Chemistry - Final Exam
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
What does n, m1, and l mean?
n is the energy level, l is the shape of orbital and subshell, m1 is the magnetic quantum number.
What is Boyle’s Law?
Pressure Is Inversely Proportional to Volume
What is Charles’ Law?
Volume is directly proportional to temperature,
What is Aufbau’s Principle?
Electrons fill up the lowest energy level first.
What is Hund’s Rule?
Electrons occupy orbitals of equal energy singly before pairing up.

What kind of orbital is this?
S Orbital
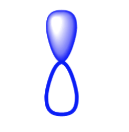
What kind of orbital is this?
P Orbital

What kind of orbital is this?
D Orbital

What kind of orbital is this?
F Orbital
A high-speed electron ejected from a nucleus during
radioactive decay is called a
Beta Particle
What is an alkane?
Saturated hydrocarbons with single bonds.
What is an alkene?
Unsaturated hydrocarbons with at least one double bond.
What is an alkyne?
Unsaturated hydrocarbons with at least one triple bond.
What are aromatic hydrocarbons?
Compounds that contain benzene rings or similar structures.
1 carbon means that the prefix is
Meth
2 carbons means that the prefix is
Eth
3 carbons means that the prefix is
Prop
4 carbons means that the prefix is
But
5 carbons means that the prefix is
Pent
6 carbons means that the prefix is
Hex
7 carbons means that the prefix is
Hept
8 carbons means that the prefix is
Oct
9 carbons means that the prefix is
Non
10 carbons means that the prefix is
Dec
For compounds with two or more double bonds, the suffixes are
diene, triene, etc.
What does (E) and (Z) mean for configuration
E means that they’re on opposite sides of the double bond and Z means that they’re on the same side of the double bond.
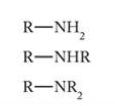
What is this functional group?
Amine
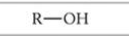
What is this functional group?
Alcohol
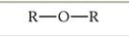
What is this functional group?
Ether
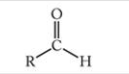
What is this functional group?
Aldehyde
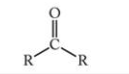
What is this functional group?
Ketone
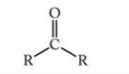
What is this functional group?
Carboxylic Acid
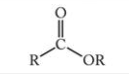
What is this functional group?
Ester
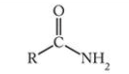
What is this functional group?
Amide
What makes something paramagnetic or diamagnetic?
When there are only paired electrons, it is diamagnetic, and when it there is at least one unpaired electron it is paramagnetic.
What is the case for formal charges?
The sum of formal charge must be equal to the total charge of the ion.
What is the formula for Formal Charge (FC)?
FC = Valence Electrons - (Lone Pair Electrons - Bonding Electrons/2)
What is a single bond in terms of sigma and pi bonds?
one sigma bond.
What is a double bond in terms of sigma and pi bonds?
one sigma bond and one pi bond.
What is a triple bond in terms of sigma and pi bonds?
one sigma bond and two pi bonds.
How do you calculate the steric number?
SN = Number of lone pairs + bonded atoms
A single bond has a bond order of
1
A double bond has a bond order of
2
A triple bond has a bond order of
3
What is the molecular geometry of a compound where SN = 2 LP = 0 and Bonds = 2
Linear
What is the molecular geometry of a compound where SN = 3 LP = 0 and Bonds = 3
Trigonal Planar
What is the molecular geometry of a compound where SN = 3 LP = 1 and Bonds = 2
Bent
What is the molecular geometry of a compound where SN = 4 LP = 0 and Bonds = 4
Tetrahedral
What is the molecular geometry of a compound where SN = 4 LP = 1 and Bonds = 3
Trigonal pyramidal
What is the molecular geometry of a compound where SN = 4 LP = 2 and Bonds = 2
Bent
What is the molecular geometry of a compound where SN = 5 LP = 0 and Bonds = 5
Trigonal Bypyramidal
What is the molecular geometry of a compound where SN = 5 LP = 1 and Bonds = 4
Seesaw
What is the molecular geometry of a compound where SN = 5 LP = 2 and Bonds = 3
T shaped
What is the molecular geometry of a compound where SN = 5 LP = 3 and Bonds = 2
Linear
What is the molecular geometry of a compound where SN = 6 LP = 0 and Bonds = 6
Octahedral
What is the molecular geometry of a compound where SN = 6 LP = 1 and Bonds = 5
Square Pyramidal
What is the molecular geometry of a compound where SN = 6 LP = 2 and Bonds = 4
Square Planar
What is the bond angle for linear?
180
What is the bond angle for trigonal planar?
120
What is the bond angle for tetrahedral?
109.5
What is the bond angle for trigonal pyramidal?
107
What is the bond angle for bent?
104.6
What is the bond angle for trigonal bypyramidal?
90/120
What is the bond angle for octahedral?
90
If SN = 2 then the electron geometry is
Linear
If SN = 3, then the electron geometry is
Trigonal Planar
If SN = 4, then the electron geometry is
Tetrahedral
If SN = 5 then the electron geometry is
Trigonal Bypyramidal
If SN = 6, then the electron geometry is
Octahedral
List the intermoleular forces from weakest to strongest.
London disperson, dipole dipole, hydrogen, ion dipole
What is a dipole dipole IMF?
IMF’s between polar molecules
What is hydrogen bonding IMF’s ?
When Hydrogen is bonded to N, O, or F
What are ion dipole IMF’s?
IMF’s between an ion and a polar molecule.
What is the BBP trick for memorizing Octet Violations?
Berrylium, Boron, and Period 3 violate the octet rule.
What are some ways that determine that a molecule is nonpolar?
Any diatomic element, any hydrocarbon, and if the dipole moments cncel out.
How do you do the net dipole moments?
Draw an arrow from the partial positive part of the bond to the central atom, if all of these end up canceling out, then the molecule is nonpolar and polar if they do not.
How do bond angles get reduced?
2.5 degrees per each lone pair.
If SN = 2, the hybridization is
sp
If SN = 3, the hybridization is
sp2
If SN = 4, the hybridization is
sp3
If SN = 5, the hybridization is
sp3d
If SN = 6 the hybridization is
sp3d2
For resonance struutures, what makes a resonance structure the best?
Fewer formal charges, negative formal charge on the more electronegative atom, , full octets are preferred, and minimized charge separation
What is specific heat?
The amount of heat energy required to raise the temperature of one unit of mass (typically 1 kg or 1 gram) of a substance by one degree Celsius
What is heat capacity?
The amount of heat required to raise the temperature of an object by 1 degree Celsius.
What are the periodic trends for atomic radii?
Atomic Radii increases going down a group and decreases going across a period.
What are the periodic trends for Ionization Energy?
Ionization Energy decreases going DOWN a group and increases going across a period.
What are the periodic trends for Electronegativity?
EN decreases going down a group and increases going across a period.
What are the periodic trends for Electron Affinity?
EA values decreases going DOWN a group and increase going LEFT TO RIGHT of a period. Note that the more negative the number is, the greater the electron affinity.
What are the periodic trends for Ionic Radius?
Increase going DOWN a group and decrease across a period. Cations result in lower atomic radii while Anions result in greater atomic radii.
What is Alpha Decay?
An alpha particle(4/2 He) is emitted from the nucleus. Two neutrons and two protons are removed.
What is Beta-Minus Decay?
A neutron is converted into a proton, so an electron is emitted (0/-1 e)
What is Beta Plus Decay?
A proton turns into a neutron and emits a positron
What is gamma radiation?
High-energy electromagnetic radiation emitted from the nucleus.
What should the n/p ratio be for smaller atoms?
1:1 ratio
What should the n/p ratio be for larger atoms?
1:5
If a atom has too many neutrons, it will likely undergo
Beta Minus Decay
If an atom has too many protons, it will likely undergo
Beta Plus Decay
If an atom has a large nucleus, it will likely undergo
Alpha Decay
At what element does the belt of stability end?
It ends at Bismuth (Bi), which has 83 protons. If an element has greater than 82 protons, it is unstable