MMET Exam 3
0.0(0)
Studied by 10 peopleCard Sorting
1/147
Earn XP
Description and Tags
Last updated 1:10 AM on 11/30/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
148 Terms
1
New cards
Electrochemical reaction
Chemical reactions between electrons and ions.
-Producing and consuming electrons
-Producing and consuming electrons
2
New cards
Oxidation
-Produces an electron
-Loses an electron
-Happens at anodes
-Loses an electron
-Happens at anodes
3
New cards
Reduction
-Consumes an electron
-Gains an electron
-Happens at cathodes
-Gains an electron
-Happens at cathodes
4
New cards
Oxidation
Fe -->Fe^+2 + 2e-
5
New cards
Reduction
2^H+ + 2e- --> H2
6
New cards
Cathode/anode/electrolyte
For an electrochemical reaction, _______ and an _____ and an ___________ is needed.
7
New cards
EMF Series
Direction of reaction and potential determined by thermodynamics
-More anodic (bottom) will corrode more than those that are cathodic (top)
-More anodic (bottom) will corrode more than those that are cathodic (top)
8
New cards
Galvanic Series
Ranks metals and alloys based on their nobility, or how cathodic or anodic they are.
-Electrolyte used is seawater
-Top is more anodic while bottom is more cathodic
-Electrolyte used is seawater
-Top is more anodic while bottom is more cathodic
9
New cards
Factors affecting corrosion
1. Materials properties
2. Environment
2. Environment
10
New cards
Material properties
-Redox potential
-Passivity
-Metallurgical factors
-Passivity
-Metallurgical factors
11
New cards
Redox potential
Is corrosion thermodynamically favorable?
12
New cards
Passivity
Formation of a protective film
13
New cards
Metallurgical factors
When an anodic area is formed
-Chemical segregation
-Presence of multiple phases
-Inclusions
-Cold work
-Nonuniform stresses
-Chemical segregation
-Presence of multiple phases
-Inclusions
-Cold work
-Nonuniform stresses
14
New cards
Environment
-Chemical nature
-Operating conditions
-Polarization
-Operating conditions
-Polarization
15
New cards
Operating conditions
-Intended service life
-Temperature
-Velocity of corrodent
-Concentration of solution
-Impurities in solution
-Aeration
-Temperature
-Velocity of corrodent
-Concentration of solution
-Impurities in solution
-Aeration
16
New cards
Polarization
The change in corrosion potential with the change in corrosion current
-An issue in batteries
-An issue in batteries
17
New cards
Types of corrosion
-Uniform
-Pitting
-Crevice
-Galvanic
-Stress corrosion cracking
-Intergranular attack
-Dealloying
-Erosion
-Pitting
-Crevice
-Galvanic
-Stress corrosion cracking
-Intergranular attack
-Dealloying
-Erosion
18
New cards
Uniform
All surface area is exposed to the corrodent (rust)
-Prevented by material choice and removal of electrolyte
-Prevented by material choice and removal of electrolyte
19
New cards
Pitting
Local corrosion damage
-Prevented by se available data on corrosion
-Prevented by se available data on corrosion
20
New cards
Crevice
Local attack in a crevice
-Prevented by good gasketing
-Prevented by good gasketing
21
New cards
Galvanic
When 2 dissimilar metals are connected and one is corroded and consumed to save the other one
-Prevent by using insulating bush in between, don't let them touch
-Prevent by using insulating bush in between, don't let them touch
22
New cards
Stress corrosion cracking
Spontaneous corrosion induced cracking of a material under static stress (environmentally assisted cracking)
23
New cards
Intergranular attack
Preferential corrosion at the grain boundaries.
24
New cards
Dealloying
One element in the alloy is preferentially removed
25
New cards
Erosion
• Liquid impact
• Liquid erosion
• Slurry erosion
• Cavitation erosion
• Liquid erosion
• Slurry erosion
• Cavitation erosion
26
New cards
Liquid impact
Protective film removal
Can be combined with mechanical material removal
Can be combined with mechanical material removal
27
New cards
Liquid Erosion
Like liquid impact, but here fluid is parallel to the surface
-Minimized by lowering fluid speed
-Minimized by lowering fluid speed
28
New cards
Slurry Erosion
Like liquid impact, but abrasive particles enhance it
-Ceramic or elastomer coatings can be helpful.
-Ceramic or elastomer coatings can be helpful.
29
New cards
Cavitation corrosion
Formed when the operational pressure is dropped below the vapor pressure of the fluid
-Causes the formation of gas bubbles that collapse at an increased velocity on the surface of the material
-Induces initial cavitation.
-Causes the formation of gas bubbles that collapse at an increased velocity on the surface of the material
-Induces initial cavitation.
30
New cards
Corrosion data
-Corrodent chemistry
-Corrodent concentration
-Temperature
-Aeration
-State of stress
-Corrodent concentration
-Temperature
-Aeration
-State of stress
31
New cards
Corrosion characteristics
1.Corrosion Data
2.Standardized lab experiments
2.Standardized lab experiments
32
New cards
Uniform and Pitting ASTM
Use immersion corrosion
33
New cards
Dissimilar metals ASTM
Galvanic attack
34
New cards
Crevice ASTM
Rubber band around sample put in liquid
35
New cards
Stress corrosion cracking ASTM
Introduce stress, observe the arc portion
36
New cards
Liquid Corrosion
See effect of velocity on corrosion rate of metals (determined by weight loss)
37
New cards
Corrosion control
1.Material Selection
2.Environmental control
3.Design
2.Environmental control
3.Design
38
New cards
Material Selection
-Coatings
-Cladding: Cold working one material onto another material
-Heat treatment
-Diffusion Treatment: Introduce chromium to steel
-Corrosion data
-Surface finish:
-Cladding: Cold working one material onto another material
-Heat treatment
-Diffusion Treatment: Introduce chromium to steel
-Corrosion data
-Surface finish:
39
New cards
True
The rougher the surface the faster the corrosion rate
40
New cards
Environmental Control
• Temperature
• Velocity
• pH: dissolved gasses
• Cleaning: can prevent buildups that lead to crevice or concentration corrosion
• Inhibitors: alter the environment
• Velocity
• pH: dissolved gasses
• Cleaning: can prevent buildups that lead to crevice or concentration corrosion
• Inhibitors: alter the environment
41
New cards
Heat Treatment
Stainless steel after welding becomes sensitized, ____ __________ and subsequent quenching addresses it.
42
New cards
Inhibitors
-Remove oxygen (makes metal less corrodible)
-Absorpitive inhibitors: slows down anodic and cathodic reactions through a passive film
-Absorpitive inhibitors: slows down anodic and cathodic reactions through a passive film
43
New cards
Design
Corrosion happens (reduce/delay it w/ design)
-Design cleanability to avoid residue corrodent
-Provide proper drainage
-Design cleanability to avoid residue corrodent
-Provide proper drainage
44
New cards
Cathodic Protection
Use electrochemical reaction in our favor
-Use sacrificial anodes like Mg or Zn for steel applications (protects multiple metals)
-Use sacrificial anodes like Mg or Zn for steel applications (protects multiple metals)
45
New cards
Anodic Protection
Senses load and applied reverse to balance it out, thus no corrosion
-More complex than cathodic protection
-More complex than cathodic protection
46
New cards
Chromium
If you want to protect steel, add ________
47
New cards
Stainless steels
-Resist corrosion even at high temperatures
-Have at least 11% Cr
-Forms a passive layer in oxidizing env.
-Have at least 11% Cr
-Forms a passive layer in oxidizing env.
48
New cards
False
SS is well suited for reducing environments (e.g. sulfuric acid).
49
New cards
Manufacture stainless steel
-Adding ferrochromium to low carbon steel scrap
-Use electric furnaces
-Ladle treatments
-Argon-oxygen decarburization (AOD)
-Use electric furnaces
-Ladle treatments
-Argon-oxygen decarburization (AOD)
50
New cards
Oxygen/carbon
Manufacturing SS is challenging because Cr reacts with ______ and ______, so special processes are used.
51
New cards
Ni/C/N
Different alloying elements change the range
of stability of phases.
__, _ and _ extend the austenite region.
of stability of phases.
__, _ and _ extend the austenite region.
52
New cards
Microstructures of SS
1. Ferrite (BCC)
2. Martensitic (BCT)
3. Austenitic (FCC)
4. Precipitated hardness
5. Duplex
2. Martensitic (BCT)
3. Austenitic (FCC)
4. Precipitated hardness
5. Duplex
53
New cards
Ferritic Stainless steel
BCC Structure
Low carbon (
Low carbon (
54
New cards
Martensitic Stainless steel
BCT Structure
Chromium carbide is present in the structure
12-18% Cr (Alloys Fe/C/Cr)
Up to 1.2% C
Quench hardening (hardenable)
Poor weldability and notch sensitivity
Magnetic
Risk of stress corrosion
Low impact strength
Chromium carbide is present in the structure
12-18% Cr (Alloys Fe/C/Cr)
Up to 1.2% C
Quench hardening (hardenable)
Poor weldability and notch sensitivity
Magnetic
Risk of stress corrosion
Low impact strength
55
New cards
Austenitic Stainless steel
FCC Structure
-Fe, Cr (16-26%), C(lowest), Ni (8-24%)
Work hardenable
Non-magnetic
Risk of stress corrosion
Highest impact strength
High weldability
Ni is austenizer
-Fe, Cr (16-26%), C(lowest), Ni (8-24%)
Work hardenable
Non-magnetic
Risk of stress corrosion
Highest impact strength
High weldability
Ni is austenizer
56
New cards
True
Austenitic stainless steel is a metastable phase
57
New cards
False
Quench is not needed to maintain the FCC structure of austenite
58
New cards
SS Identification System
AISI: 3- digit system
1st letter shows composition
-200 = Cr, Ni, Mg
-300 = Cr, Ni
-400 = Cr
1st letter shows composition
-200 = Cr, Ni, Mg
-300 = Cr, Ni
-400 = Cr
59
New cards
Carbon/weldable
Lowering ______ makes them more ________
60
New cards
True
Molybdenum prevents pitting
61
New cards
What are ferritic SS used for
Non-structural and high temperature applications
62
New cards
What are martensitic SS used for
Structural applications and cutting tools
63
New cards
What are austenitic SS used for
Chemical and creep resistance, tanks, piping
64
New cards
Physical properties of SS
• Density: similar to other iron-based alloys
• Structure: affects mechanical properties and magnetism
• Conductivity: low electrical (one sixth of carbon steels) and thermal conductivity (less than half of carbon steels)
• Expansion: austenitic alloys can have 50% larger thermal expansion. Can be problematic in bimetal strips. Other structures are similar to carbon steels.
• Modulus of elasticity: slightly lower than carbon and alloy steels --> for the same section size SS has more elastic deformation
• Structure: affects mechanical properties and magnetism
• Conductivity: low electrical (one sixth of carbon steels) and thermal conductivity (less than half of carbon steels)
• Expansion: austenitic alloys can have 50% larger thermal expansion. Can be problematic in bimetal strips. Other structures are similar to carbon steels.
• Modulus of elasticity: slightly lower than carbon and alloy steels --> for the same section size SS has more elastic deformation
65
New cards
Forming
-Austenitic SS has high ductility --> no fracture in huge deformations
-Ferritic SS as a group are not as formable as carbon steels
-Ferritic SS as a group are not as formable as carbon steels
66
New cards
Machining
-If not modified (adding sulfur and phosphorous), much lower machinability compared to B1112
-Ferritics are gummy
-Austenitics tend to cold work
-Ferritics are gummy
-Austenitics tend to cold work
67
New cards
True
Corrosion resistance from chromium oxide layer
68
New cards
Pickling and Passivation
-To achieve the maximum corrosion resistance a uniform passive film is needed
-Pickling removes oxides (made from welding, heat treatment)
-For passivation nitric acid, phosphoric acid or citric acid is used
-Pickling removes oxides (made from welding, heat treatment)
-For passivation nitric acid, phosphoric acid or citric acid is used
69
New cards
Sensitization
Chromium carbides in austenitic SS can form at grain boundaries, making them susceptible to corrosion.
70
New cards
How to prevent sensitization
-Reduce exposure time to high temperatures
-Reducing carbon shrinks region of _____________, making them more weldable
-Reducing carbon shrinks region of _____________, making them more weldable
71
New cards
Heat treatment
-Ferritic: only annealing useful
-Austenitic: after annealing quenching is needed to prevent sensitization
-Austenitic: after annealing quenching is needed to prevent sensitization
72
New cards
Sensitization
Cb (Nb), Ta and Ti prevent ___________
73
New cards
Sulfur/selenium
______ and ________ lower corrosion resistance (but easier machining)
74
New cards
Limitations of SS
• Prone to pitting
• Best in oxidizing environments
• Susceptible to crevice corrosion
• Prone to attack in chloride and reducing acids (bleach solution, sea water, other Cl water)
• Some prone to stress corrosion cracking
• Susceptible to intergranular corrosion when sensitized
• Susceptible to galvanic corrosion between grains
• Best in oxidizing environments
• Susceptible to crevice corrosion
• Prone to attack in chloride and reducing acids (bleach solution, sea water, other Cl water)
• Some prone to stress corrosion cracking
• Susceptible to intergranular corrosion when sensitized
• Susceptible to galvanic corrosion between grains
75
New cards
Ferritic/Martensitic/Austenitic
-430(_______)
-416, 420, 400C (___________)
-302, 304, 316, 304L, 316L (__________)
-416, 420, 400C (___________)
-302, 304, 316, 304L, 316L (__________)
76
New cards
Cupola
Cast iron is often melted in a ______
77
New cards
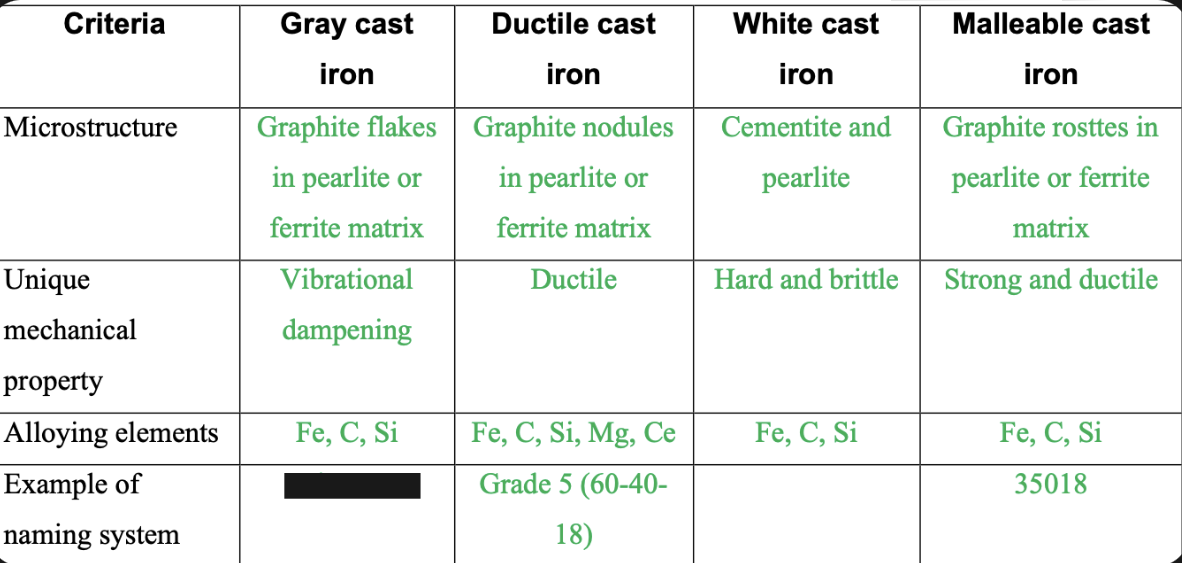
Cast Iron Types
1. Gray- General Purpose
2. Malleable- Heat treated for ductility
3. White- Hard and wear resistant
4. Ductile- Some ductility as cast
2. Malleable- Heat treated for ductility
3. White- Hard and wear resistant
4. Ductile- Some ductility as cast
78
New cards
Fluidity/cast
Silicon in cast iron increases ________, making it easier to ____
79
New cards
True
Cast iron has b/w 2% and 4% carbon
80
New cards
Graphite flakes in pearlite or ferrite matrix
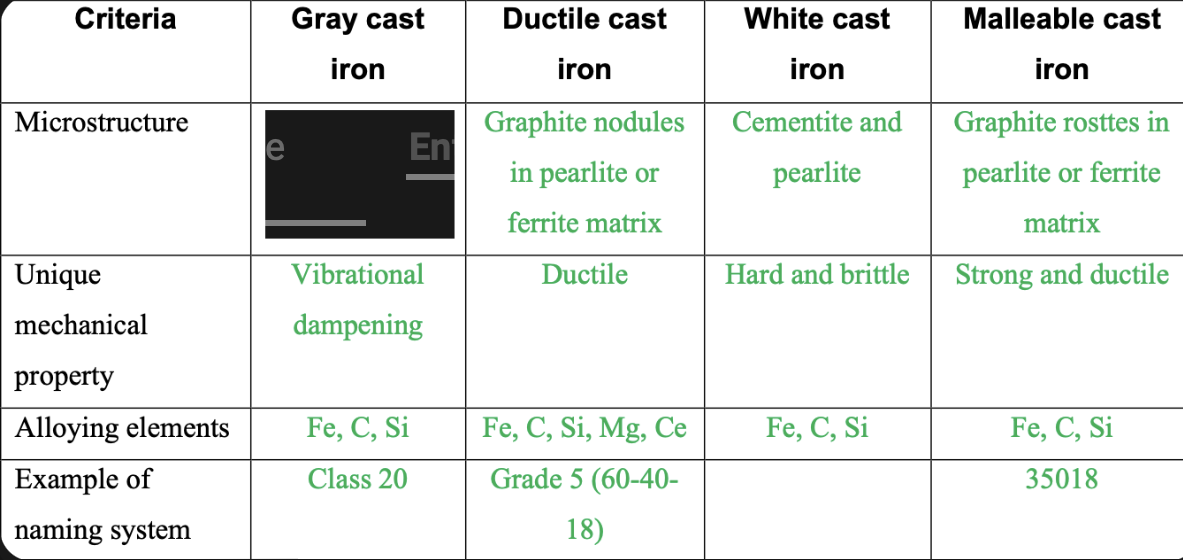
81
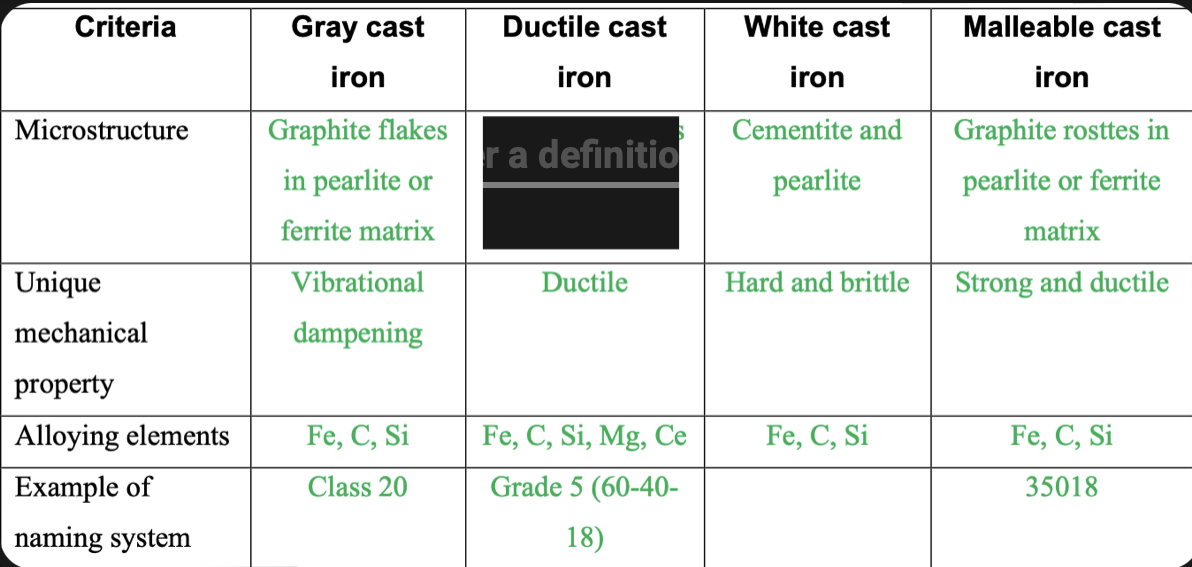
New cards
Graphite nodules in pearlite or ferrite matrix
82
New cards
Cementite and pearlite
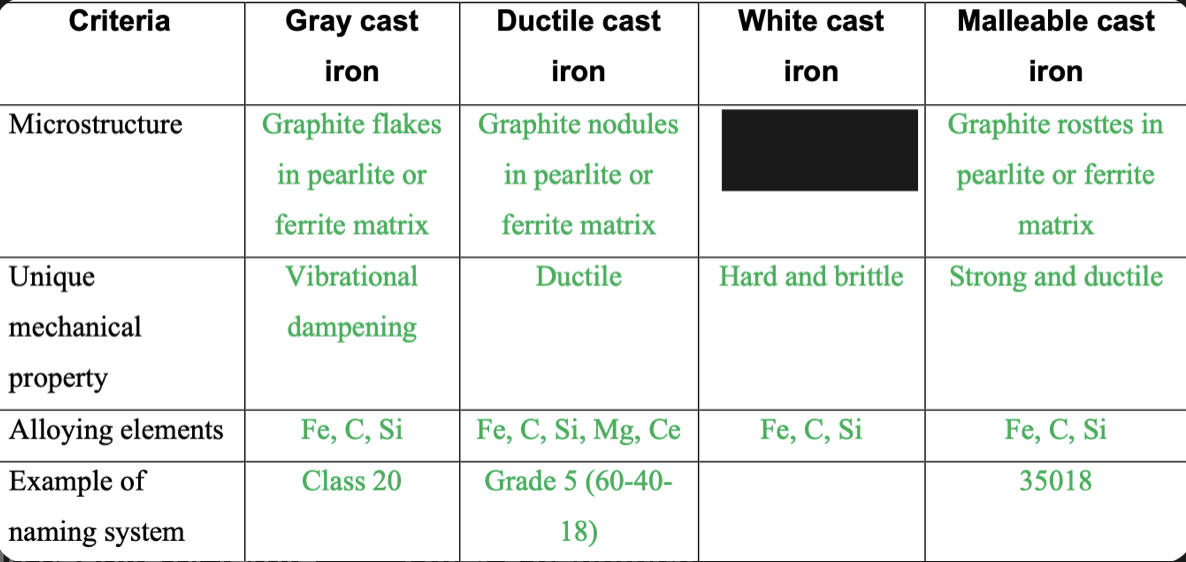
83
New cards
Graphite rosettes in pearlite or ferrite matrix
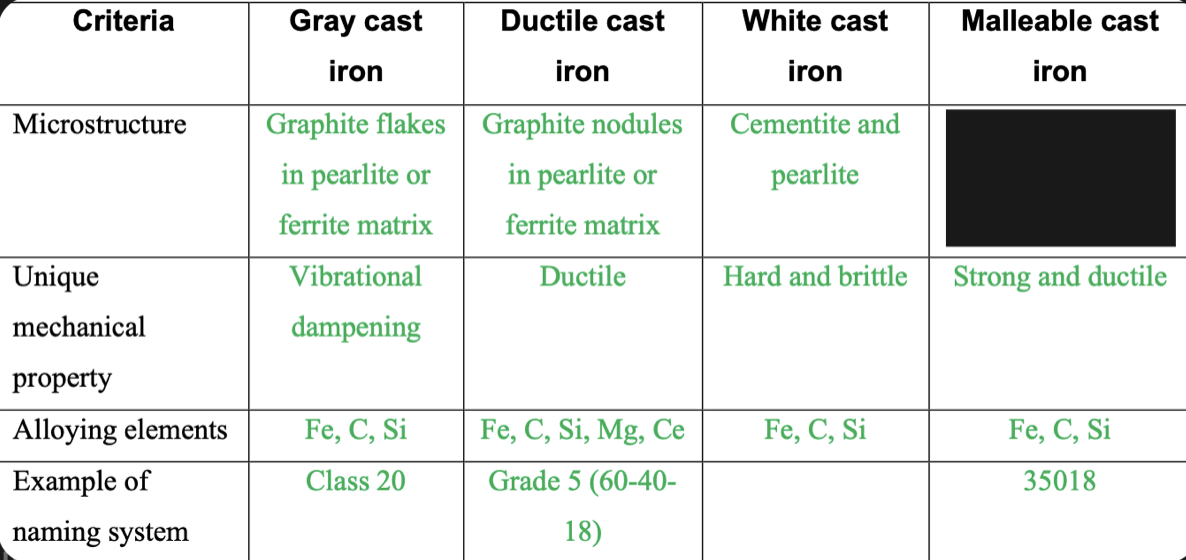
84
New cards
Vibrational dampening
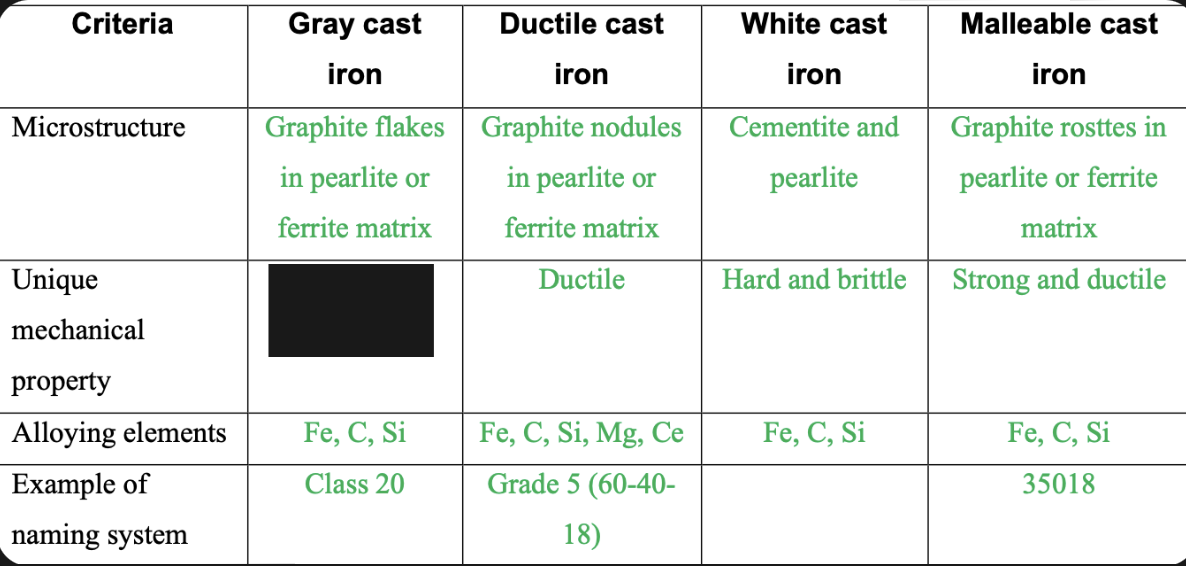
85
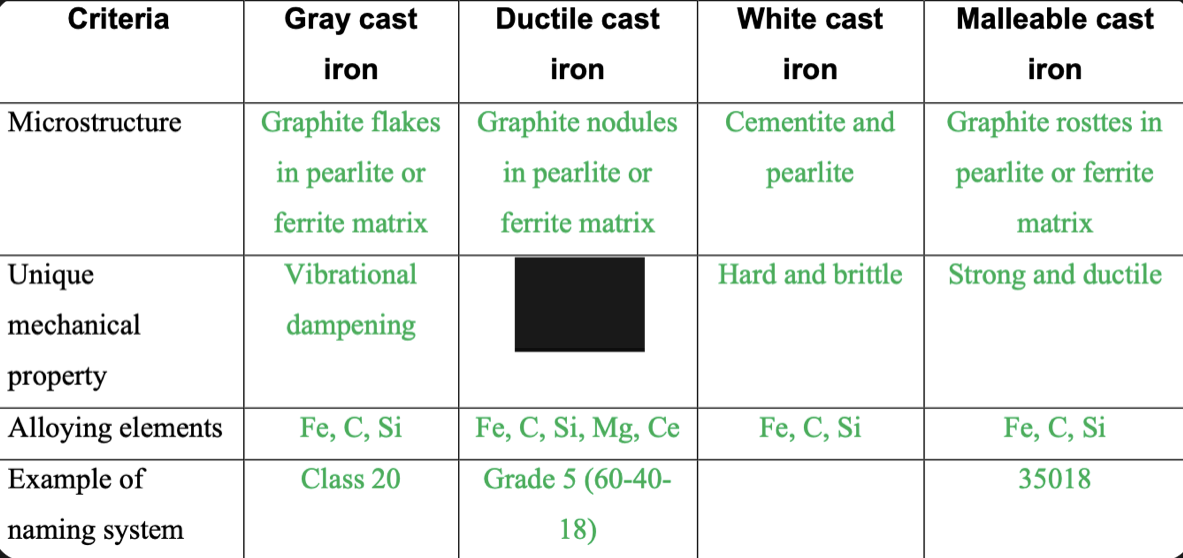
New cards
Ductile
86
New cards
Hard and brittle
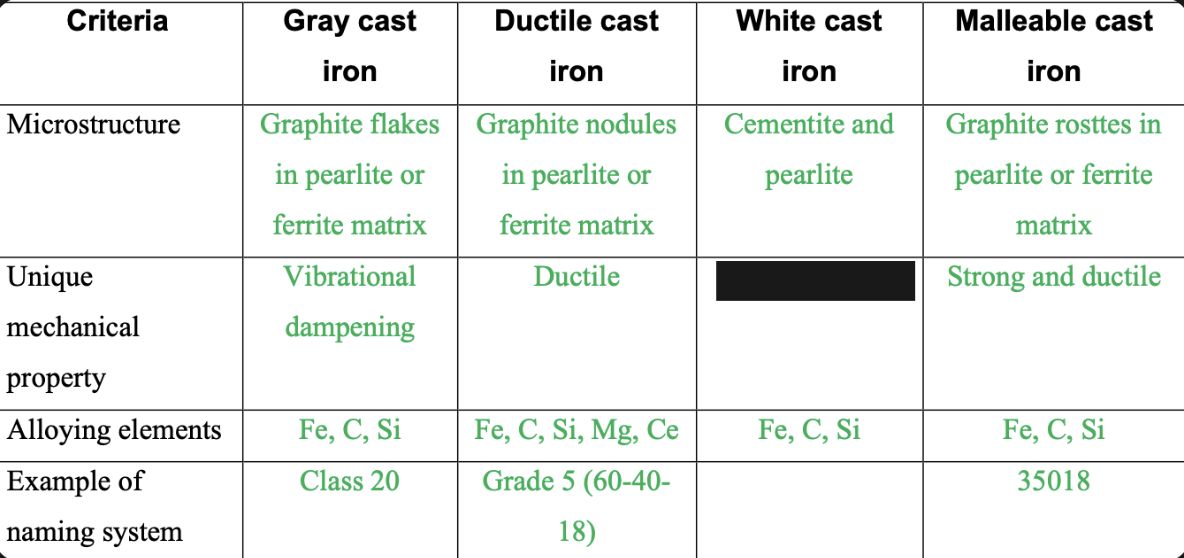
87
New cards
Strong and ductile
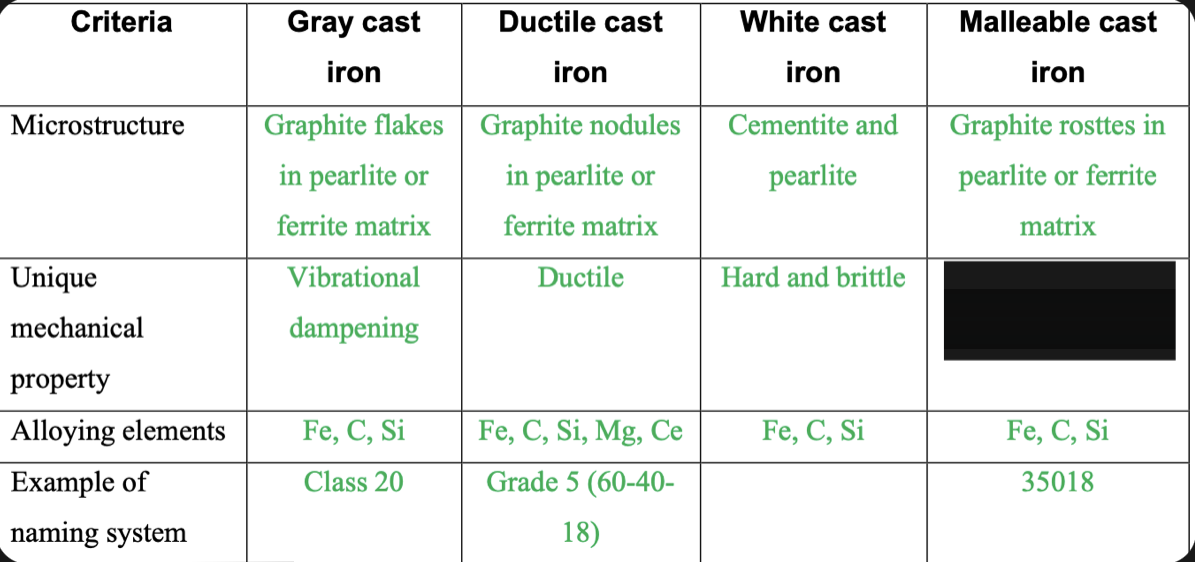
88
New cards
Fe,C,Si
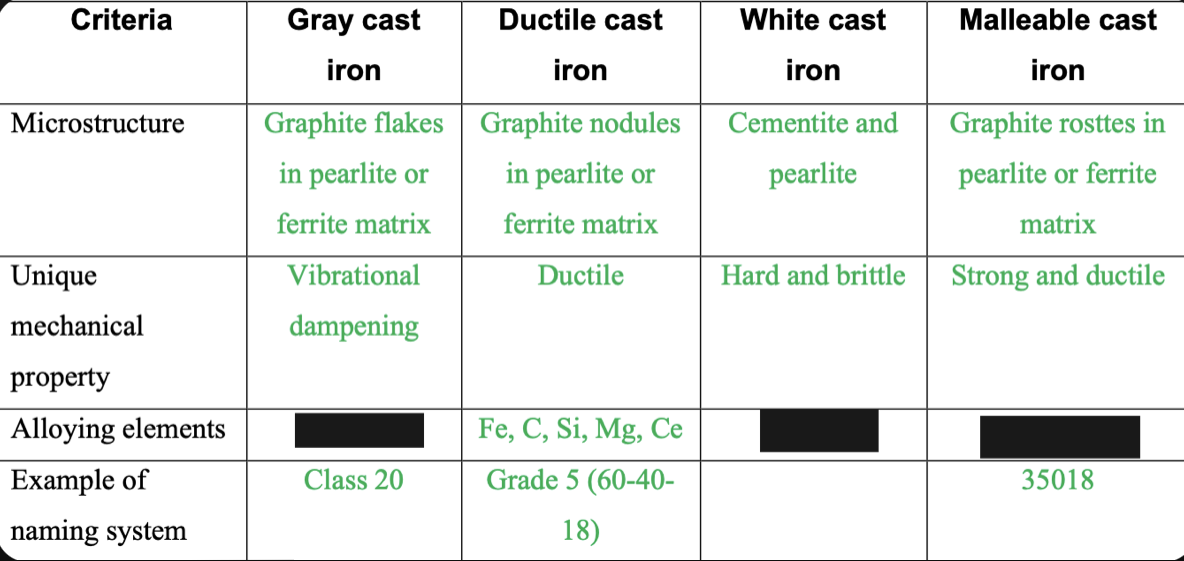
89
New cards
20/60/increase
• As increase from __ to __, the following ________:
-All strengths
-Quality of finish for machined surfaces
-Resistance to wear
-Modulus of elasticity
-All strengths
-Quality of finish for machined surfaces
-Resistance to wear
-Modulus of elasticity
90
New cards
20/60/decrease
As increase from __ to __, the following ________:
-The ability to dampen vibration
-Resistance to thermal shock
-Machinability
-Castability
-The ability to dampen vibration
-Resistance to thermal shock
-Machinability
-Castability
91
New cards
Class # (20-60)
92
New cards
Secant modulus
Need to use ______ _______ to fine Gray CI's modulus of elasticity
93
New cards
Numbering system for Ductile CI
-Grade number and properties
-Minimum Tensile strength in ksi
-Minimum Yield strength in ksi
-% elongation
Ex: Grade 5(60-40-18)
-Minimum Tensile strength in ksi
-Minimum Yield strength in ksi
-% elongation
Ex: Grade 5(60-40-18)
94
New cards
White
Fracture surface for white CI is _____
95
New cards
Numbering system for malleable CI
ASTM 47 with a 5 digit number
-32510:
-Minimum yield strength – 325
-% elongation – 10
-35018
-32510:
-Minimum yield strength – 325
-% elongation – 10
-35018
96
New cards
Malleable CI
Gotten from heat treating white CI (800-900C) for up to 30 hours
97
New cards
Modulus of elasticity
White CI has the highest _______ __ __________
98
New cards
Physical properties of Aluminum
-Low density →one third the weight of steel
-Good thermal and electrical conductivity
-High strength to weight ratio
-High reflectivity
-Good corrosion resistance (passive aluminum oxide layer)
-Not magnetic
-Good thermal and electrical conductivity
-High strength to weight ratio
-High reflectivity
-Good corrosion resistance (passive aluminum oxide layer)
-Not magnetic
99
New cards
Aluminum fabrication
-Easy to cast and machine
-Most alloys are weldable
-Can be given a hard surface by anodizing and hard coating
-Ductile
-Most alloys are weldable
-Can be given a hard surface by anodizing and hard coating
-Ductile
100
New cards
Aluminum
-Most abundant metal in nature (8% of earth’s crust)
-Nontoxic
-Nontoxic