Lecture 11 – Protein Functions
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Innate vs. Adaptive Immunity
vertebrates have immune systems
Our immune system can be broken down into innate and adaptive systems
What are innate systems?
◦Skin, mucous membranes and secretions, and the normal microflora found on our skin and other areas
◦We also have phagocytic cells that can envelop and destroy pathogens, inflammatory processes, and antimicrobial substances
What is a big advantage we have over some other organisms?
◦a big advantage we have over some other organisms is the adaptive system
◦Composed of two main components B and T cells and antibodies
How is the adaptive immune system “adaptive”?
It is able to change, depending on what it has encountered previously
This change is mediated by the B cells and T cells
B cells
produce the antibodies and retain the memories of the adaptive system
T cells
help out presenting antigens and carry out eliminating the antigens from the system
What is A big role in the adptive immune system and the things that can change over time?
Antibodies
◦They can be variable, and can be created to attach to many different antigens
What is an antigen?
A substance that elicits an immune response
◦So, it’s not just infectious particles, it can be a wide variety of substances, including oneself
How do antibodies get produced in large numbers?
immunoglobin receptors on B cell surface recognize and attach to antigen, which is then internalized and processed. Within the B cell a fragment of the antigen combines with MHC class II
MHC clas II- antigen fragment complex is displayed on B cell surface
Receptor on the T helper cell Th recognizes complex of MHC class II and antigen fragment and is activated- roducing cytokines, which activate the B cell. The tH cell has been previously activated by an antigen displayed on dendritic cell
B cell is activated by cytokines and begins clonal exapnsion. Some of the progeny become anti-body producing plasma cells
Why are antibodies useful?
Five reasons – Not just useful for adaptive immunity!
Agglutination, Neutralization, Opsonization, Activation of complement, and Facilitate binding
Agglutination
◦when pathogens are clumped together by antibodies (especially IgM antibodies), they cannot induce target cells to engulf them, and the clumps can be easily phagocytized
Neutralization:
antibodies block the surface protein’s ability to bind to receptors on the target cells
Opsonization:
an antigen that is coated with antibodies is easier to phagocytize
Activation of complement
◦initiates the inflammatory response, resulting in cell lysis
Complement activation is a crucial component of the innate immune system, consisting of over 30 plasma proteins that, upon activation, form a cascade to destroy pathogens, induce inflammation, and clear damaged cells.
Facilitate binding
◦Facilitate binding of certain effector cells that can destruct large antigens
How are antibodies made? Step one:
An immature B cell is exposed to antigens
◦This produces the first wave of effector B cells (plasma cells), which produce soluble antibodies for the threat
◦Other populations of the adaptive immune system target and eliminate the antigen
How are antibodies made? step two
Exposure to a second antigen at a later date triggers the memory cells replicate and become:
◦New effector B-cells (plasma cells) and
◦New memory cells
This whole process happens much faster and with a considerably more vigorous response than the previous time
◦The reason why vaccines work so effectively…
Two developmental paths for stimulated B lymphocytes.
Exposure to antigen causes two kinds of cells to develop from immature B lymphocytes. Effector B cells, or plasma cells, synthesize soluble antibody. Memory cells carry membrane-bound antibody to allow a rapid and enhanced response to a second exposure of the same antigen.
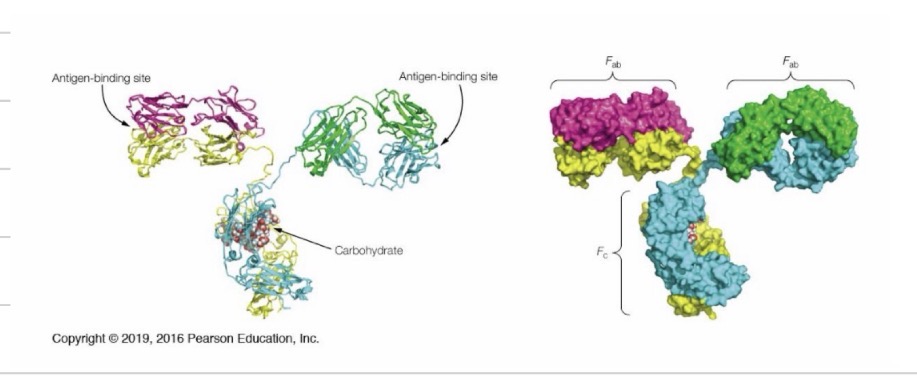
Antibodies – Basic Structure
They are immunoglobulin proteins
◦Globular proteins that are involved with the immune system
◦This is why they are sometimes shortened to (Ig)
What do antibodies look like?
◦Four individual immunoglobulin protein molecules make up one antibody
◦Two identical heavy chains
◦Two identical light chains
◦All the molecules are bonded together by di-sulfide bridges
antigen binding site
Each of the four chains of an antibody has a variable (V) region, and a constant (C) region
The variable region of one heavy and one light chain together to make up one antigen binding site that is specific to one epitope
Most antibodies have two identical antigen-binding sites (the two parts between the short and long ends of the chain)
epitope
(also called antigenic determinant) The specific portion of an antigen particle that is recognized by a given antibody or T-cell receptor.
The Fc region
The “stem” of an antibody is made up of the lower arms of the constant regions on the heavy chains that are not paired to the light chains
◦The Fc region
◦The constant regions on the heavy chains do not vary between antibodies for different epitopes, but do change through the course of an infection
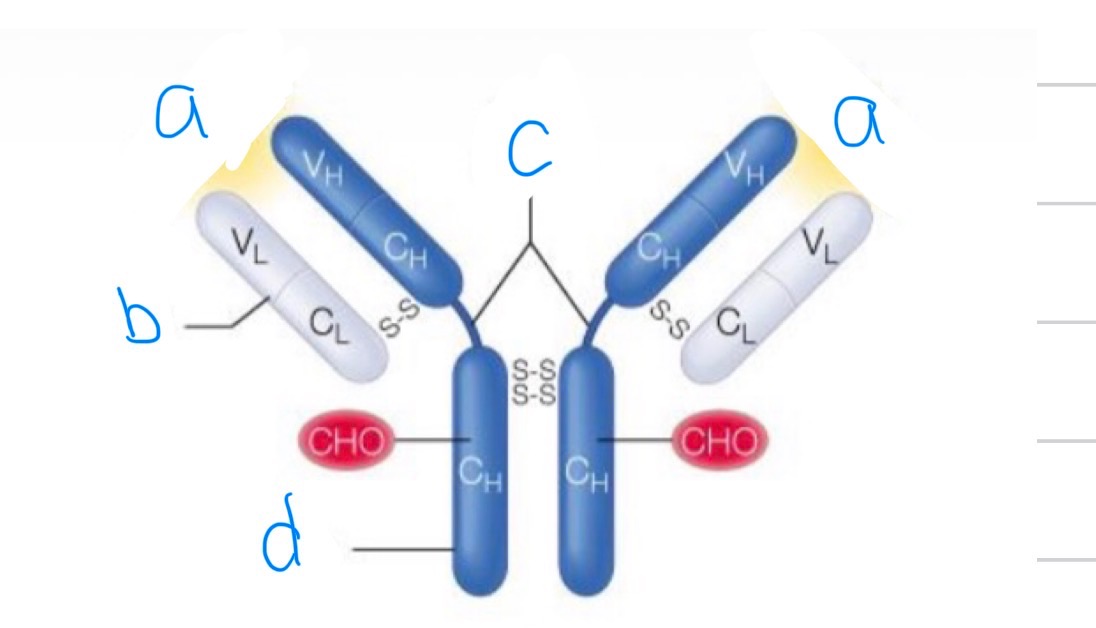
What is this? Label it
An anti body
A) antigen binding site
B)Light chain
C) Hinge region
D)Heavy chain
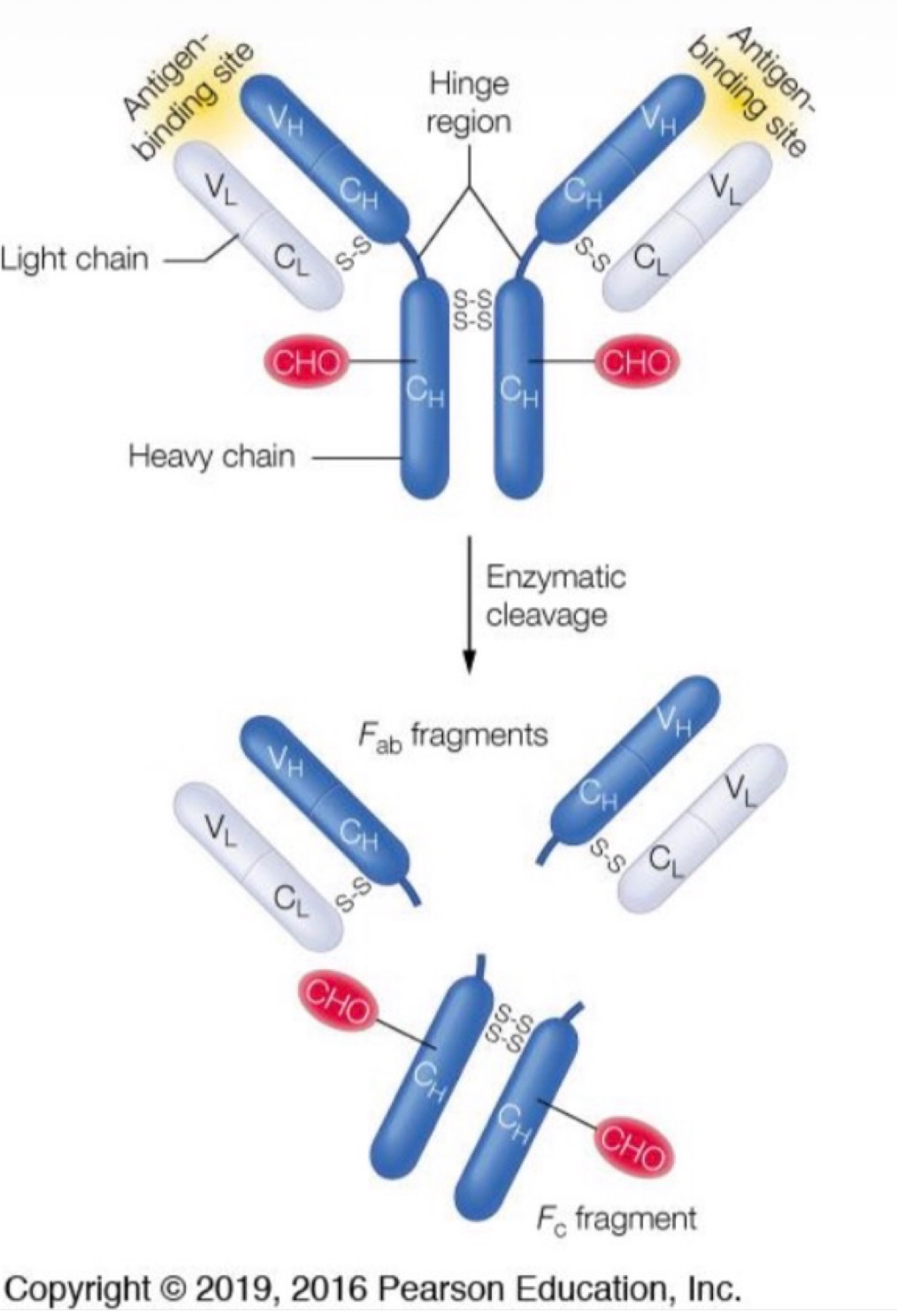
Explain this image
Rather than breaking down the Ig by heavy chains, light chains, and variable regions, the Ig can also be cleaved at its hinged regions producing two F(ab) fragments and one Fc fragment
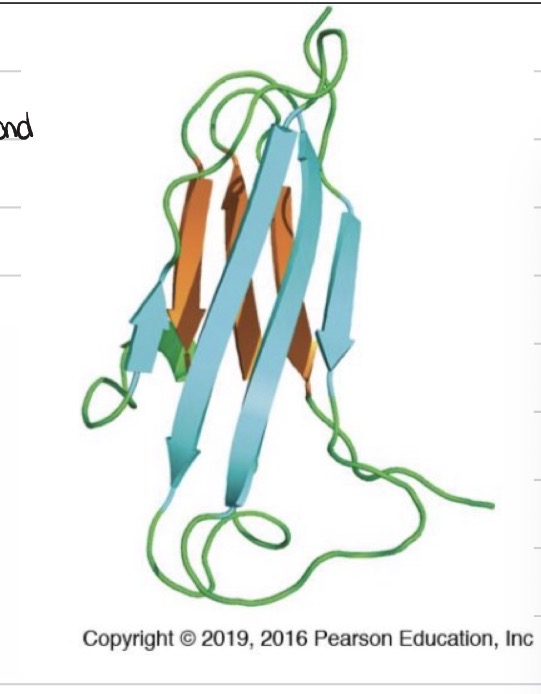
The Ig Domain
Consists of two anti-parallel beta sheets, linked together by a sulphur bond
The coiled portions are the “variable” bits of the region
Out of the five classes of immunoglobins, which one is the most important?
the igG molecules
The most abundant circulating antibodies
Can transverse blood vessel walls
can cross the placenta
Triggers the complement system
Antibody – Antigen Interactions
These loops on the on the variable regions of the light and heavy chains contain the Ig domain
◦Two anti-parallel beta sheets mixed with hypervariable loops
Most of the Ig-Antigen interactions happens with these loops
◦They are the complementarity determining regions (CDRs)
In regards to the complementarity determining regions (CDRs), what do we mean by complementarity?
these regions are structurally and chemically complementary to a specific antigen epitope, allowing a precise, lock-and-key, or "induced fit" binding, similar to how a key fits a lock
Shape and Charge Complementarity
Not only do Ig molecules match the shape of the antigens, locking them into place, they also have matching charges (- with +, non-polar with non-polar, etc.)
Again, this is due to these hypervariable loops having many different R groups which can accommodate different sizes and charges of antigens.
Take away from the figure of Hepatitis B antigen interacting with the CDR region of a complementary Ig:
Note, again, it is the side chains of the amino acids in the loop regions that do the bulk of the interactions with the antigen
How do novel antibodies get generated?
This occurs through two separate mechanisms:
1.The nucleic acid sequences programming the amino acids in the heavy and light chain variable regions are shuffled, creating new patterns for interaction with antigens
2.The sequences of the CDR loops are prone to mutate at a higher rate
Note: Point mutations in these sequences are not harmful, since it may not affect the structure of the Ig domain itself, just the loop features
What does the respiratory system rely on?
Relies on two separate oxygen transport proteins
◦Myoglobin and hemoglobin
◦Without these, the amount of oxygen from respiration would be ~100 times lower
◦Not only do these molecules need to attach oxygen, they also need to release it
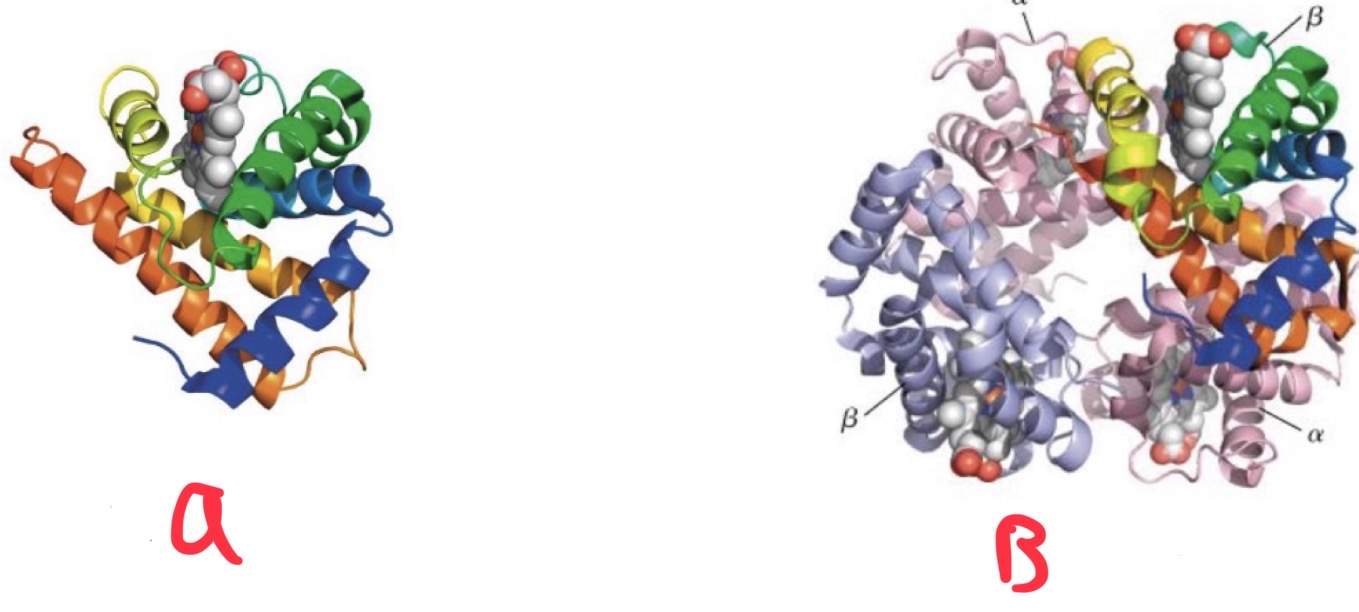
Hemoglobin
◦Present in red blood cells, tetrameric protein
◦Does not bind as readily to oxygen
Changes its shape when bound. More O2 bound, less likely to bind the next molecule
Myoglobin
◦Present in tissues
◦Binds oxygen more readily, transports it to mitochondria
Figure 7.12 Role of the globins in oxygen transport and storage.
Hemoglobin transports O2 from the lungs or gills to the respiring tissues, where it is used for aerobic metabolism in the mitochondria
Inside cells, dissolved O2 diffuses freely or is bound to myoglobin which aids transport of O2 to the mitochondria. Myoglobin can also store O2 for later use (as in deep-diving mammals)
CO2 produced by oxidative processes in the tissues is carried back to the lungs or gills by hemoglobin, or in plasma as HCO3- and releases
Where does O2 bind?
The globins contain bound prosthetic groups (protoporphyrin IX), which itself contains Fe(II)
O2 bids to this Fe(II) molecule
prosthetic groups
A prosthetic group is a non-protein, organic or inorganic molecule tightly, often covalently, bound to a protein (specifically an enzyme) that is essential for its biological activity
A heme group is an essential iron-containing prosthetic group
Histidine
an essential amino acid necessary for protein synthesis, tissue repair, and the production of histamine,
How does O2 bind to the heme group?
On each side of the heme group there are histidine molecules from the globins which stabilize the structure
proximal histidine
is in direct contact with the heme group
The distal histidine
◦is in contact, via hydrogen bonds, with the oxygen molecule when there is one in place
Dynamics of globin binding
Myoglobin binds to oxygen at relatively low concentrations of oxygen
◦Only requires a partial pressure of 3-4 mmHg
This is helpful, as the partial pressure in tissues is around 30 mmHg
This is also considerably lower than hemoglobin
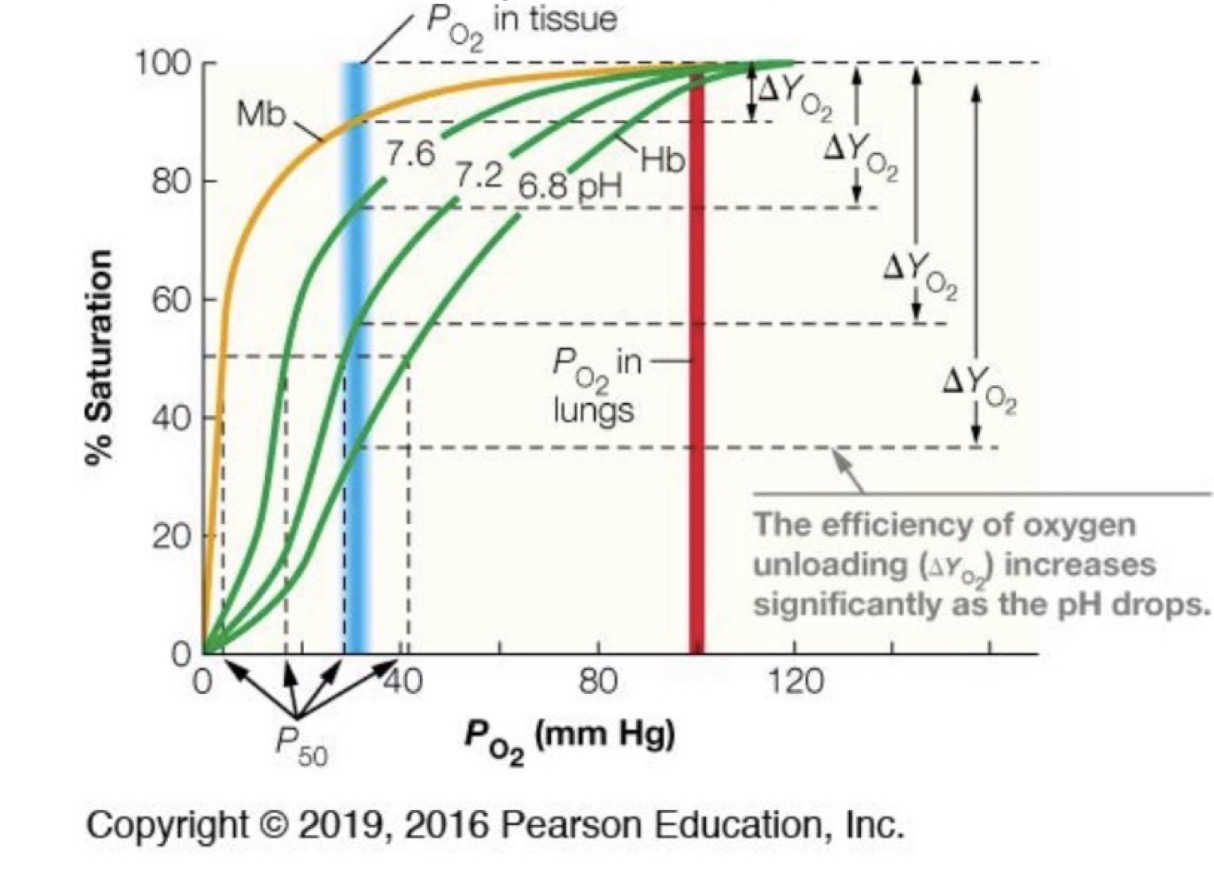
So, if myoglobin is so effective, why hemoglobin?
Both hemoglobin and myoglobin bind oxygen very readily at the partial pressures found in the lungs (100 mm Hg)
At lower pressures, hemoglobin’s binding efficacy decreases in a sigmoidal way
At pressures like those in tissue (30 mmHg), hemoglobin loses its affinity for oxygen more readily
Why does at pressures like those in tissue (30 mmHg), hemoglobin loses its affinity for oxygen more readily?
◦There is a conformational change that occurs with the hemoglobin molecule. The more oxygen is bound, the more likely the next site is able to uptake another oxygen. The reverse is also true.
This is not possible with myoglobin, with only one binding site
Why is hemoglobin O2 transporter better if myoglobin has high O2 affinity?
If the transport protein had a high O2binding affinity, it would achieve 100% O2 binding in the lungs but would release very little O2 in the capillaries
to achieve optimum O2 delivery to tissues, an ideal oxygen transport protein would be nearly saturated at 100 mm Hg, deliver sufficient O2 to tissues at rest to support basal metabolism, yet maintain a significant O2 reserve for periods of high demand
How else does hemoglobin give up its oxygen?
Through the manipulation of allosteric sides, which can be (negatively) modified by
◦pH changes (acidity)
◦Carbon Dioxide
◦Cl- and
◦2,3-bisphosphoglycerate (2,3-BPG)
The Bohr effect
Ph changes: Increases in acidity, whether from increased presence of CO2 or vigorous activity producing lactic acid, promote extra release of oxygen
◦Named the Bohr effect
◦In lower pH solutions, the H+ ions help stabilize a different conformational state of hemoglobin (T-state), which allows it to more readily release its oxygen molecules
Hemoglobin and CO2 Transport
CO2 has the effect of lowering hemoglobin’s affinity to oxygen
First, it does this by lowering the pH
But, it also directly attaches to the hemoglobin, allowing for its release
carbamates
CO2 does not attach to hemoglobin at the heme site, rather, it attaches to the n-terminal regions of the dimers
◦These molecules are called carbamates
once the RBC reaches the lungs, in an area with high levels of oxygen, the hemoglobin molecule changes its shape to that favouring oxygen, which allows for carbon dioxide release

What is this reaction?
This carbamation reaction allows hemoglobin to aid in the transport of O2 from tissues to lungs or gills, and the protons released by carbamate formation contribute to the Bohr effect.

Label this
(a) Transport protein efficient in binding but inefficient in unloading (hyperbolic-binding curve).
(b) Transport protein efficient in unloading but inefficient in binding (hyperbolic-binding curve).
(c) Transport protein efficient in both binding and unloading because it can switch between higher- and lower-affinity states (sigmoidal-binding curve).
(d) Switch from lower- to higher-affinity states yields the sigmoidal curve.