Group 2, the alkaline earth metals
1/46
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What ions do group 2 elements form when they react?
2+ ions.
What do the electron configurations of all group 2 elements end in?
s2 subshell.
What is the trend in atomic radius going down group 2 and why.
Atomic radius increases going down group 2, this is because the number of shells increases.
Describe and explain the trend in first ionisation energy going down group 2.
First ionisation energy decreases going down group 2, this is because atomic radius decreases meaning the attraction between the nucleus and the outer shell of electrons decreases and shielding increases meaning less energy is required to remove an electron.
Why does the first ionisation energy decrease going down group 2 despite the increasing number of protons?
Because the increasing strength of the shielding going down group 2 overrides the increase in the number of protons going down group 2.
Describe the general trend in melting point going down group 2.
Melting point decreases.
What type of structure do group 2 elements form?
Metallic structure (2+ metal ions attracted to delocalised electrons).
Explain why the melting point generally decreases going down group 2.
The size of the metal ion increases going down group 2 whereas the number of delocalised electrons and the charge of the ion remains the same. Larger ions mean there is a greater distance between the positive nucleus of the metal ions and the delocalised electrons meaning the attractive forces between them are weakened and require less energy to break.
How and why is magnesium an exception in the melting point trend going down group 2?
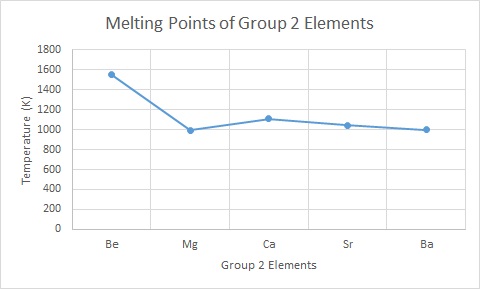
Magnesium is structurally different to other group 2 metals meaning it has weaker attractive forces (metallic bonding) that require less energy to overcome.
What do group 2 elements form when they react with water?
Metal hydroxides (bases) and hydrogen gas.
Give the general balanced symbol equation for the reaction between a group 2 element and water.
(M = group 2 element).
M (s) + 2H2O (l) → M(OH)2 (aq) + H2 (g)
Describe the trend in reactivity with water going down group 2.
Reactivity with water increases going down group 2.
True or false? Beryllium reacts with water at room temperature.
False. BERYLLIUM DOES NOT REACT WITH WATER AT ROOM TEMPERATURE.
Explain the trend in reactivity with water going down group 2.
Atomic radius increases the attraction between the nucleus and outer shell electrons decreases as well as shielding increasing, this means electrons are lost more easily so increased reactivity.
True or false? Magnesium reacts slowly with cold water but more vigorously with steam.
True.
What is the difference in the products of the reaction between magnesium and steam as opposed to the products of the reaction between magnesium and water?
The reaction between magnesium and steam produces magnesium oxide whereas the reaction between magnesium and water produces magnesium hydroxide.
Give the balanced symbol equation for the reaction between magnesium and steam.
Mg (s) + H2O (g) → MgO (s) + H2 (s)
True or false? Group 2 hydroxides and group 2 sulfates have opposite solubility going down group 2 (one increases, one decreases).
True.
Describe the trend in group 2 hydroxides solubility going down group 2.
Solubility of group 2 hydroxides increases going down group 2.
(Sparingly soluble → slightly soluble → fairly soluble → very soluble).
Describe the trend in the solubility of group 2 sulfates going down group 2.
Solubility of group 2 sulfates decreases going down group 2.
(Very soluble → fairly soluble → slightly soluble → sparingly soluble).
What group 2 based substance do you use to test for sulfates?
Barium chloride (BaCl2).
OR
Acidified barium chloride (allows you to skip step of adding acid).
What equipment do you need to test for sulfates?
Test tube.
Dropping pipette.
Talk me through how to test for sulfates using barium chloride.
1) Add some of the sample to a test tube.
2) Add some hydrochloric acid to the test tube (to remove any carbonates in the sample that could give a false positive result).
3) Add barium chloride to the test tube.
What would a positive result for the test for sulfates with barium chloride look like? What substance is the positive result?
Formation of white precipitate ((insoluble) barium sulfate (BaSO4)).
Give the general symbol equation for the reaction between barium chloride and a sulfate.
BaCl2 (aq) + XSO4 (aq) → BaSO4 (s) + XCl2 (aq)
Give the ionic equation for barium chloride reacting with a sulfate.
Ba2+ (aq) + SO42-(aq) → BaSO4 (s)
True or false? Group 2 compounds can be used to neutralise bases.
False. GROUP 2 COMPOUNDS ARE BASIC THEMSELVES SO CAN BE USED TO NEUTRALISE ACIDS.
Give one use of calcium hydroxide.
Neutralising acidic soil.
Give one use of magnesium hydroxide.
Antacid.
What is barium meal?
Barium sulfate.
Give one use of barium meal.
When ingested it can identify problems within the digestive tract (patient drinks barium sulfate suspension that coats the linings of soft tissue in the digestive tract, barium sulfate absorbs x-rays so soft tissues show up on x-rays).
True or false? Barium compounds are toxic.
True.
Why are people able to safely ingest barium meal despite it being toxic?
Barium sulfate is insoluble so cannot be absorbed into the blood.
Which group 2 metal is used to extract titanium from it’s ore?
Magnesium.
Why is titanium useful?
It is lightweight but strong.
What is titanium ore?
Titanium oxide (TiO2).
Describe what happens in the first step of extracting titanium from titanium ore.
The titanium ore (TiO2) is heated with carbon and chlorine gas, converting it into titanium chloride (TiCl4).
Describe what happens in the second step of extracting titanium from titanium ore.
The titanium chloride produced in the first step is passed through a fractional distillation column to increase it’s purity.
Describe what happens in the third step of extracting titanium from titanium ore. Give a balanced symbol equation for this reaction.
The purified titanium chloride is reduced using magnesium in 1000 C furnace, producing titanium.
TiCl4 + 2Mg → Ti + 2MgCl2
Talk me through the entire process extracting titanium from titanium ore. Include any relevant equations.
1) Titanium ore is converted to titanium chloride by heating it with carbon and chlorine.
2) The titanium chloride is passed through a fractional distillation column to increase it’s purity.
3) The purified titanium chloride is reduced using magnesium in a 1000 C oven, 2Mg + TiCl4 → 2MgCl2 + Ti
Give one use of both calcium carbonate and calcium oxide.
Remove sulfur dioxide gas (a pollutant) from emissions.
What is wet scrubbing?
A process in which an alkali is used to remove sulfur dioxide (acidic gases) from flue gas, the alkali is sprayed onto the sulfur dioxide gas.
Give the balanced symbol equation for wet scrubbing with calcium carbonate.
CaCO3 (s) + 2H2O (l) + SO2 (g) → CaSO3 (s) + 2H2O (l) + CO2 (g)
What is the name of CaSO3?
Calcium sulfite.
Give the balanced symbol equation for wet scrubbing using calcium oxide.
CaO (s) + 2H2O (l) + SO2 (g) → CaSO3 (s) + 2H2O (l)
What is one use of calcium sulfite?
Production of plasterboard.
Why does barium chloride need to be acidified (or add acid to sample) when using it to test for sulfates?
The acid neutralises any carbonates that may be present in the sample that could react with barium to form barium carbonate, another white precipitate that would be a false positive result.