rotational spectroscopy
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
for rigid rotor - what is the general spectrum shape (absorption vs frequency)
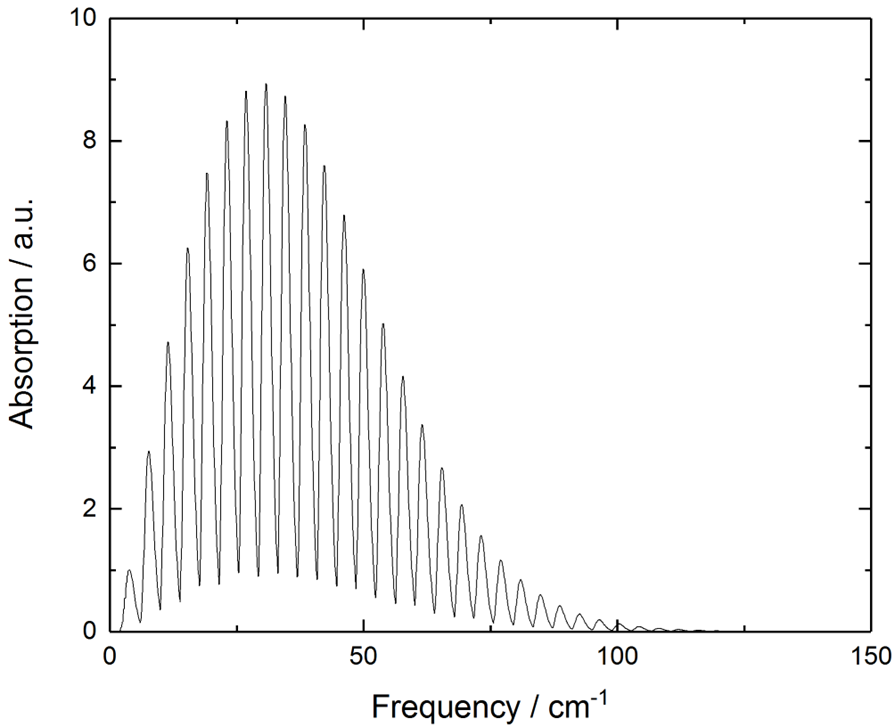
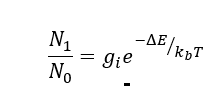
how do you find Jmax from this?
what is Jmax? what do you need to remember when calculating it
differentiate to find maximum
Jmax is the Jth level that gives max absorption
round down to an integer value (as it is a quantum number)

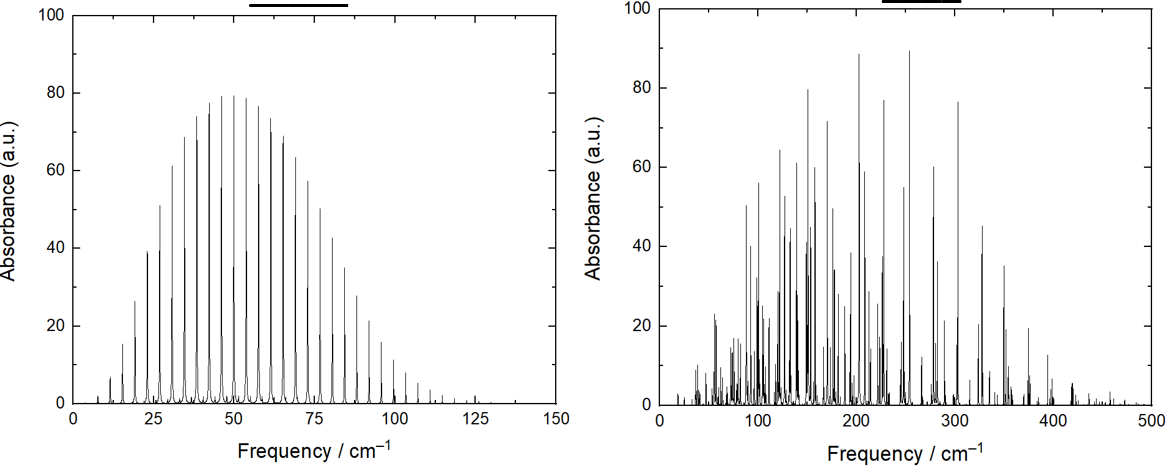
explain why these rotational spectra are different (CO2 vs H2O)
CO2 - moment of inertia is the same in all 3 directions
H2O - different moments of inertia for x,y,z means different rotational spectra
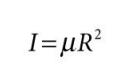
put in terms of m for general formula
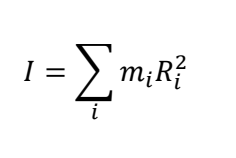
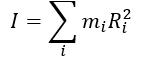
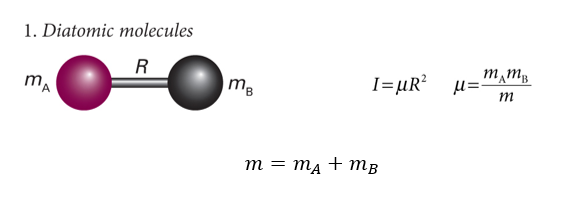
show diagram of diatomic molecule
show mA, mB and R
what is m? what is I?
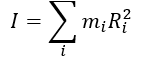
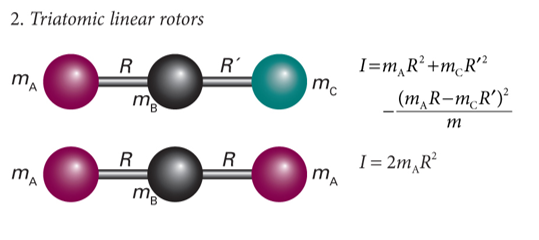
show diagram of triatomic linear rotors
label mA, mB, mC and R
what is I? what is the specific selection rule?
ΔJ = ±1
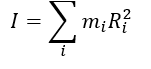
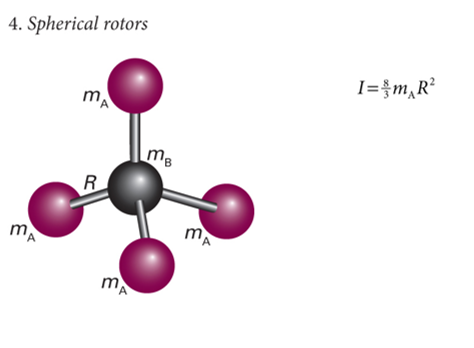
show diagram of spherical rotors with 4 atoms
label mA, mB, mC etc and R
what is I?
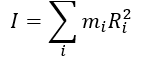
show diagram of spherical rotors with 6 atoms
label mA, mB, mC etc and R
what is I?
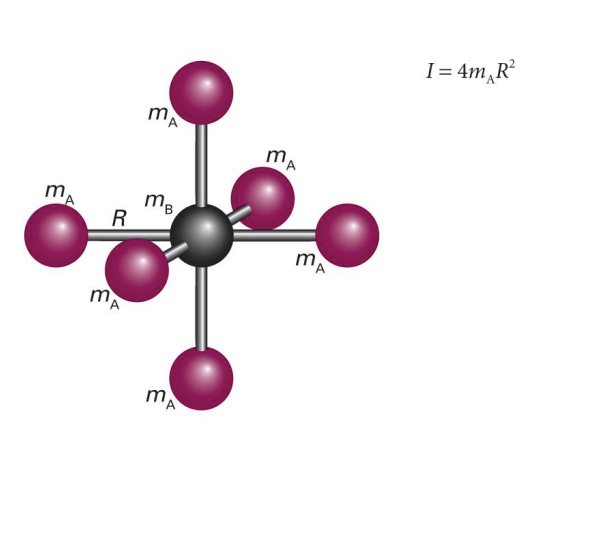
what is the specific selection rule for spherical rotational molecules?
J=0 transitions also allowed

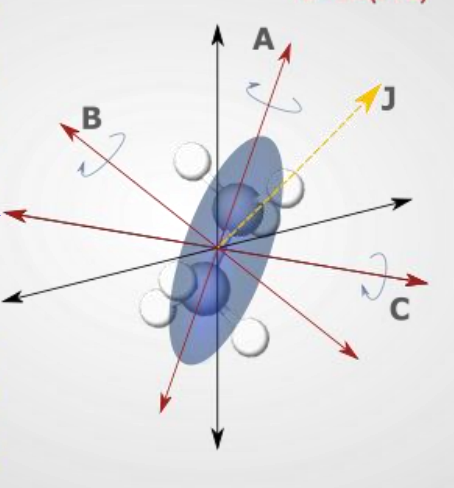
what is a symmetric rotor?
taken spherical and changes mass of one atom or removed one
in specific selection rules - what is K?
the projection of J onto the principal axis of the molecule
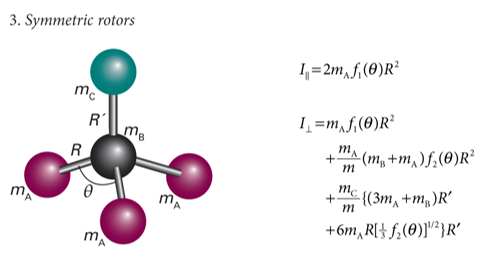
show symmetric rotor with 5 atoms
label mA, mB, mC etc and R and any angles of importance
what is I?
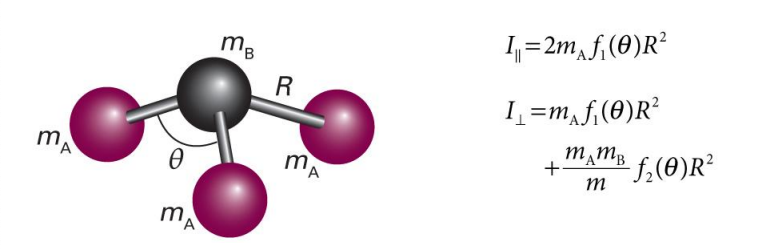
show symmetric rotor with 4 atoms
label mA, mB, mC etc and R and any angles of importance
what is I?
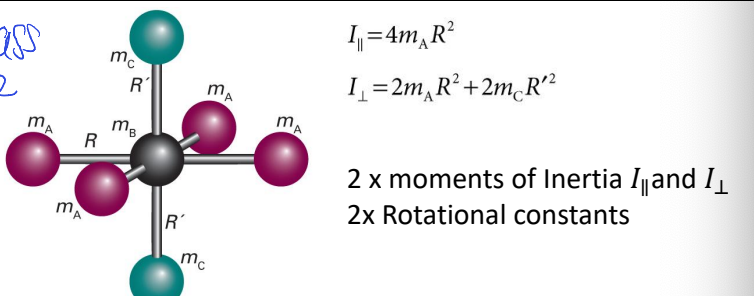
show symmetric rotor with 7 atoms
label mA, mB, mC etc and R and any angles of importance
what is I?
what are the rotational specific selection rules for symmetric rotors?
ΔJ = ±1 and ΔK = 0
new rotational level which is K
what are the 3 vibrational specific selection rules for symmetric rotors?
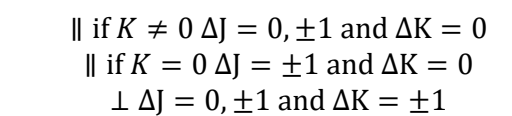

how are transitions different for diatomic molecules vs symmetric rotors?
diatomic, only in K = 0
symmetric has multiple sets of K which each have their own sets of J
they have transitions up the ladders but not between (ΔK=0 in the specific selection rules)
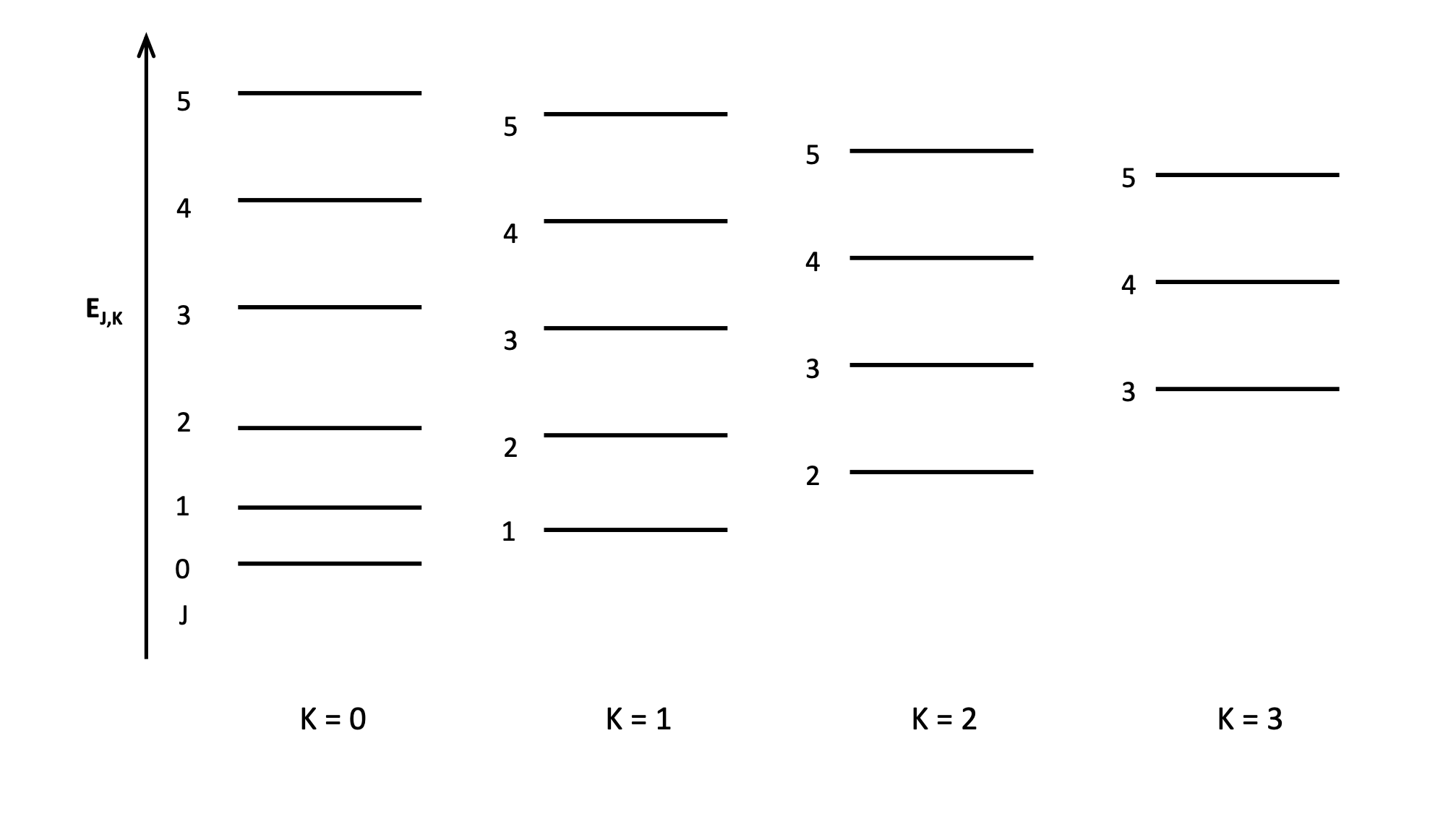
explain how symmetric rotors having transitions in multiple sets of K affects spectra?
spectra are much more complex
series of transitions all stuck on top of each other but randomly offset
show linear molecules diagram
what are the quantum numbers?
what is the degeneracy?
what is the energy equation?
J and m
2J+1
E=BJ(J+1)
where is the mass centred?
all mass around one axis
show spherical molecules diagram
what are the quantum numbers?
what is the energy equation?
what is the degeneracy?
E=BJ(J+1)
J,k,m quantum numbers
degeneracy 2J+1
what are the rotational constants?
A=B=C
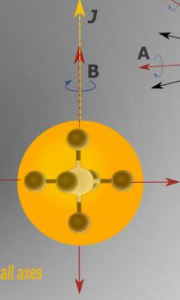
where is the mass centred?
mass distributed equally around all axes
show asymmetric molecule diagram
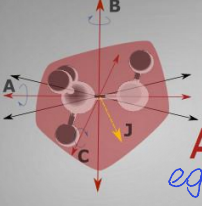
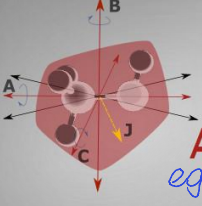
what are the quantum numbers?
what is the energy equation?
what is the degeneracy?
J, Ka, Kc, m
no standard energy equation
degeneracy is 2J+1
what are the two options for symmetric molecule shapes?
oblate (wide and fat)
prolate (long and thin)
show oblate molecule diagram
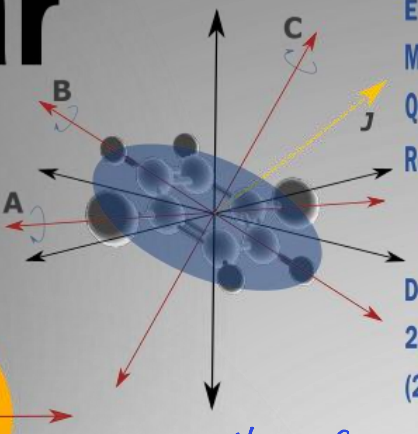
what are the rotational constants?
A=B>C
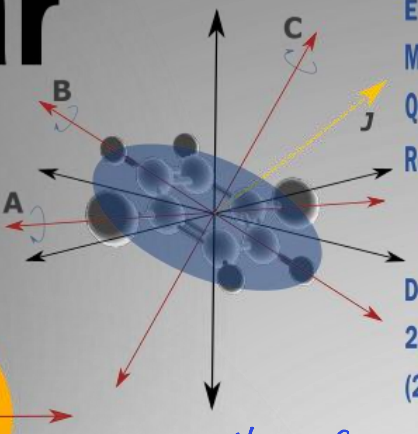
what is the energy equation?
what are the quantum numbers?
quantum numbers J,k,m
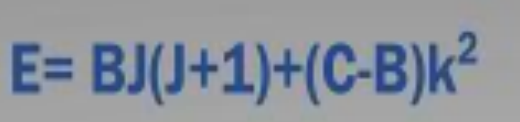
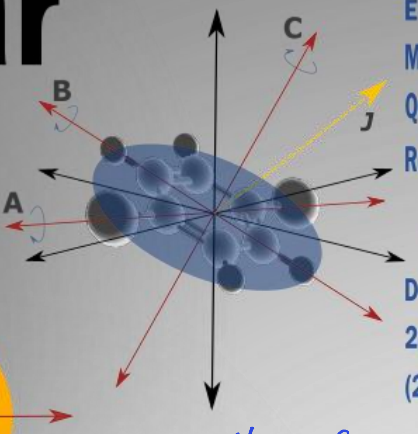
what is the degeneracy?2

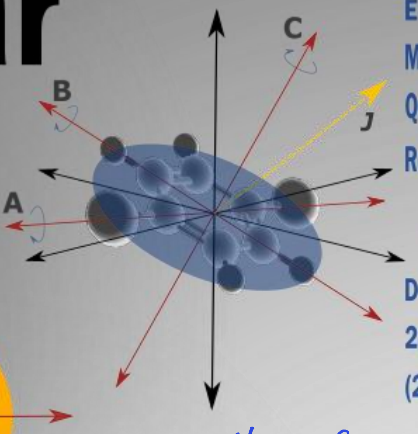
where is the mass centred?
most mass about a plane
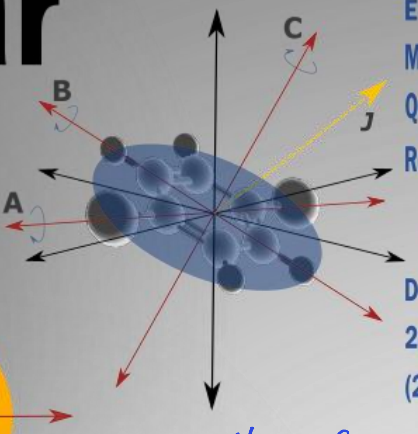
show prolate molecule diagram
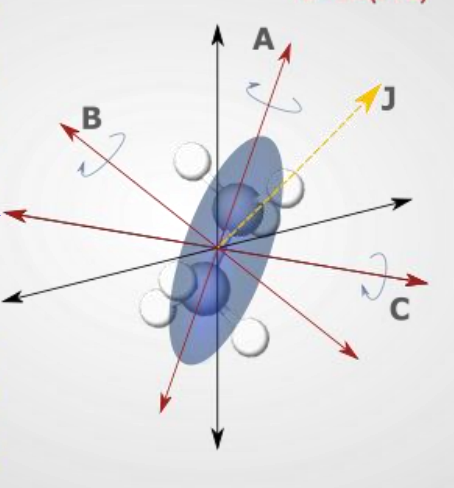
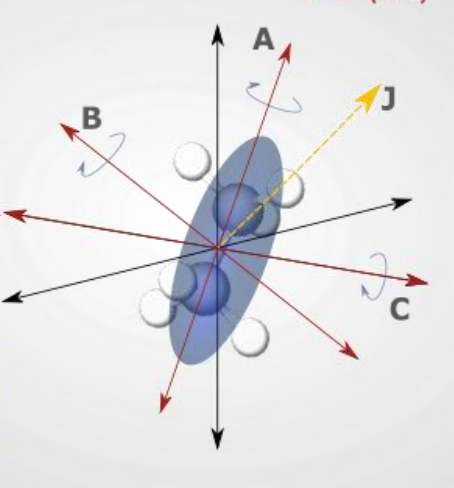
what is the energy equation?
what are the quantum numbers?
quantum numbers J,k,m

what are the rotational constants?
A>B=C
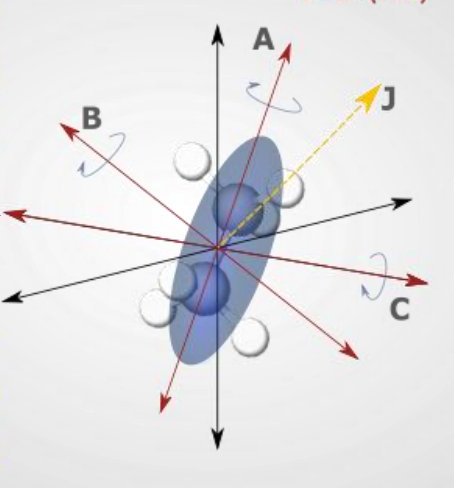
what is the degeneracy?

where is the mass centred?
most mass about one axis
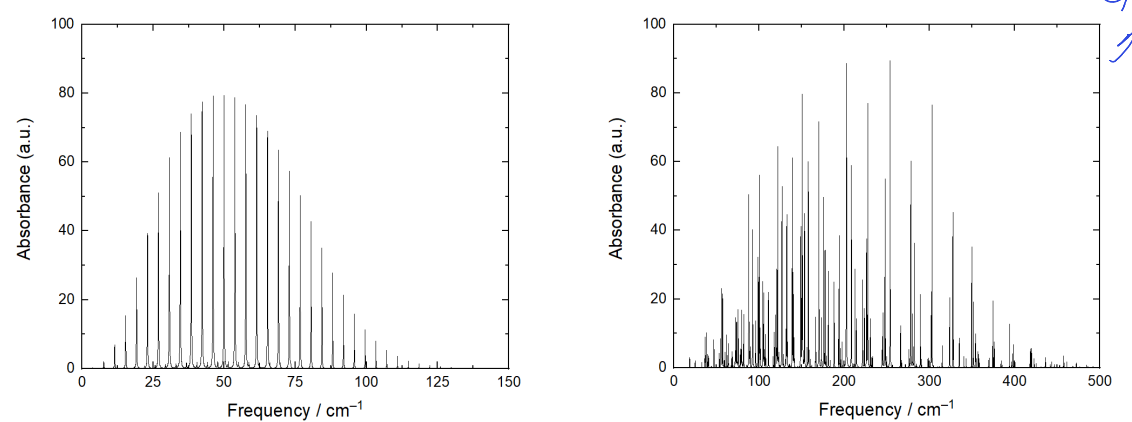
why is spacing clear for CO but very different for water?
CO has one rotational constant which is small
water has very different rotational constants which spread spectra out
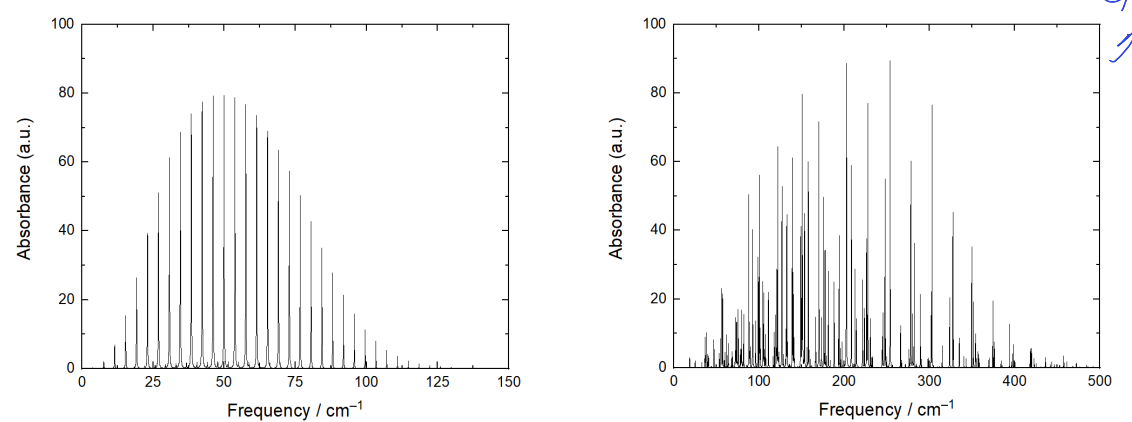
why is overall shape of water and CO spectra similar?
overall shape is governed by population of energy levels
explain born Oppenheimer approximation
nuclei are an order of magnitude heavier than electrons
nuclei move, electrons move instantaneously
can be treated separately
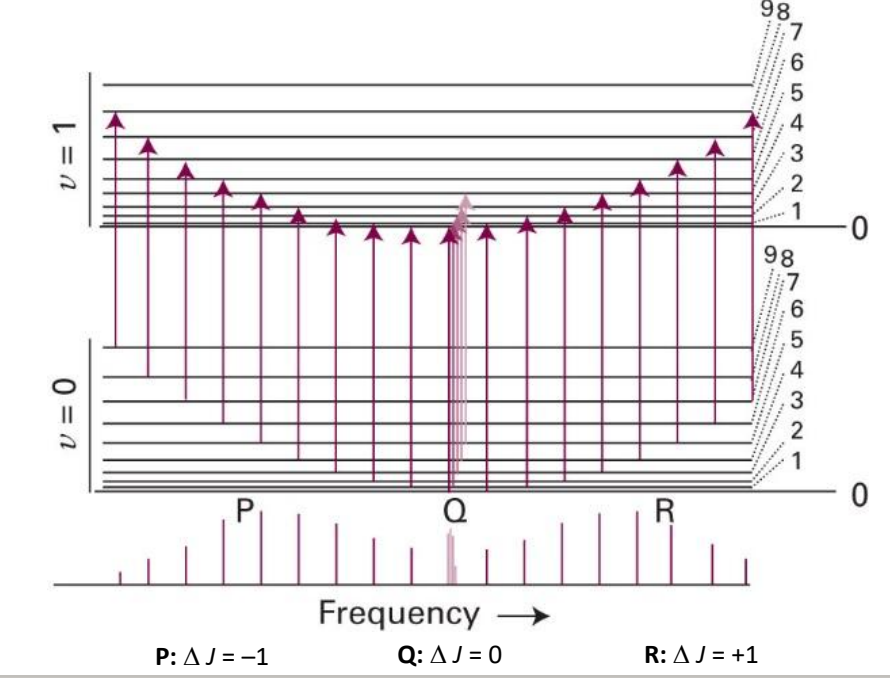
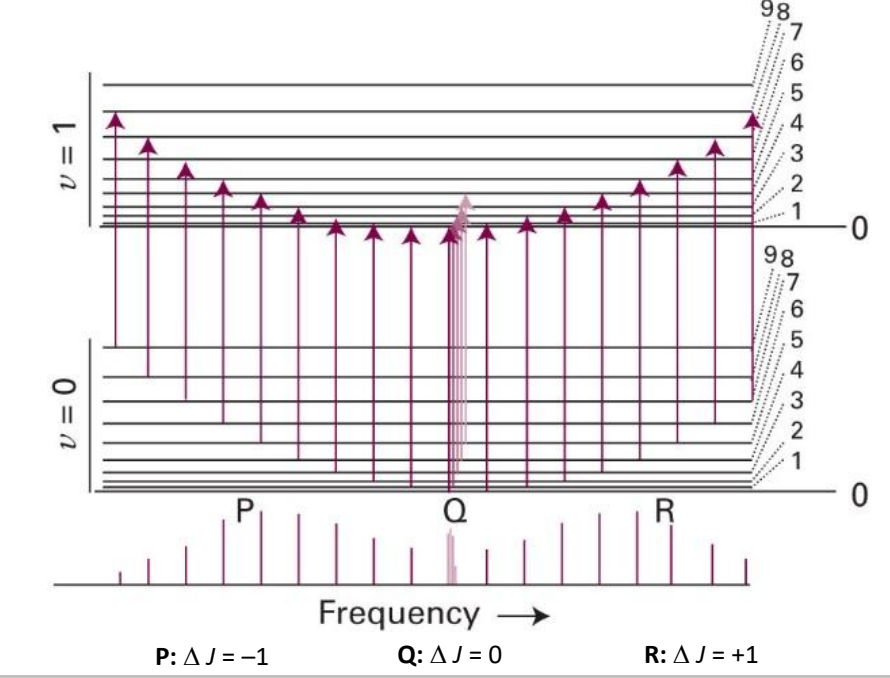
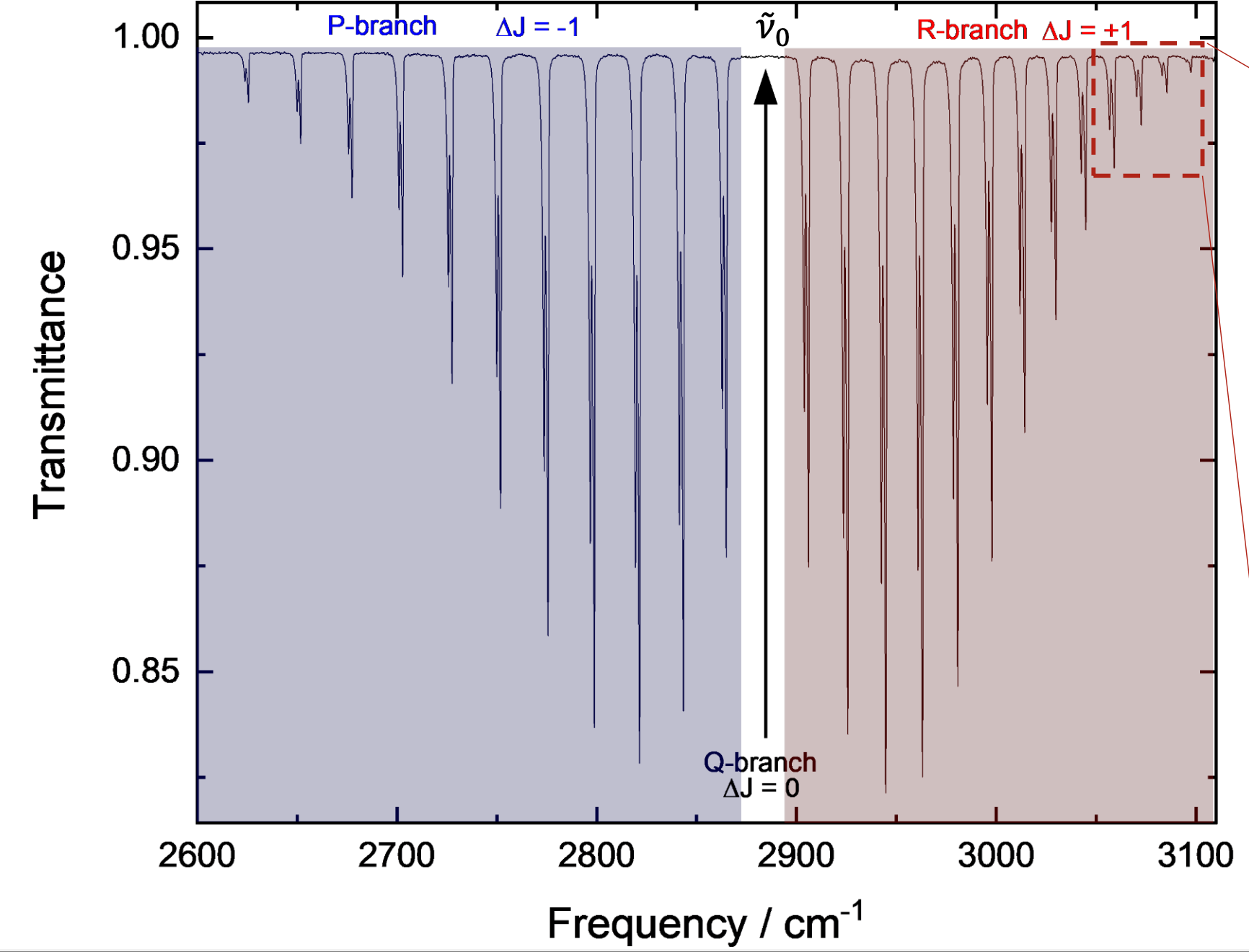
what transitions are occurring in a ro-vibrational spectra?
transition from v=0 to v=1 at same time as change in rotational number
what are P, Q and R branches?
P: ΔJ=-1
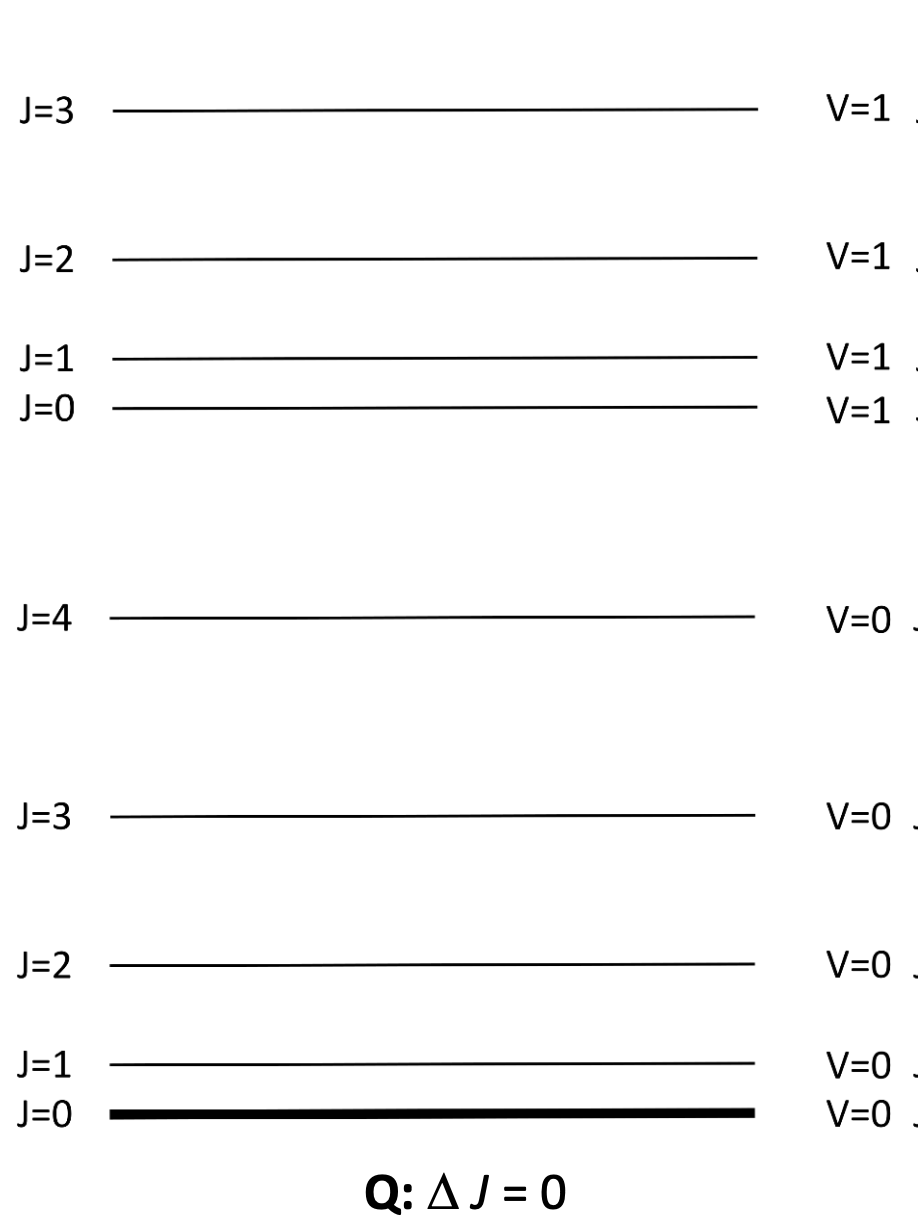
Q: ΔJ=0
R: ΔJ=+1
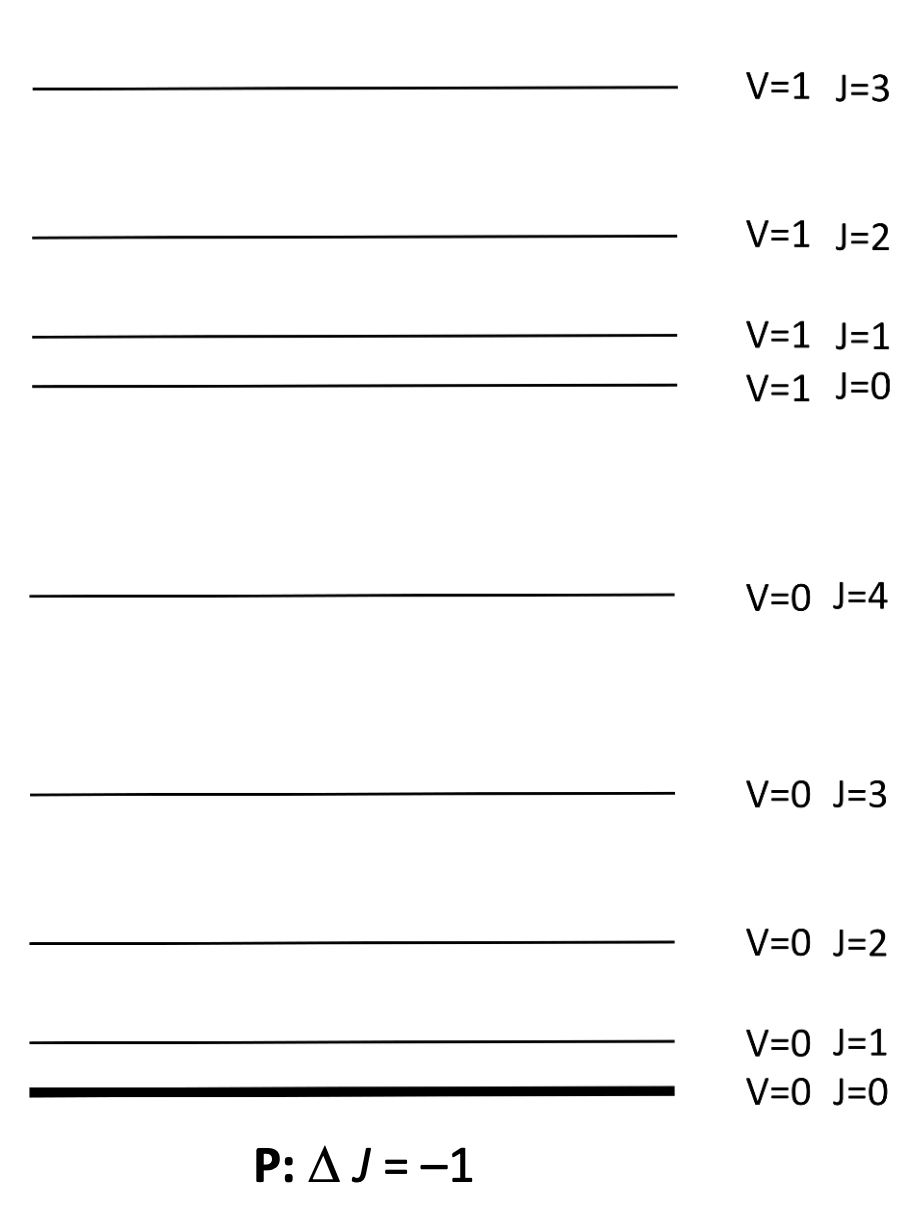
show ro vibrational transition
where go from vibrational ground state to excited state, and J also changes by -1
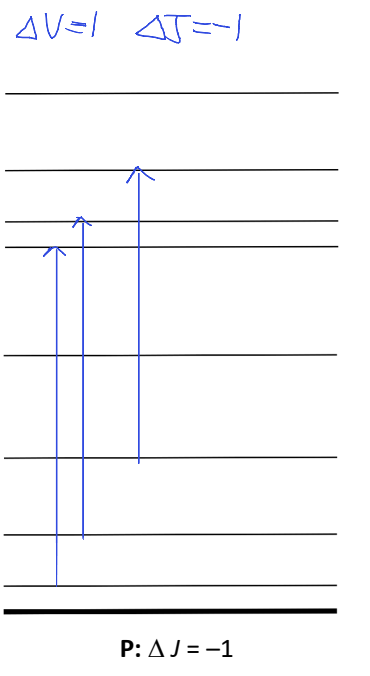
show ro vibrational transition
where go from vibrational ground state to excited state, and J also changes by 0
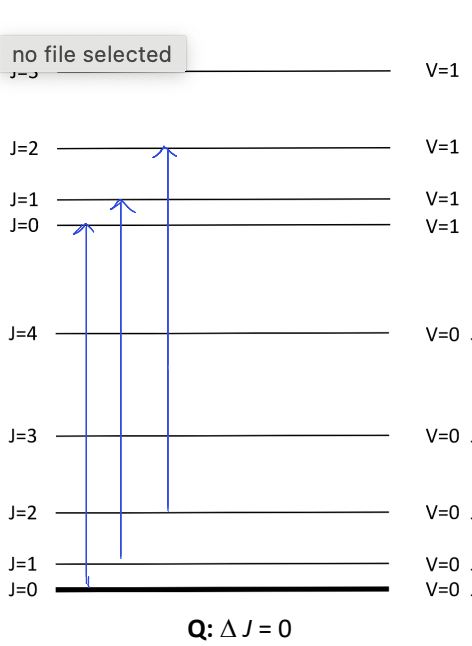

label P, Q and R transitions
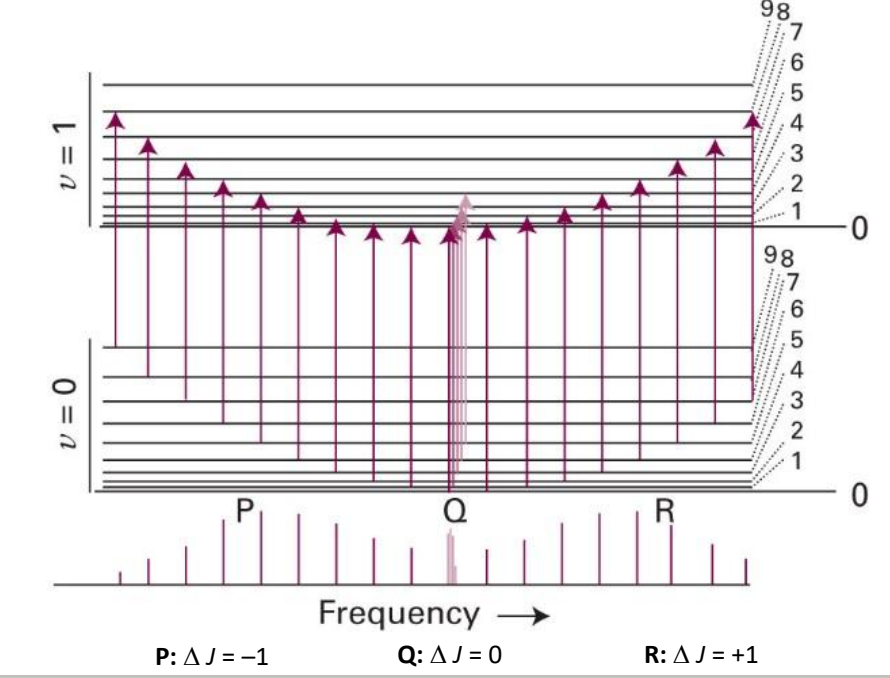
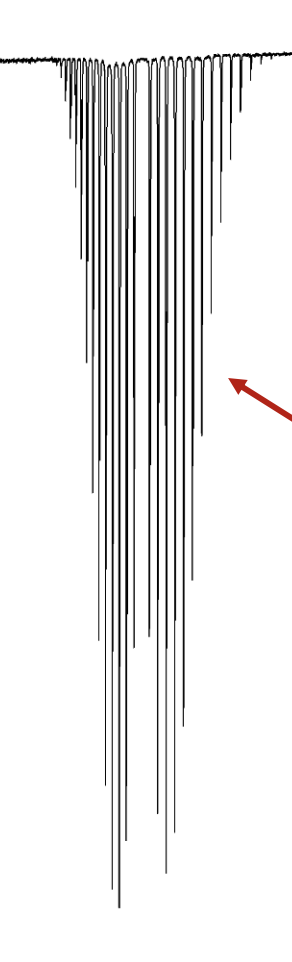
explain shape
intensity of lines is governed by the population of each EL so it is a Boltzmann distribution
what governs the position of lines?
whether or not a transition is allowed
for HCl, the big peak is the stretch
why is there a series of lines on either side?
molecule is rotating at the same time
P branch and R branch
combination of both effects
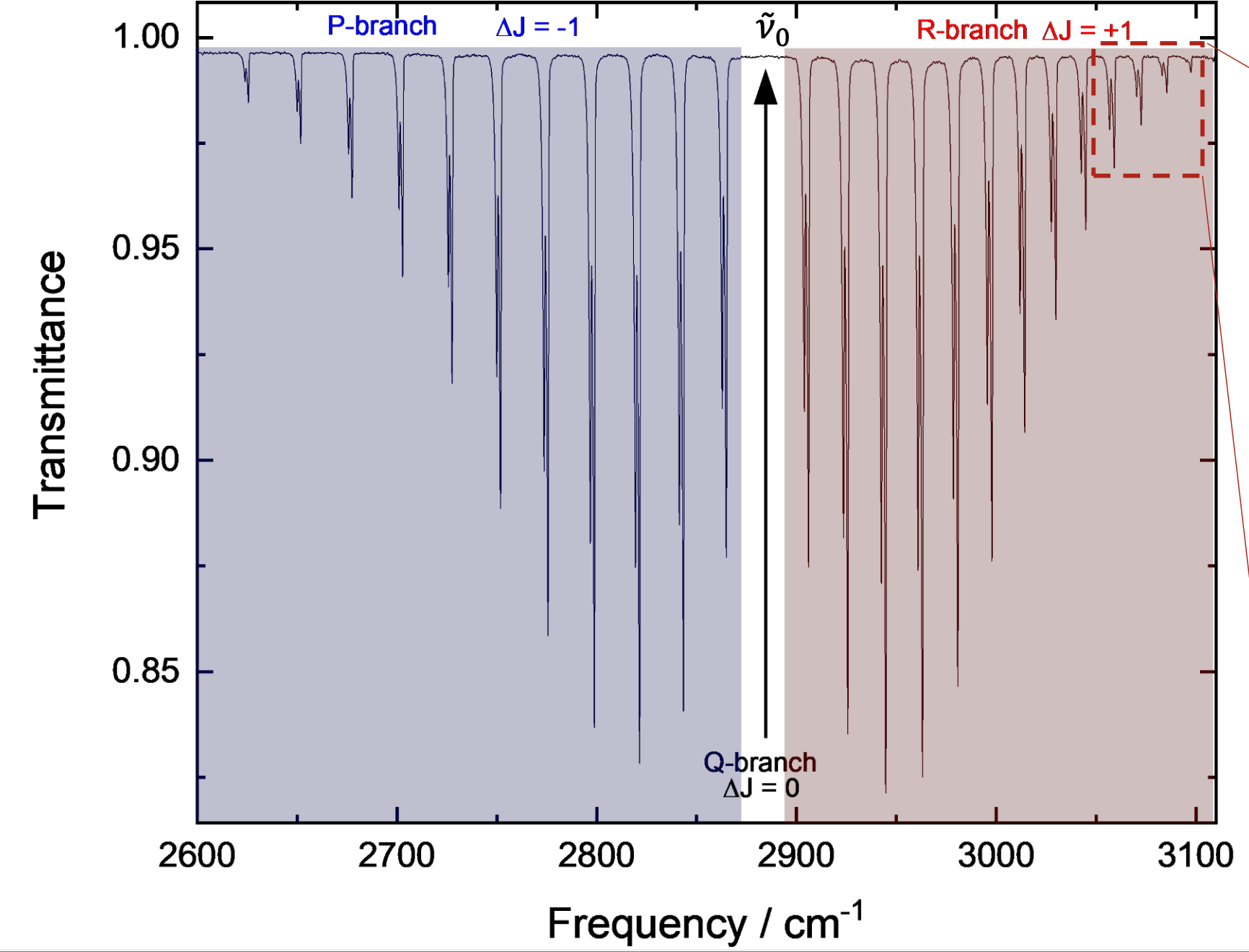
why is there a gap for the Q branch for H-Cl?
a ΔJ=0 is not allowed in a diatomic molecule
Q branch is not allowed
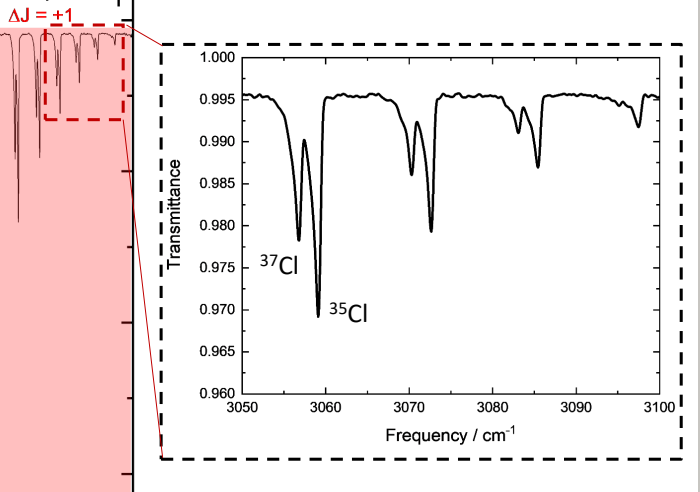
why are there two peaks for each peak of H-Cl?
isotopes
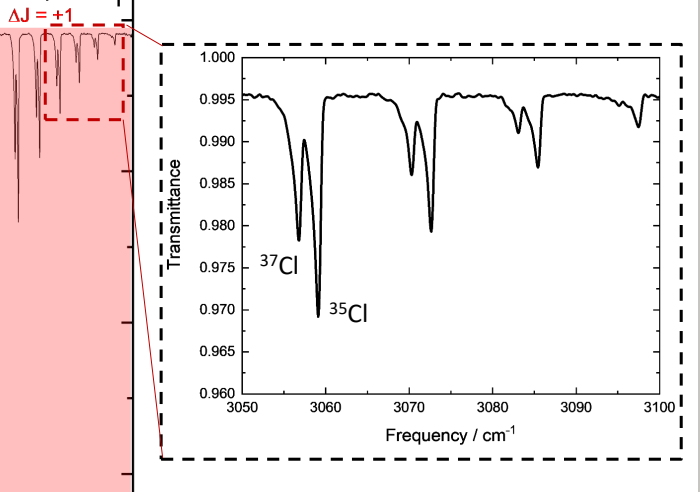
why is there no D-Cl peaks if both Cl isotopes are seen?
governed by reduced mass and DCl would shift and no longer be part of spectrum
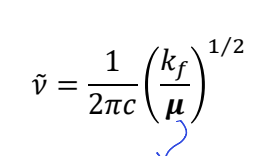

why do they have different rotational behaviour?
H2O vs NO2
rotational behaviour depends on masses
NO2 - all masses essentially the same so operates like prolate top (long and thin)
water heavily distorted as mass is different
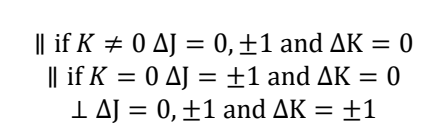
what does this mean?
if dipole moment of vibration is perpendicular to principal rotational axis, then the bottom selection rules apply
why might a Q peak not be present?
if the dipole change is small
how to work out how many times a diatomic molecule vibrates in one rotation?
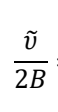
show harmonic oscillator vs morse potential
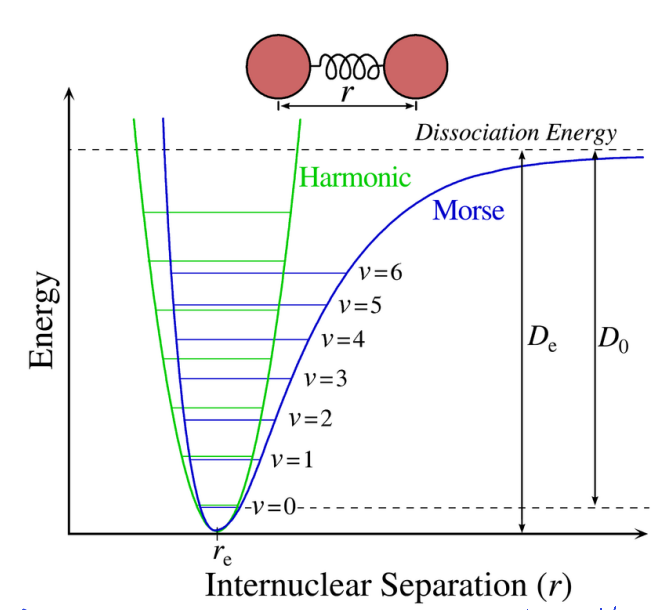
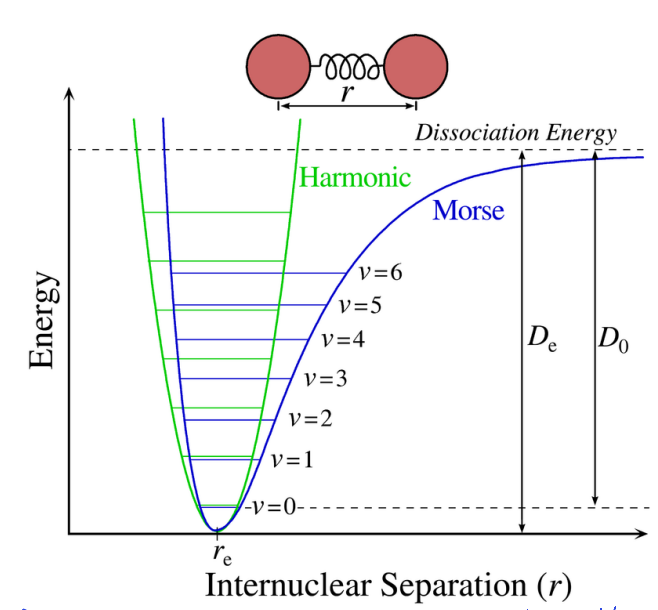
explain difference in bond lengths with vibrational levels and how this affects rotation
if in different vibrational EL, it would experience different bond length
rotation sees average bond length, but vibrational is faster so sees different bond lengths
each vibrational level has slightly different rotational constant
different moment of inertia (related to bond length)
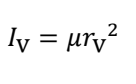
what is a centrifugal effect? how does this change with speed?
pushes bonds outwards
spins faster, bond lengths stretch out
distorts shape of molecule
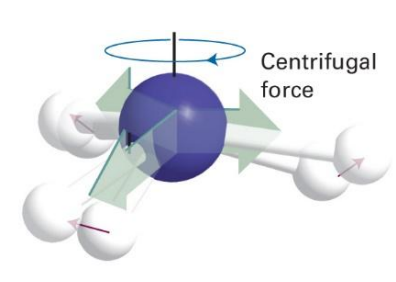
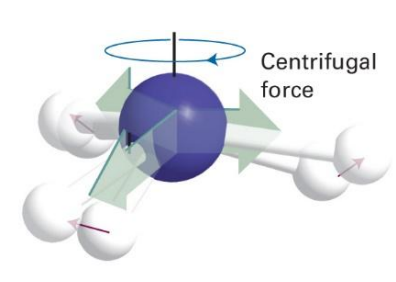
how can this be accounted for?
increase moment of inertia and hence decrease rotational constant
when are combination bands observed?
when 2 or more fundamental vibrations are excited simultaneously
what are overtones?
where vibrational mode changes by more than one
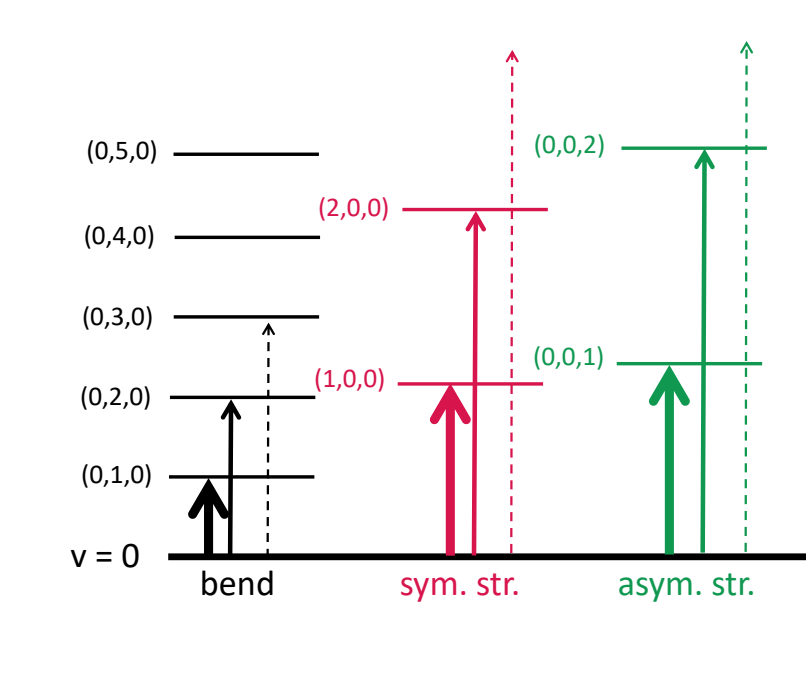
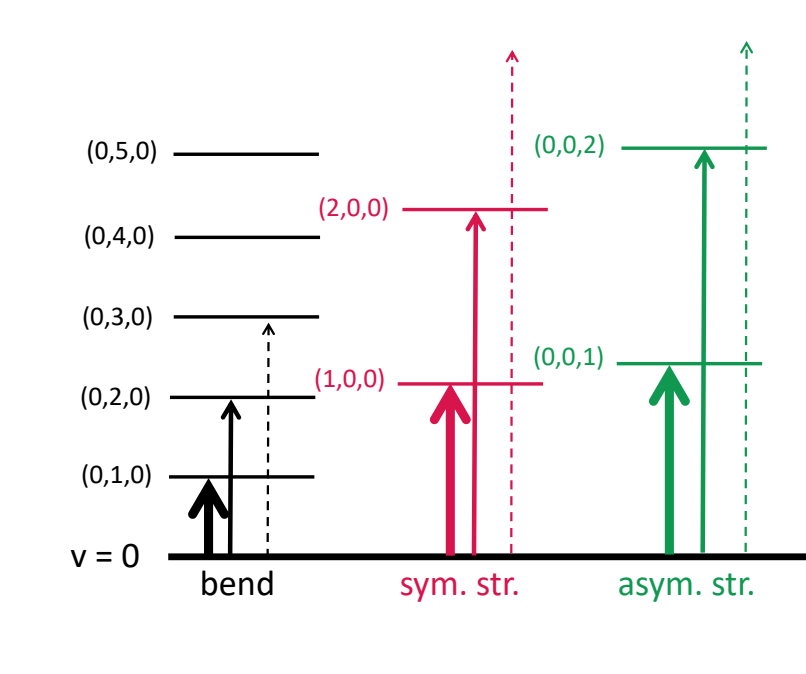
why are overtones less intense? what frequency are they at?
probability is lower
roughly twice the frequencies
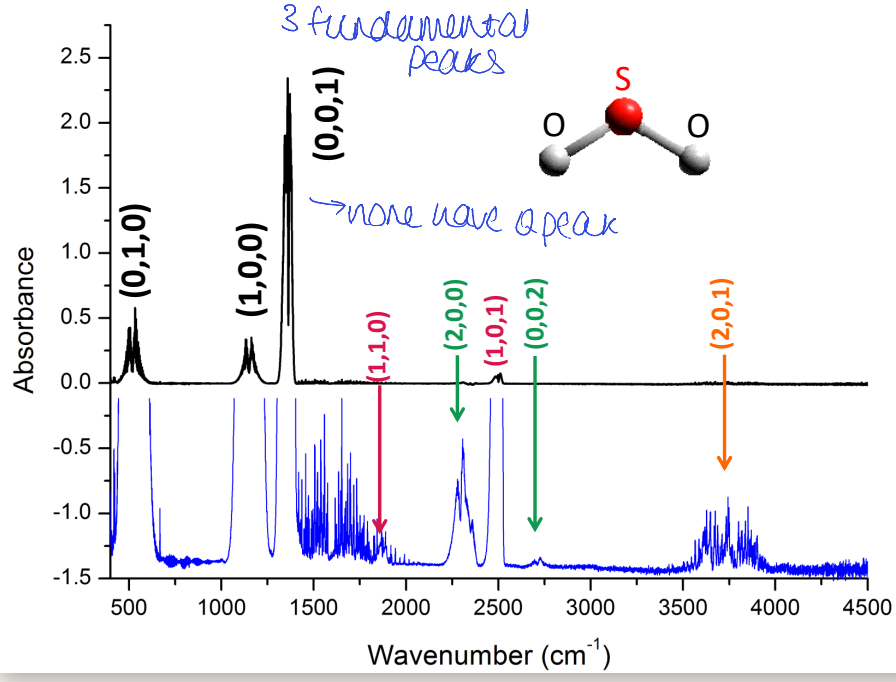
what is intensity of combination bands related to (orange)?
change in dipole
population of vibrational states
complex rules based around overlap of wavefunctions
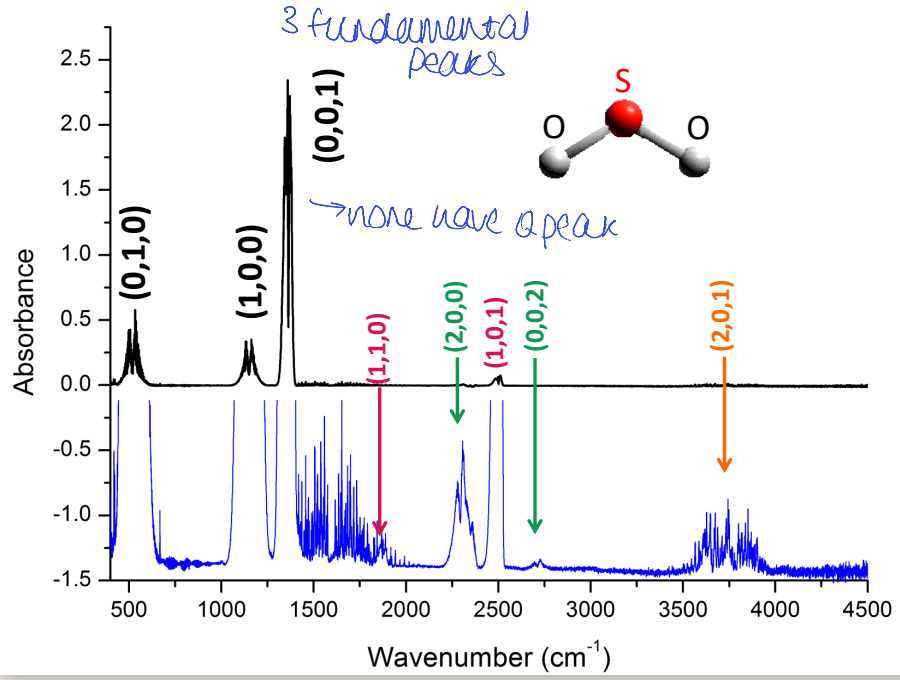
why do some overtones/combinations have Q peak when fundamentals do not?
changed selection rules so allowed to have Q peak