Chem 215-1: Quiz 2
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What is an enantiomer?
Configurational isomers that are mirror images of each other
What are diastereomers?
Configurational isomers that are not mirror images of each other
What is chirality?
Something that can’t be superimposable with its mirror image
What is one way to determine the chirality of a molecule?
A molecule that has a plane of symmetry must be achiral
What is a chiral center?
A sterocenter, asymmetric atom; is when it and its mirror image are nonsuperimposible, mirror plane goes down center of atom
Can other elements also be chiral centers?
Yes, nitrogen atoms can be sp3 hybridized and can have tetrahedral geometry; it typically doesn’t though, but can
What happens when a molecule has exactly one chiral center?
Molecule must be chiral
What happens when a molecule has two chiral centers?
Usually chiral, but can be a chiral
What does meso mean?
A molecule is meso if it contains at least two chiral centers but has a plane of symmetry that makes it achiral overall
How to define stereochemical configurations?
Each tetrahedral stereocenter has two possible configurations
One is R and one is S
One configuration is the opposite of the other
What is the Cahn-Ingold-Prelog system?
Assign priorities 1 through 4 to the substituents (highest is 1)
Orient the molecule properly so the lowest priority points away
Observe the arrangement of substituents 1 through 3 and assign the configuration as R or S
If priority 1 through 3 is arranged clockwise, R
Counterclockwise, S
How to assign relative priorities?
Assign substituents based on the atomic number at the points of attachment (higher = 1)
Assign substituents priorities based on the sets of three atoms located one bond away from the points of attachment
Repeat step 2 but go out another bond
How to determine priority with double or triple bonds?
An atom that is doubly bonded to another atoms is treated as having two single bonds to the atom (so list same atom twice)
An atom that is triply bonded to another atom is treated as having three single bonds to the atom (list same atom three times)
What do you do when the fourth priority substituent does not point away?
You swap the 1st and 4th priority so the lowest one points away, whatever is the opposite of the current assignment (R or S) is the correct answer
What happens when a molecular and its mirror image rapidly interconvert?
The molecule is effectively achiral
How to determine if a cyclohexane is a chiral?
Don’t use the chair conformation, draw it flat or Hayworth with wedges and dashes, then use this to determine chirality
Why is Nitrogen often achiral?
Nitrogen rapidly interconverts, the intermediate leaves an unhybridized p orbital is achiral (sp2)
What can diastereomers come from?
Can arise from double bond geometry or compounds with at least 2 chiral centers
How to determine Z/E system for double bond configurations?
Z (cis), E (trans); Divide the double bond in half define priority on either side of the line; if higher priority groups are on the same side, Z
What is the maximum number of configurational isomers that can exist for a molecule?
With n chiral centers, 2n
How to draw all stereoisomers of a given structure?
Identify all chiral centers
Find the configuration at each chiral center
R or S (if you have two chiral centers and switch their positions and configurations ex (1R, 3S) to (1R, 3S) for different molecules, it’s an enantiomer)
Flip at each and check for redundancies
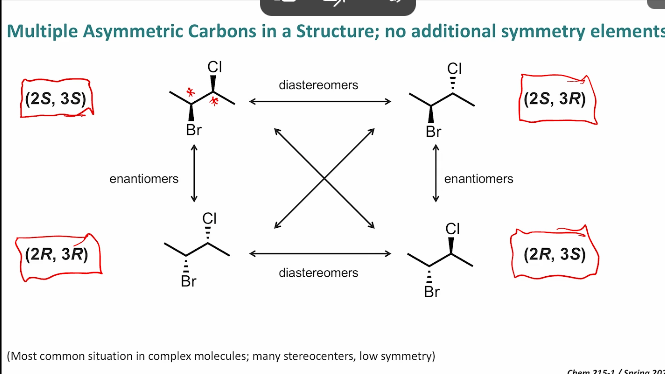
What are physical and chemical properties of isomers?
Constitutional isomers have different physical and chemical properties (ex boiling point)
Why do we are about enantiomers?
Both are identical in terms of physical properties; in achiral environment, have the same physical and chemical properties, in chiral this isn’t true
What are the physical and chemical properties of diastereomers?
Have different chemical properties depending on molecule and properties differences can be large or small
How to use chromatography to separate enantiomers?
Have a chiral stationary phase that will associate with only one (R or S) of the enantiomers
One that sticks more moves more slowly
How can you form salts to separate enantiomers?
Have a mixture of a simple chiral molecule and get a single enantiomers → S combines with either R or S of OG molecule
Resulting salts will be diastereomers of each other
How do chiral molecules interact with plane-polarized light?
Polarized light passing through a chiral medium is going to rotate with the light
Dextrorotatory vs levorotatory?
Chiral compounds that rotate light clockwise (+) direction are dextrorotary; don’t need the actual word
What is a racemic mixture?
Contains equal amounts of the (+) and (-) enantiomers of a chiral molecule
optically inactive
Excess percentage that favors one enantiomer is the enantiomeric excess
specific rotation of mixture = %ee (specific rotation of pure enantiomer)/100
What is a proton transfer reaction?
Bronsted-Lowry acid-base reaction, accept and donate protons
Changes occur simultaneously (concerted)
Curved arrow notation?
Double barbed means that 2 valence electrons are moving