribute to tumour therapy (2)
1. target specific protein overexpressed on specific tumour cells
2. target proteins normal expressed on certain cell types and also on tumour cells
give an example of a protein that is overexpressed on tumour cells (protein and what tumour)
HER2 on breast cancer cells
give an example of a normally expressed cell found on tumour cells (protein and what tumour)
CD20 on B cell lymphomas
what is the HER group of receptors
human receptor for EGF
how is HER2 involved in tumorigenesis
activation of HER1,3,4 activates HER2. Amplification of the HER2 gene leads to overexpression of HER2 proteins, increasing cell division and growth.
what type of therapy is used to target HER2
monoclonal antibodies
how can targeting CD20 on B cell lymphomas prevent tumorigenesis
Using a monoclonal antibody that targets and binds the CD20 antigen on B lymphocytes.
The binding marks the B cell for destruction via CDC (complement dependant cytotoxicity) and ADCC (antibody dependant cellular cytotoxicity), depleting CD20 cells.
what are tumour antigens
any antigen expressed by the cancer cells that elicits an immune response
what is a neoantigen
mutations in normal human proteins that are recognised as foreign by the immune system
what will display the neoantigens to T cells
type I MHC
will a T cell be activated once shown a neoantigen?
no, they need the second co-stimulatory signal
what can a tumour do to prevent T cell activation once bound
up regulate CTLA4 and PD1
why does upregulating CTLA4 and PD1 prevent T cell activation
they are inhibitory surface co-stimulatory receptors that will prevent T cell activation when binding.
what therapy can be used to inhibit PD1 and CTLA4
monoclonal antibodies
what is CAR T-cell therapy
personalised immunotherapy that modifies a patients T lymphocytes to recognise and destroy cancer cells
what does CAR T stand for
chimeric antigen receptor T cell
what feature of CAR T cell cytoplasm enhances tumour killing
it contains extra co-stimulatory domains
what is the benefit of personalised anti cancer therapy as opposed to universal
less likely to be rejected by the body