Cell Bio - Test 1 flashcards
1/51
Earn XP
Description and Tags
Test 1 flashcards
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
A cell with endoplasmic reticulum most likely belongs to the domain _____.
Bacteria
Eukaryotes
Archaea
Cannot be determined
Eukaryotes
Name the purine nucleotides.
Adenine and Guanine
The lenses in an electron microscope are actually_____.
Concave Glass
Convex Glass
Heavy metal calipers
Magnets
None
Magnets
Which amino acid is incompatible with the formation of alpha helices?
Proline
Which of the following is/are present in all cells?
Cytosol
Plasma Membrane
Ribosomes
All of the above
Cytosol, Plasma Membrane, Ribosomes (All of the above)
A monosaccharide with 4 carbons will have ____ oxygens and ____ hydrogens.
4 and 4
4 and 8
8 and 12
8 and 8
none
4 and 8
At least some of the illumination source passes through specimens in all of the following types of microscopes, except?
Bright field
Phase contrast
Scanning electron
Transmission electron
Name four types of proteins.
Structural, defensive, hormonal and receptor
Which type of microscopy requires coating the specimen with heavy metal?
Bright-field
Phase‑contrast
Scanning electron
Transmission electron
All of the above
Scanning electron
Name two basic amino acids
Histidine, Lysine and Arginine
The earliest light microscopes were noted in the _____.
1400’s
1600’s
1800’s
1900’s
1600’s
Sucrose is a disaccharide made of _____.
2 glucose monomers
Fructose and galactose
Fructose and mannose
Fructose and glucose
None of the above
Fructose and glucose
Scientists estimate that prokaryotes were first found on earth approximately ___ billion years ago.
0.9
2.2
3.5
5.1
3.5
The cell theory was proposed by _______.
Hooke and Scwhann
Schleiden and Schwann
Schleiden and Thomas
Schwann and Van Leeuwenhoek
None of the above
Schleiden and Schwann
Extra credit: Name the three domains of life.
Eukaryotes, Bacteria, and Archea
Which of the following are true of ALL proteins?
They have ligands.
They have substrates.
They have quaternary structure.
All of the above are correct
None of the above is correct
None of the above
The amino acid sequence of a polypeptide is its ____ structure.
Primary
Secondary
Tertiary
Quaternary
Primary
When was the cell theory proposed?
1665
1839
1931
1953
1839
Which of the following polymers are created by dehydration or condensation reactions?
Polysaccharides
Polypeptides
Nucleic acids
All of the above
None of the above
All of the above
Which of the following is correct about alpha helices?
They are a type of tertiary structure.
They form when the side chain of one amino acid bonds with the alpha carbon of another amino acid.
When they are in proteins in the cytoplasm, hydrophobic side chains are on the outside of the protein.
They are stabilized by hydrogen bonding.
All of the above are correct
They are stabilized by hydrogen bonding
Seeing things clearly using a microscope depends on all of the following, except _______.
Contrast
Magnification
Resolution
Microscope working distance
All of the above are important
Microscope working distance
Which of molecules below demonstrates the ability of chains of carbons to form ring structures?
Monosaccharides
Nucleotides
Steroids
All of the above
None of the above
All of the above
How many nanometers are in 1.78 centimeters
1.78 × 10^7 nanometers
Which fatty acid type contains NO double bonds?
Omega‑3
Polyunsaturated
Saturated
Unsaturated
All of the above
Saturated
The simplest amino acid is ____, and its R group is ___.
Alanine . . . a hydrogen atom
Alanine . . . a methyl group
Glycine . . . a hydrogen atom
Glycine . . . a methyl group
Glycine… a hydrogen atom
Beta-sheets in which the amino (N)-termini of the beta-strands are aligned are described as ____.
Antiparallel
Conservative
Oblique
Parallel
None of the above
Parallel
What is the carbohydrate in DNA, and how many carbons are in it?
Deoxyribose, 5 carbons
How many valence electrons are in carbon ___?
2
3
4
5
4
Which of the following chemical groups is not present in the compound in the figure?
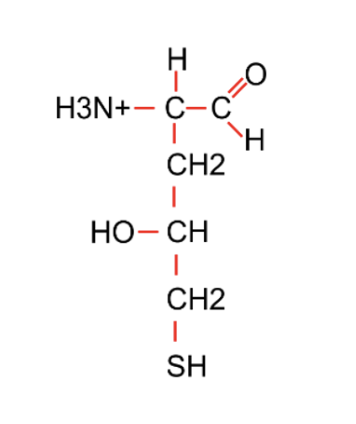
Amino
Carboxylic acid
Hydroxyl
Sulfhydryl
All of the above
Carboxylic Acid
A derivation of the nucleotide adenine forms the energy molecule ____.
ATP-adenine triphosphate
What living things are the universal building blocks of all living things?
Cells
DNA
Lipids
Proteins
All of the above
Cells
Name two of the uncharged, polar amino acids
Serine (Ser, S)
Threonine (Thr, T)
Cysteine (Cys, C)
Tyrosine (Tyr, Y)
Asparagine (Asn, N)
Glutamine (Gln, Q)
What would the resolution of a microscope using a light source with a wavelength of 400 nm and a numerical aperture of 1 be?
400 nm x 0.61/1=
244
Which of the following is a part of all amino acids?
Aldehyde group
Amino group
Hydroxyl groups
Ketone group
All of the above are parts of all amino acids
Amino group
Which of the following chemicals bonds is strongest in cells?
Covalent
Hydrogen
Ionic
Van der Waals interactions
Covalent
The majority of amino acids have side chains that are ____.
Acidic
Basic
Nonpolar
Polar
Nonpolar
-
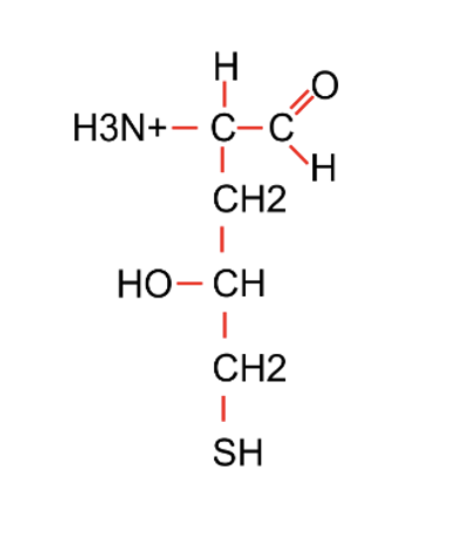
Which of the following chemical groups is not present in the compound in the figure?

Amino
Carboxylic acid
Hydroxyl
Sulfhydryl
All of the above are present
Carboxylic acid
Which of the following types of bonds act in protein folding?
Electrostatic attractions
Hydrogen bonds
Hydrophobic forces
van der Waals interactions
All of the above
All of the above
Which of the following polysaccharides is made by humans?
Amylose
Cellulose
Glycogen
All of the above
None of the above
Glycogen
In a polar covalent bond consisting of hydrogen and oxygen, the _______.
Hydrogen has a partial negative charge because it is more electronegative than oxygen
Hydrogen has a partial positive charge because it is more electronegative than oxygen
Oxygen has a partial negative charge because it is more electronegative than hydrogen
Oxygen has a partial positive charge because it is more electronegative than hydrogen
Oxygen has a partial negative charge because it is more electronegative than hydrogen
The percentage of water in cells ranges from _____ percent.
70-95%
Name the four macromolecules of life.
Carbohydrates, proteins, fatty acids, nucleotides
An electron microscope would be needed to view which of the following?
Mitochondria
Nucleus
Ribosomes
All of the above
None of the above
Ribosomes
Name 4 things that all living things do.
Cellular Respiration, Reproduce, Consume Nutrients, Excrete Nutrients
Who showed in the laboratory that spontaneous formation of organic compounds that could form the macromolecules of life could occur in conditions found on primitive Earth?
Gunther Rudenberg
Robert Hooke
Stanley Miller
Theodor Schwann
None of the above
Stanley Miller
There are ___ common amino acids.
10
20
40
50
20
Which of the following chemical bonds is due to the random, ever-changing accumulation of electrons in one part of a molecule?
Hydrogen bonds
Covalent bonds
Van der Waals interactions
Hydrophobic forces
Van der Waals interactions
Two microscopes with the same lenses are using different light sources. Microscope A is using visible light. Microscope B is using ultraviolet light that has a wavelength of 350 nm. Which of the following is true?
The magnification of microscope A is greater than the magnification of microscope B.
The magnification of microscope B is greater than the magnification of microscope A.
The resolution of microscope A is better than microscope B.
The resolution of microscope B is better than microscope A.
The resolution of microscope B is better than microscope A.
If the pH of cells the normal range of approximately 7.2, the ______ group in amino acids can donate a hydrogen to lower the pH.
Amino group
Carboxyl group
Sulfhydryl group
All of the above
Carboxyl group
Which of the following atoms is most likely to form an anion in a dissociated ionic bond?

Carbon
Fluorine (F)
Nitrogen
Lithium (Li)
Fluorine (F)
Which of the following elements make up 96% of all living matter? (select all that apply)
Calcium
Carbon
Hydrogen
Nitrogen
Oxygen
Carbon
Hydrogen
Nitrogen
Oxygen