MCAT Chem
1/102
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
103 Terms
Photon energy
hf = hc / wavelenght
Lewis Base
A chemical species that donates a pair of electrons
Lewis Acid
A chemical species that accepts a pair of electrons
Keq < 1
favors reactants
+ Delta G
nonspontaneous
Keq = 0
nothing favored; equal
0 = delta G
at equilibrium
Keq > 1
favors products
- delta G
spontaneous
Ionic bond
A bond that results from electrons being transferred completely from one atom to another
Covalent bond
A bond in which both atoms contribute electrons to be shared between the nuclei of the two atoms an electron coming from both atoms
Coordinate ionic bond
Doesn’t exist.
Coordinate covalent bond
A type of covalent bond where the electrons shared comes from one atom
Coordinate number
The total number of atoms, ions, or molecules directly bonded to a central atom
Ionization energy
The minimum energy required to remove the most loosely bound electron. Trend: increases right and up.
Henderson Hasselbalch Equation
pH = pKa + log ([A-]/[HA])
Hess’s Law
Delta H (enthalpy) = sum of delta Hf products - sum of delta Hf reactants
Ideal gas law
PV=nRT
ideal gas constant R
R = 8.314 J / mol
R = 0.082 L atm / mol K
Ideal molar volume for ideal gases
22.4 L/mol
Strong Acids
HCl
HBr
HI
HClO4
HNO3
H2SO4
Strong Bases
LiOH
NaOH
KOH
RbOH
CsOH
Ca(OH)2
Sr(OH)2
Ba(OH)2
Ka and Kb relation to Kw
Ka x Kb = Kw
Relationship between Ka and pKa
pKa = - log (Ka)
pH equation for acid concentration
pH = -log [H3O+]
pOH equation of base concentration
pOH = -log [OH-]
Equation to find Ka
Ka = [H3O+] [ A-] / [HA]
Equation to find Kb
Kb = [BH+] [OH-] / [B]
Gamma Decay
No change in atomic number. Radiation is emitted through gamma rays like high energy photons when the nucleus moves from a higher energy state to a lower energy state
Alpha Decay
A He particle of neutrons and protons is removed from the nucleus as radiation called the alpha particle. The result is a daughter nucleus with an atomic number decreased by 2
Beta minus decay
Releases an electron and increaes atomic number by 1
Beta plus decay
Releases a positron and decreases the atomic number by 1
Oxidation # Rules
Free elements (H2,O2,ect) = 0
Hydrogen = +1
Group 1 = +1
Group 2 = +2
Metals are dependent on the other atoms
Group 17 / halgoens = -1
Oxygen = -2 (with few acceptions)
What happens if a molecules oxidation number goes up
It gets oxidized and it is the reducing agent
What happens if a molecules oxidation number goes down
It gets reduced and is the oxidation agent
What happens if there is no change in the oxidation number
No redox reaction occured
Electron affinity trend
Increases to the right and up
What happens to boiling point when pressure is decreased
boiling point decreases
Avagadro’s Number
6.02 × 10²3
Inductive effect
The shifting of electrons from less electronegative atom towards more electronegative atoms
Absolute configuration the amino acids
Most AAs S & L. Cystene = S & R. Glycine = no stereocenter
SN1 rxn facts
substrate only in the rate
3 > 2 > 1
weak nucleuophile
polar protic solution
sterochem = racemic
SN2 rxn facts
substrate & nucleophile rate dependent
1 > 2 > 3 (no rxn)
strong nu ( - charge)
polar aprotic (DMSO, acatone)
inverted sterochem
Charge of amino acids at pH 2
+1 cation —> N terminus = +, C terminus = 0
Charge of amino acids at neutral pH
0 zwitterion —> N terminus = +, C terminus = -
Charge of amino acid at pH 9
-1 anion —> N terminus = 0, C terminus = -
Isomerization
The process in which a molecule is transformed into an isomer with a different chemical structure (but same chemical makeup) ex: enolization, tautomerization
Electron Capture
Type of beta decay where a proton captures an electron near the nucelus and converts into a neutron without a positiron or electron emission and decreases atomic number
Polarizability
The extent to which an electron cloud of an atom can be distorted by an external charge or by an applied electric field to produce a dipole. Increases down and to the left
Electronegativity
The tendency of an atom to attract electrons within a bond. Increases up and to the right
Electron affinity
The tendency of an atom to accept an additional electron by measuring the energy change when an electron is added to an atom. increases right and up
Ionization energy
Measures the energy required to remove an electron from an atom. increases right and up
Standard temperature and pressure (STP) conditions
standard pressure = 1 atm
standard temp = 273K or 0C
1 mole of any gas at STP has a volume of 22.4L
Qualifications to be arromatic
Conjugated pi bonds in a cyclic structure
Unhybridized p orbitals present in each atom
Plana geometry, forming a continuous ring of parallel, overlapping unhybridized p orbitals
4n+2 pi electrons (Huckels rule), where n is a non-negative integer
Dispersion
The phenomenon of different frequencies of light having slightly different refractive indicies
Heat of combustion
The measure of the energy released when the compound is combusted with oxygen. The amount of energy to break bonds within a molecule
How to find the energy of activation
The activated complex minus the energy of the reactants
What is aceylation?
The addition of an acyl group (CH3CO)
What group is alkali metals
group 1
what group is alkaline earth metals
group 2
Linear
2 Electron group
Bond angle 180
Trigonal Planar
3 Electron groups
Bond angle 120
Tetrahedral
4 Electron Groups
Bond Angle 109.5
Trigonal Bipyramidal
5 Electron Groups
Bond angles 90 and 120
Octahedral
6 Electron groups
Bond angle 90
What is -CHO the condensed form for
Aldehyde
OH IR peak
3300 cm
Carboxylic acid peak
3000 cm
Amide / N-H peak
3300 cm
Carbonyl peak
1750 cm
sp3 carbons NMR
0 to 3 ppm
sp2 carbons NMR
4.6 6o 6.0 ppm
sp carbons NMR
2 to 3 ppm
Aldehyde NMR
9 to 10 ppm
Carboxylic acids NMR
10.5-12 ppm
Aromatics NMR
6.0 - 8.5 ppm
Absorbance and concentration relationship (if path length and wavelength are constant)
A1/C1=A2/C2
London Dispersion forces
Weak, temporary intermolecular forces are present in all atoms and molecules cause by random electron motion forming instantaneous dipoles
Critical Point
The end point of the liquid-gas coexistence curve representing the specific temp and pressure above which distinct liquid and gas phases exist
Sublimation
Solid to gas
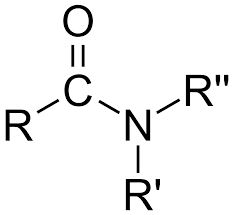
What is it?
Amide

What is it?
Amine
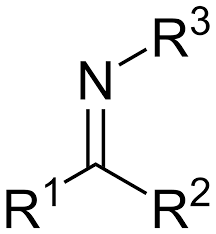
What is it?
Imine
What is it?
Acyl Halide
What is it?
Alcohol
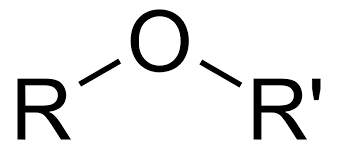
What is it?
Ether
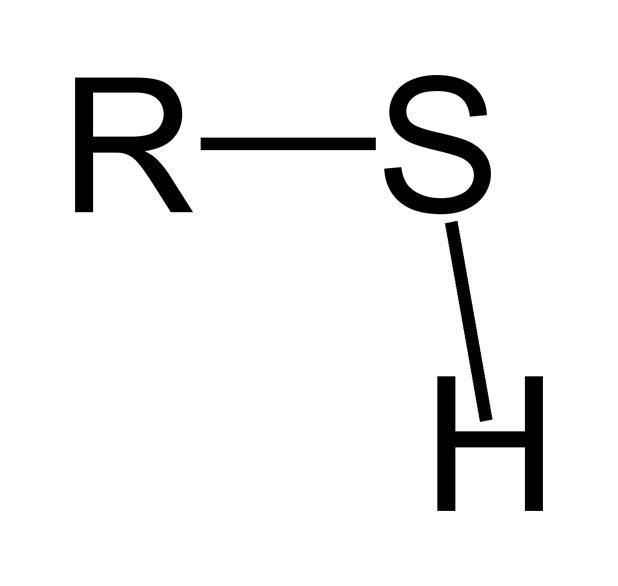
What is it?
Thiol
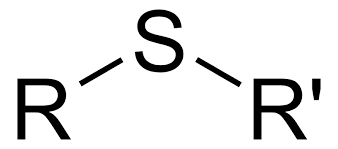
What is it?
Sulfide
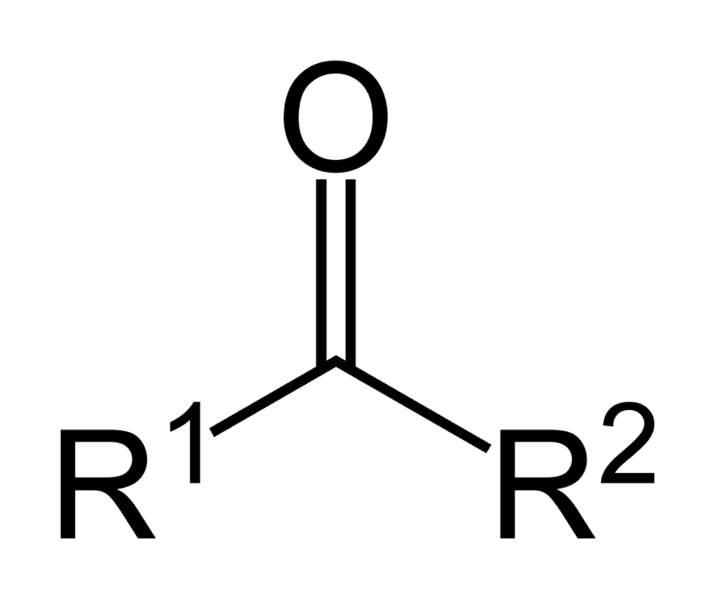
What is it?
Ketone
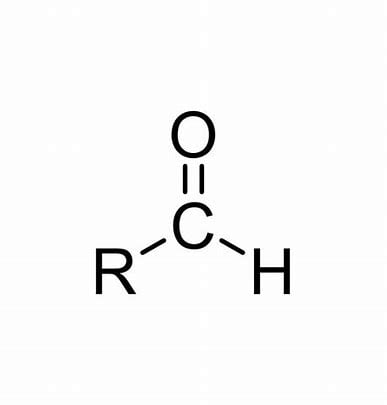
What is it?
Aldehyde
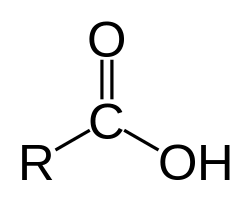
What is it?
Carboxylic Acid
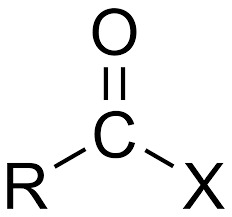
What is it?
Acyl Halide
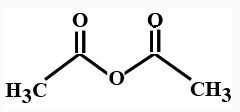
What is it
Anhydride
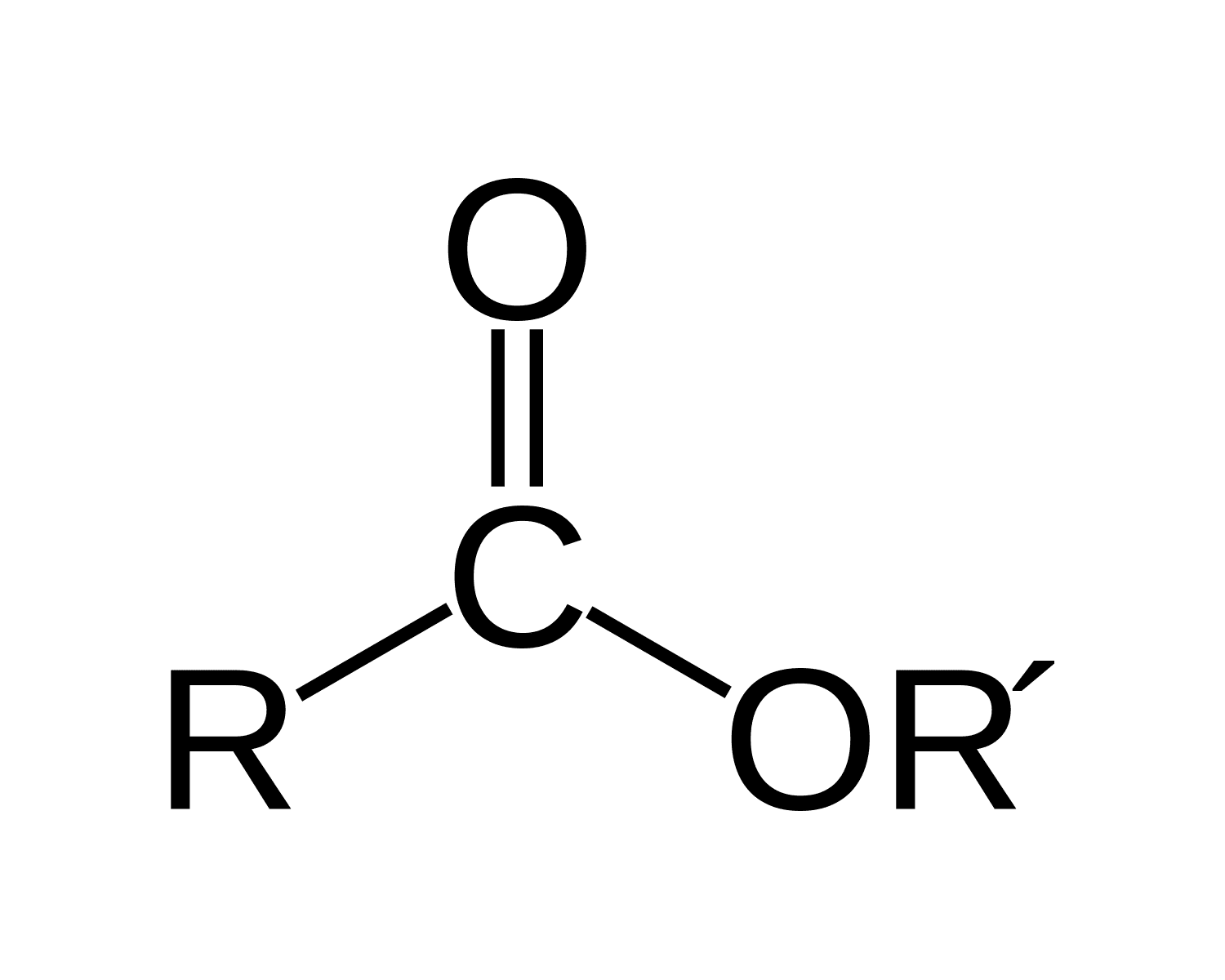
What is it?
Ester
Important oxidizing reagants
PCC, CrO3, CrO3/H2SO4 (Jone’s reagent)
Importat reducing agents
LiAlH4, NaBH4
What makes a good nucleophile
Charge: negative charge
Electronegativity: low electronegativity
increases right and down
What makes a good electrophile
Charge: positive charge
Electronegativity: high electronegativity
increases left and up
Ether layer
Non polar product
Aqueous layer
Polar product
What is true about resistors and capacitors in parallel?
They share the same voltage
How to add resistors in parallel
Use the inverse
How to add resistors in series
Use the normal sum